Polarization
Diving into how atoms and dielectrics respond to electric fields — getting polarized, forming tiny dipole moments, and what that α actually means!
From here on we’re doing “Electrostatic fields inside matter.”
Up through chapter 3, we didn’t consider what body we’re inside, what kind of matter we’re inside, or anything like that, right. It was all vacuum, vacuum!
So for permittivity we just used epsilon-zero without a second thought.
From now on, we’re going to care about what kind of matter we’re inside.
When a charge is in vacuum, when it’s in water, or when it’s in just any kind of matter or any kind of gas?
The electric field in each case is different.
The matter’s response to that electric field is going to be different, right!!~
If the electric field is different, then of course the potential will be different too.
So now we’re gonna do this kinda stuff, yeah~
First, let’s just put one normal, neutral atom inside an electric field E. (We want to see the atom’s response to the electric field)
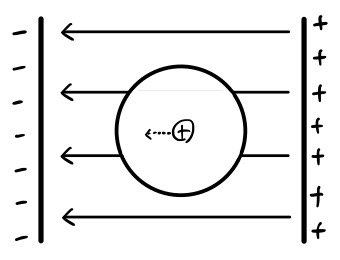
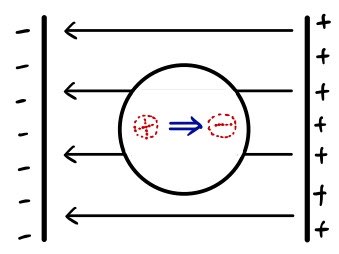
Because of the electric field E, the atom’s nucleus (positive charge) will get slight~~~ly pulled to the left, and the atom’s electron cloud (negative charge) will get slight~~~ly pulled to the right!!
Now because of these shifted charges, a new electric field will form inside!!!!
And as long as the external field E isn’t super-megaton huge, it will reach equilibrium.
This is what we call being “polarized.”
A polarized atom like this has a very tiny dipole moment (dipole moment p).
Naturally this p will be proportional to the external electric field E.
Say we put a variety of kinds of atoms into the same E. Will each of those atoms have the same p? Of course not.
For the same E, some will shift a bit more, some will shift a bit less.
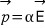
Written this way, α is called the “atomic polarizability.” This means we’re looking at the atom microscopically.
For this part, it’s enough to just know “ah, there’s something like this.”
In the classical model, we can’t look at atoms microscopically…
(Of course, if you invest 218371273981279 hours you could look at it microscopically, but~ what I mean is it has no meaning.)
Up the~re we put an atom into an electric field, so this time let’s put in a piece of dielectric made of “atoms.”
In the same way, the many atoms also get polarized, and the piece of dielectric as a whole gets polarized.
But now since we’re dealing with a ‘piece’ of dielectric, let’s define P.
P : the dipole moment contained in unit volume (Polarization: polarization density) ← this is also a vector, a vector
What we’re looking at now is: we shove it into an electric field and it gets polarized,
and then we’re going to remove the electric field again! Actually there are also materials where if you remove the electric field the polarization disappears, but for now we’ll assume it doesn’t disappear @@
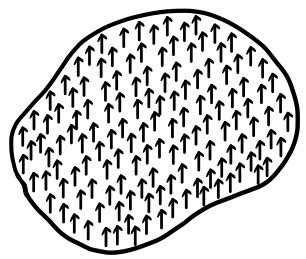
Let’s saaay it got polarized like this!
Then let’s set it up like this
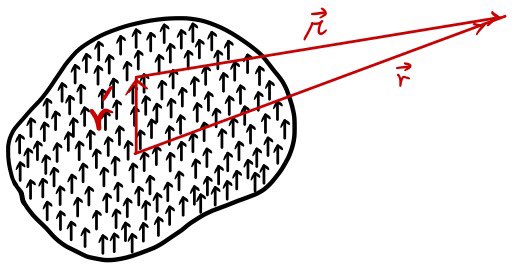
When we have a material polarized like this (dipole moment P contained in unit volume)
how much is the electric field created by this polarization density????
At a point r away from this body, what is the potential?!?!?!
First, the potential!
If we just sum up allll the potentials at r from every individual dipole, that oughtta do it~
First, the potential at r due to a single dipole (located at r’) is
V(r) = pr/4πε, so
summing up alll~ those potentials means
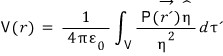
Let’s manipulate this equation.
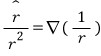
Let’s use this to transform the equation a bit. And since P(r’) is uniformly polarized, I’ll write it as P.

But here
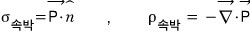
we have these relations. Using this!
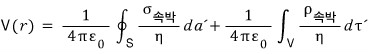
So when a body gets polarized, if you look inside the body, there will be -+-+-+-+-+-+-+-+-+-+-+-+-+ ~~~ all arranged like this.
You can think of these charges as being bound (held in place).
But then why is the bound surface charge density over there — why is it equal to the dot product of P with the surface-normal vector,
and why is the volume charge density inside the body the divergence of P;;; this….
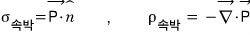
Honestly, it was realllly really hard for me too.
I reached my own conclusion and pushed on with the material, but it still feels pretty sloppy.
In the next post I’ll try to talk about that.
Originally written in Korean on my Naver blog (2014-11). Translated to English for gdpark.blog.