Rutherford Scattering and the Rutherford Scattering Formula
Deriving the Rutherford scattering formula while stumbling through the wild history of how we went from Thomson's pudding model to discovering the atomic nucleus — scary math incoming lol.
Ah, today while studying I got so sleepy that
I figured I’d post the piping hot Rutherford scattering formula I just studied.
Alright, let’s go!!!!!!
From a history-of-science perspective, I used to mistakenly think the structure of the atom had been figured out by around the 1600s? 1700s?
Around the time classical mechanics and electromagnetism were completed by Newton and Maxwell,
I was under the illusion that at least the structure of the atom had all been revealed.
But while reading this book called Quantum Story this time,
I learned that the rise of ‘atomism’ was in the late 1800s, and that Thomson’s discovery of the electron and Rutherford’s discovery of the atomic nucleus around this time
is what modern physics is — I learned that this time. (I probably knew this before too,,,, but this time I properly organized it….)
Actually, the reason Planck (a master of classical thermodynamics) jumped into blackbody radiation research was because he wanted to shut the atomists up, which is why he started that research,
(according to the atomists’ logic, an assumption that went into his proof of the second law of thermodynamics would be broken; that assumption was apparently “matter is continuous”.)
And then he somehow…..became the leader of the atomists, and also,,, threw open the doors of quantum mechanics wide open lololol it was fun hehehe
Anyway, in 1898 J.J Thomson discovered the electron and discovered that it carries a negative charge.
And he proposed that at least a few hundred negatively-charged electrons would be embedded in some positively-charged something, proposing the so-called ‘pudding model’.
That pudding model ended up being broken by the New Zealand physicist Rutherford.
Rutherford witnessed a strange phenomenon during his α-ray scattering experiment,
(α-rays are helium ions from a helium atom with two positive charges that have lost their outermost electrons, but here let’s just call them α particles! Back then they probably just thought it was a ray?)
“Hey hey hey hey hey!! The positively-charged particle isn’t spread out evenly like pudding!!!”
“It seems to be clumped up ttak!!! somewhere!!!!!” Let’s call this the nucleus!!! he said, and then he
“Turns out the nucleus has the same charge amount as the electron?!?!??!”
So Rutherford’s conclusion was to propose an “orbital model” for the atom,,,,,
(Rutherford probably knew too that the orbital model was wrong……hehe
But through the α-ray scattering experiment it’s clear it’s not the pudding model.,,, I think there just wasn’t really any other way…hehe
Now the world just waits for the next luck-blessed~~~~~~~ genius…!!)
The “Rutherford scattering formula” that Rutherford obtained after the α-ray scattering experiment is

From now on I’m going to derive this…………..haaa…………
I’m already scared lololololololololololololol
How do I actually do this lololololol aaargh!
Alright, let’s think about it like this…
From now on assume I’m channeling Rutherford
From now on the writer is Rutherford (Rutherford still thought it was the pudding model)
“Ohoho~~ pudding, you say? Then the α particles would all pass through~~ Assistant~~~~ fire the α rays~~~~~”
(Ting!~ting!ting!ting! Taeng!~taeng!taeng!taeng!) - the sound of α particles bouncing off
“What on earth is this!!!!!!! A frying pan game!!!!!!!!! Attendants!!!! Are you outside!!!!!!!!!!!!! Fetch me my pen and paper at once!!”
“This must surely mean there is some densely packed sardine that bounced off our powerful α particles, methinks.”
“Then let us assume the following”
“First, let us call that densely packed particle the nucleus, and since that nucleus is small and the α particle is also freaking small, let us think of them as point masses and point charges. And since they both carry positive charges, they will produce a repulsive force!!!!”
“My good fellow, tell me how this repulsive force arises”
(Yes my lord, I understand that the repulsive force has a coefficient of 1 over four-pi-epsilon-zero, and is inversely proportional to the square of the distance and proportional to the product of the charges…)
“Correct, it is the Coulomb force
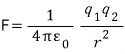
indeed”
“For now, let’s set aside the head-on collision and think about being scattered by the repulsive force when slightly off to the side. This is my assumption.”
“Also, I am inclined to think that the nucleus is very heavy compared to the α particle, because there are α particles that collide head-on and come back out. So I made the assumption that it is heavy, and therefore let us assume that during the time the repulsive force acts, the nucleus does not move.”
“If no one objects, behold this figure.”
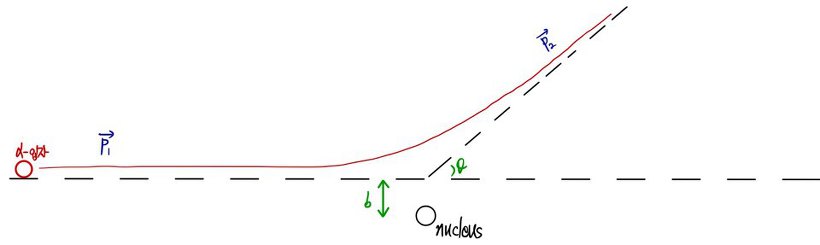
" This is a figure of the α particle being deflected by about b. "
" Watch carefully, what I am about to do is derive the relationship between that b and θ. "
" But since we assumed that the nucleus thing stays put, energy will be conserved. "
" Then what does that mean, the magnitude of the momentum p1 and p2 are equal. "
" The change in momentum is Δp=p2-p1, any objections "
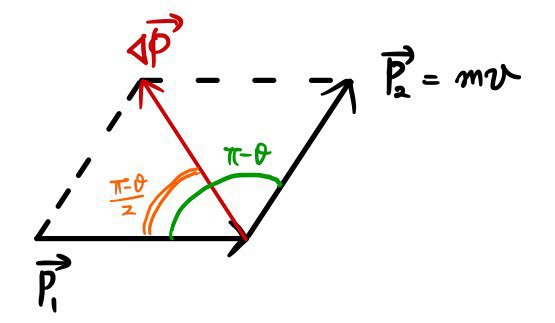
" Let us say the speed and mass of the α particle is mv "
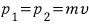
" Alright, first let me throw out a question. What caused that change in momentum (it’s a vector so there is a change) "
“The electric repulsive force, my lord”
" Yes, correct. Let us move on. "
" Alright then
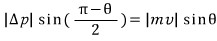
if you look at the figure this relation is obvious so we will move on “
" Alright, the sine function, if you shift it by pi over two, becomes cosine, and sin is an odd function, cos is an even function, massaging the equation with that gives "
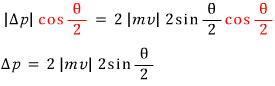
“We have derived the relation between delta p and theta. Now let us write delta p differently. "
" What caused delta p was the electric repulsive force as I said earlier,
I will draw that electric force at each position. (the blue is the direction of the delta p vector) "
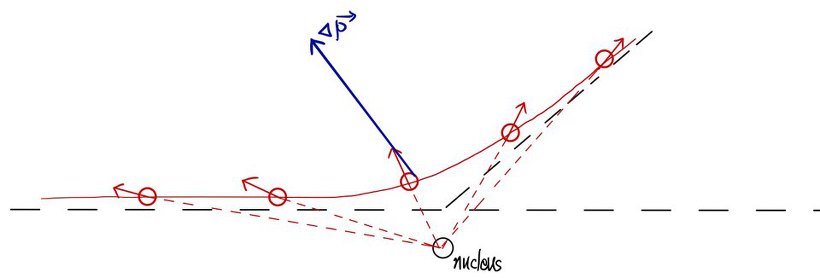
“Ohoho, these fellows face a headwind as they approach and a tailwind as they pass by, I see that’s how energy is conserved.
Alright then, the total sum of each F (vector) would have caused Δp (vector). "
Did you not learn this in 10th grade
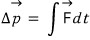
(it’s like dropping an egg on a cushion versus dropping it on asphalt…heh)
But let us be crafty, instead of summing all those F’s, let’s project the components only in the direction of Δp and sum those up.
That is,
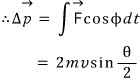
“Alright then, from where to where would it be reasonable to integrate?????? "
“Yes my lord, this humble servant thinks from -∞ to ∞ would be reasonable.”
" Correct. Even when α was really really far away, the electric force was just small but not zero, so as you say, let us do it that way”
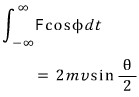
Alright… the integration variable is t
Who can integrate this!
Ugh T_T
Right, let’s change the integration variable to angle too…. Let’s use the chain rule
I will change it;;

" The reason I specifically did phi is because the rate of change with respect to time of phi can be described as circular motion, and I thought we could manipulate it that way, "
“Let us continue”
“Now now now, the magnitude of p is constantly constant….. then that means the angular momentum r × p is also constant. "
Alright, we can now witness the birth of b right here, watch carefully!!!
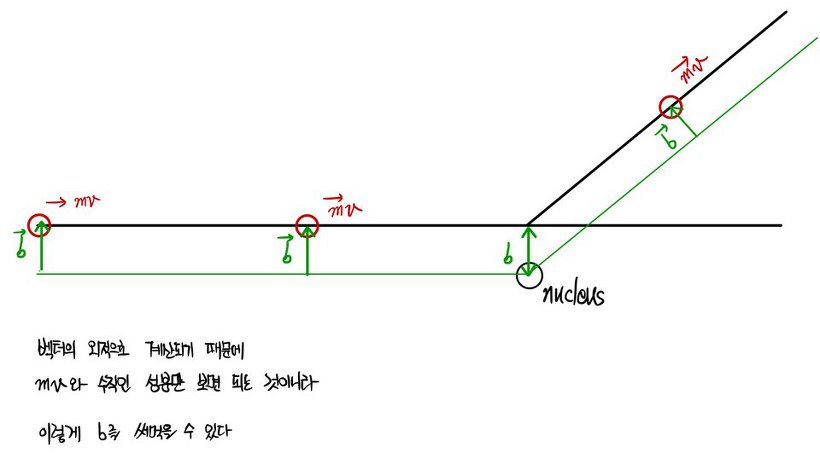
" Alright, angular momentum is constant……yes. "


“Let us plug it into our equation.”
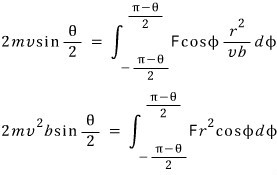
Ohoho
Right, now let us calculate the electric force between the α particle and the nucleus.
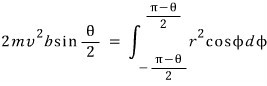
I will mash these equations together
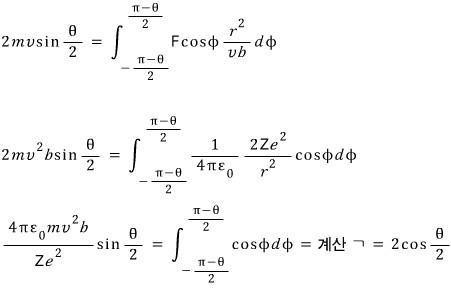
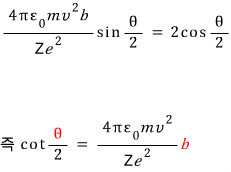
Alright, we have pulled out the relationship between b and theta, heh heh heh heh heh heh heh heh heh
Ah but let us clean up the equation just one more time
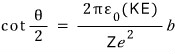
Done done. This is the equation I made with my assumptions..
Uh huh… my thinking was pretty much “Where could this thing called the nucleus be! >_<” and I built the equation like that,,,,,
Haaa~~~ how to use that equation experimentally is really a problem T_T T_T T_T T_T
First of all, many atoms together would form matter, and matter has thickness, doesn’t it T_T?
It would be hard to make it graze past just one atom T_T.T_T
How do I use this equation………
Alright!!!!!!!!!!!!!!!!!!!!!!!!!!!!!
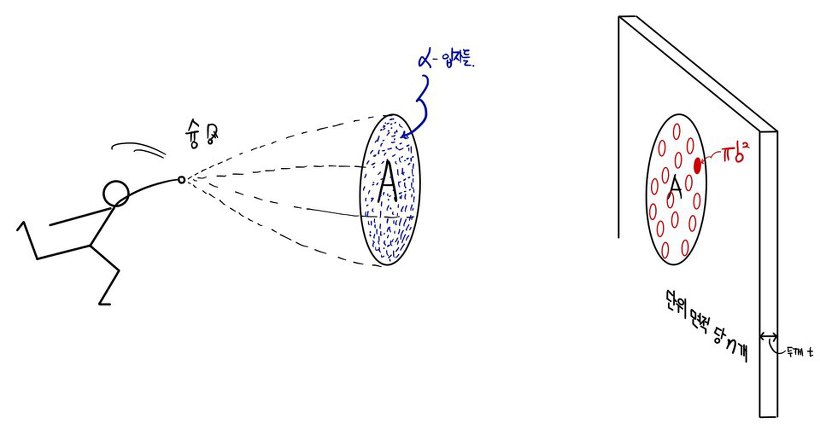
First, suppose an α particle coming in at spacing b gets scattered by theta,
then an α particle coming in at a smaller spacing than b will be deflected at an angle greater than theta.
That is, α particles incident within π(b.^2) will be deflected at angles greater than theta!!!!!
Okay, and, let’s say matter has n atoms per unit area,
and the thickness is t (where t is sufficiently small).
Alright alright alright alright alright, then how shall we fire our α particles,
we will fire α particles uniformly over an area of A~~~~~~~~
Then the α particles will encounter ntA nuclei,~~~~~~~~~~
and among those, only the α particles that went into π(b.^2) will be deflected by angles above theta, hehehehehehe
Does everyone agree?!!?!!?!???!?!
Alright then, we will find the ratio of α particles scattered above theta to α particles scattered below that angle.
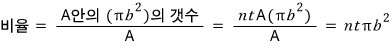
Okay, using the equation from before, let us express the ratio as an equation in theta.
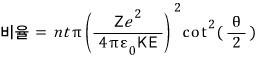
For reference, I Rutherford have drawn the cotangent graph.
Other than theta, what’s in front is just constants so you don’t need to worry about them~~~
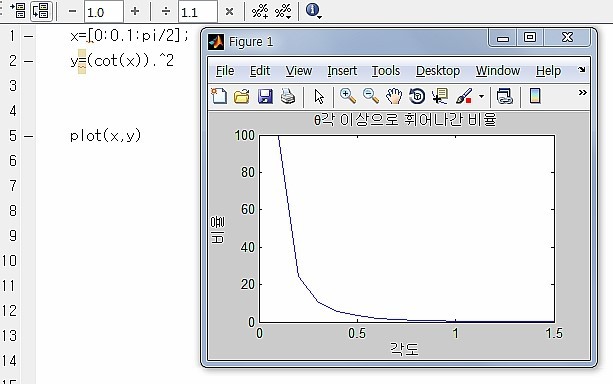
I Rutherford shall think like this
If I think about “when θ changes by thiiiiiiiiis~~~~~~ much, how does that ratio change????”
then shouldn’t I just differentiate that equation with respect to theta??!?!”
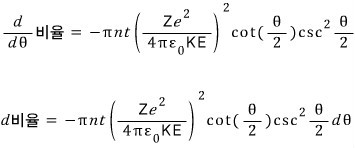
Let us think about the α particles scattered at angle dθ.

That is, if we call that region dS, this humble Rutherford can predict the number of α particles reaching dS, can he not~?
(for dS do a spherical integral, it’s not a difficult integral.)

Done…
I will now go change majors,
may you all live well and eat well….keh keh keh


Sorry for making it feel like I posted this while high on drugs…hehe
Originally written in Korean on my Naver blog (2015-05). Translated to English for gdpark.blog.