The Photoelectric Effect
Ever wonder why Einstein won the Nobel Prize for the photoelectric effect and not relativity?! Let's dig into the experiment that changed everything!
Thanks to the movie Interstellar, now everyone knows him (not necessarily because of the movie though)
physicist Einstein!!!!!!!!!!!!!!
In 1905 he published the ‘Special Theory of Relativity,’ something that would flip the world upside down,
which at the time was treated as nothing more than absurd nonsense,
and then 11 years later, in 1916, he published General Relativity,
but this too was taken as just more nonsense,
until a few years later, in 1919, Eddington’s solar eclipse observation proved it
and only then did Einstein become a superstar, so the story goes…
With a theory that flipped the world on its head, he must’ve gotten the Nobel Prize for it, right!??!?!?!?!?
That’s no no
I myself don’t really know why he didn’t get the Nobel Prize in Physics for this theory,
but I do know that Einstein is a Nobel laureate!!!!!!!
And I know what he got it for!!! It was precisely this photoelectric effect experiment!!!!!!!!!
Just what on earth did this guy figure out from this experiment, and for what contribution,
did he end up getting the prize not for that amazing relativity theory but for the photoelectric effect experiment?
Okay, first, the historical background goes like this.
During some experiment, Hertz found out that if you shine ultraviolet light on a certain metal ball,
discharge happens waaaaay more easily,
but since that wasn’t the purpose of the experiment, he just moved on, apparently.
But then other scientists went
“Ohhh?!?! Wait a sec?!?!?? Why?!?!?! What’s this?!?!?!?!”
and that’s how research in this area got started~~
sweeeet then, start — go go
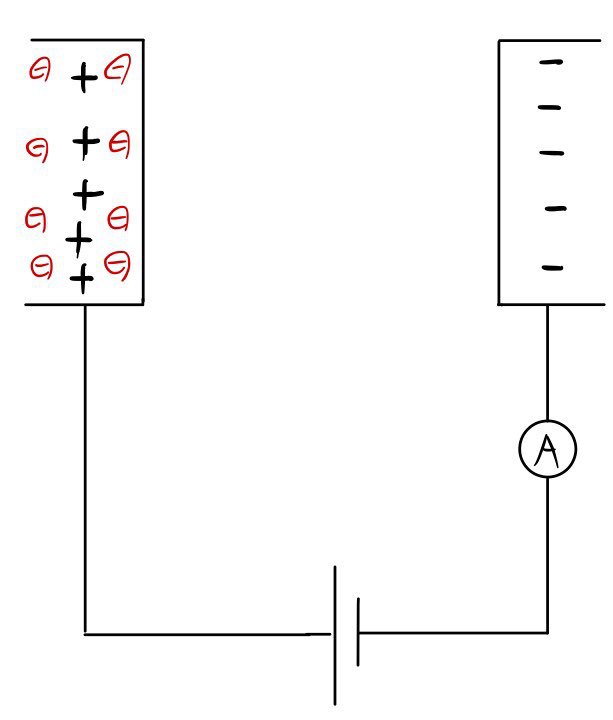
If you hook up a battery between two conductors like thaaaat~~~~, the charges will pile up like this,
and the electrons will end up in a bound state!!!!!
But if you shine some light on it!!~~
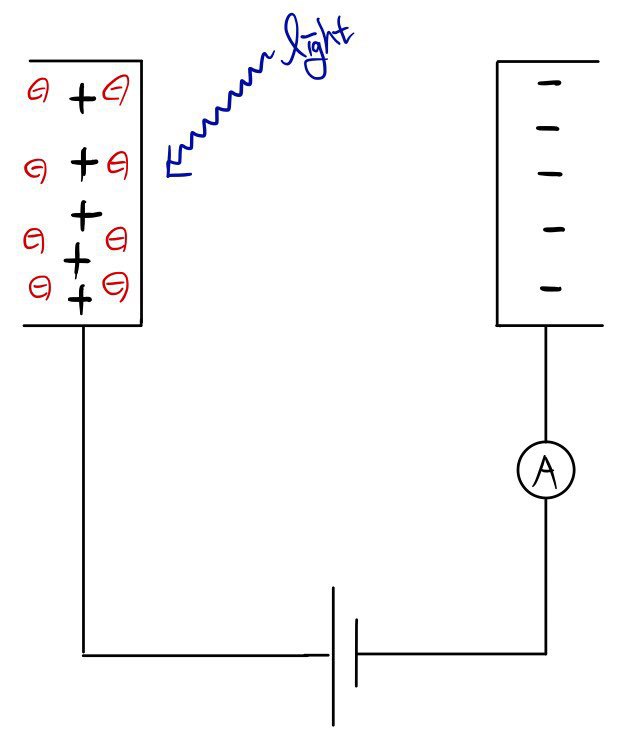
then the electrons go ping~~@@ and fly out~~~~~~, that’s the thing
(whether they ping’d out or not, you can find out by measuring the current)
Okay, so from here, we gave it a few changes.
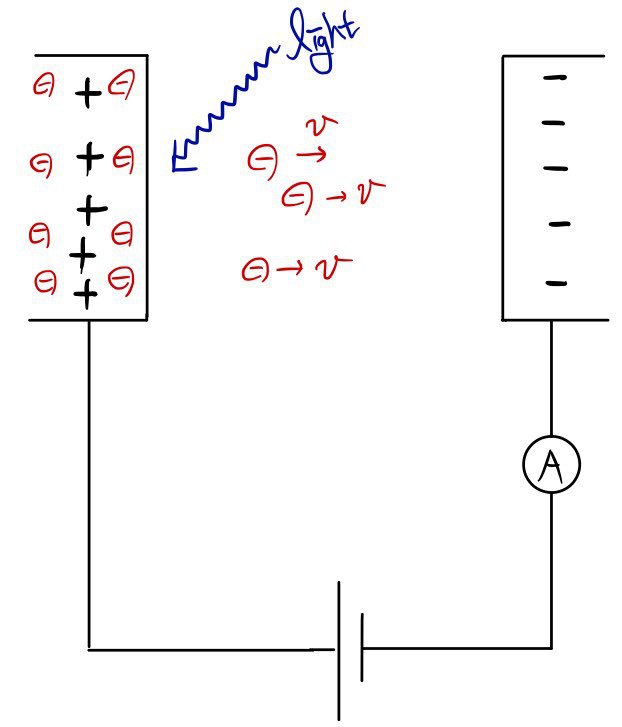
First, let’s slide the Voltage up and down
What happens if you just crank V all the way up??!?????
The circuit will break…. can’t do that…
let’s just raise it a decent amount…hehehehe (just kidding lol)
The flying electrons whose energy isn’t big enough won’t be able to overcome the attraction from the left plate
and the repulsion from the right plate, and they’ll head Back,
and only the strong electrons, strong enough to ignore that stuff, will zoom~~~ into the opposite plate
But the more you raise the potential,
the fewer electrons will zoom~ into the opposite plate~!
In other words, “the higher the potential, the fewer electrons reach the other side”
Therefore, “the higher the potential, the smaller the current that flows”
and if you creep the potential up little by little, then bam!!!!~~
there will be some voltage where the ammeter reads 0, right?! Let’s call that potential value
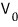
!!!!!!
Among the electrons ping’d~~!!!!~!!! out like that,
there are ones with small kinetic energy and ones with large kinetic energy,
and the higher you push the potential, the more the electrons with small kinetic energy fail to reach the opposite plate,
and as the number of such electrons grows, the flowing current drops.
The more you raise the potential, the fewer electrons reach the opposite plate,…
and at some point the number reaching will become just 1!
That one will be the electron with the number-one thumbs-up kinetic energy among all the flying ones!!!!!
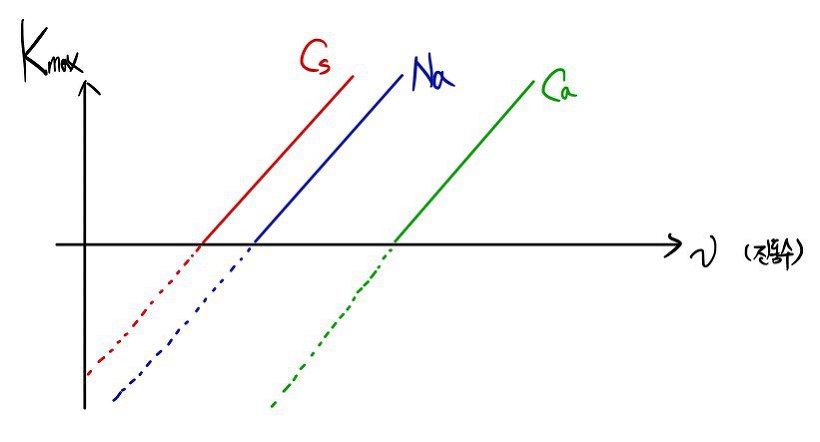
If we call the kinetic energy of that number-one thumbs-up electron
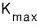
then,
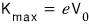
(charge times potential is ’energy,’ after all)
And
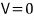
from this point
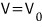
up to here, creeping it up little by little,
watching the

registered on the ammeter, let me plot that data on a graph
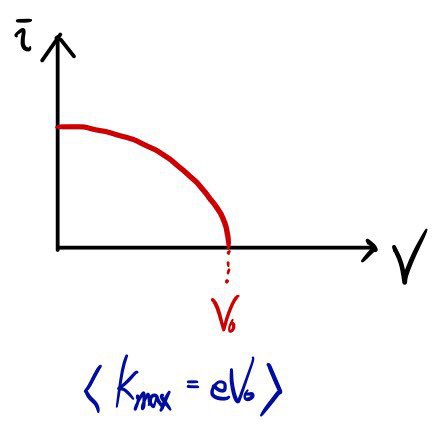
Gaaaaaasp!!! It turned out like this!!!!!!!!!!!!!!
Alright, now, second, let’s do the work of varying another parameter
I checked how the graph behaves for ’light of different colors (different frequencies)~~’
red → green → violet
Raising the frequency like this, I repeated the same experiment, drew the same kind of graph 3 times,
and when you lay all 3 graphs right on top of each other,
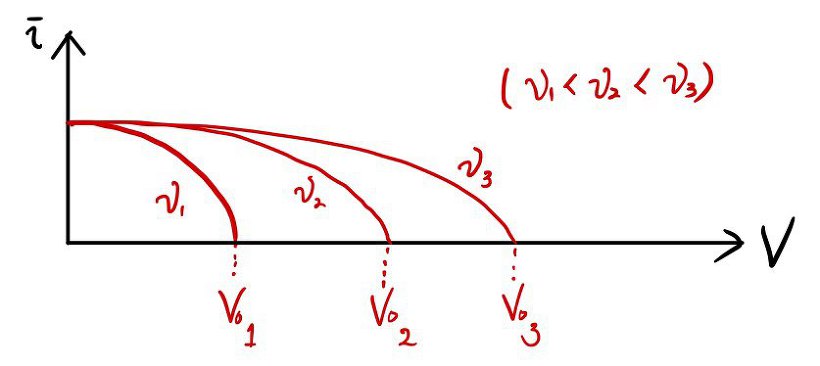
it turns out like this~~~~~~~~~
Third experiment, variable change gogogogogogo
So then, what’s the relationship with ‘brightness of light’~~~~~~????
With that curiosity, using light of the same color and cranking up its ‘brightness,’ if you watch the changes on the graph
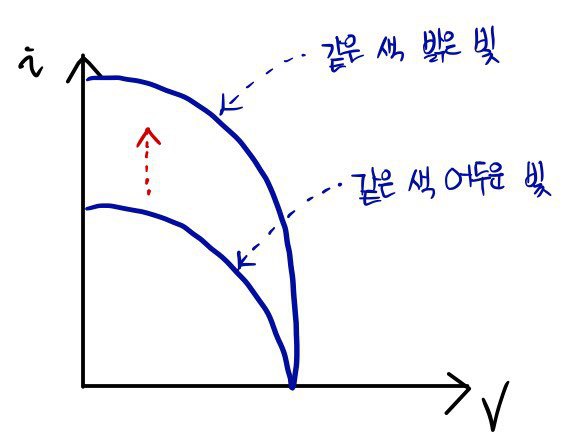
it turns out like this~~~
Now, how we interpret this experimental result data is the key.
Rather than explaining it by bringing in Planck’s energy quantum theory, to put it simply,
it can be explained with this one line
“Hey!!!! Light is a chunk” — we can say that data came out that says exactly this~~
- When we raise the frequency, why does
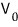
(the stopping voltage) get bigger~?
:
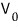
being big means the electron’s speed is big,
so that means raising the frequency made the electron’s speed bigger?
Light energy is proportional to frequency!?>?!?!!!! Yes~
- When V=0, if you make the light brighter, why does i get bigger~~~?
: i being big means many electrons arrived!!!
Even with light of a different frequency (different E),
when V=0 and you shine light of the same brightness, the i value is the same,
and that means the number of somethings hitting the electrons is the same.
If we call the brightness of light the ’number of somethings,’ it fits. In other words, we can say the brightness of light is the number of light chunks called ‘photons.’
To drive the stake in, finally, they tried switching up the conductor material.
With one material, for red light i doesn’t flow (= electrons don’t fly off),
but starting from light with a higher frequency (= higher energy) than red, i gets detected….. whoaaa….
Don’t you get this huge POP!!! feeling now that things, one by one, are lining up with ’light is a chunk’ from above!?!?!?!!
If you plot the relationship between the frequency of the incoming light and the max kinetic energy of the electrons that come flying out,
holy crap, and the slope is exactly~~~ Planck’s constant h~~~~~~~~~~~~~~~~~
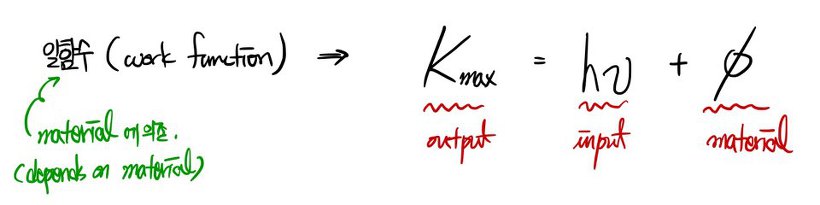
Light => particle(photon) : a particle with zero rest mass (E = hυ = pc)
- A scale that’s nice to have an intuitive feel for:
In vacuum, the energy of a 620nm light chunk is 2eV (calculate it yourself)
=> The energy of red (about 450nm) light is about 3eV
Note) the ’electron that pops out’ is, unlike other electrons, specially called a ‘photoelectron.’
Originally written in Korean on my Naver blog (2015-08). Translated to English for gdpark.blog.