Wave Properties of Particles
If light can act like a particle, why not flip it — turns out matter has wave properties too, and that wild idea leads straight to de Broglie wavelengths and Schrödinger's equation!
Up until just now, we learned that waves have particle-like properties.
We showed that light, known as an electromagnetic wave, has particle-like properties.
(It’s not that light is actually a particle~ it’s literally just the property-property.)
So if we think of an electromagnetic wave as some kind of photon,
the light energy is
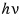
and if we think ’this thing? a particle?',
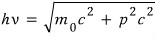
we can write an equation like this,
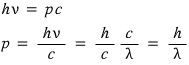
the momentum of light being h over lambda, so we can think of light as some equivalent particle(?) with about that much momentum!!!
The important thing is that light hasn’t actually become a particle now — we’re just thinking of it as a particle; its essence is still a wave as a wave.
Now from here on, it’s the opposite idea!!!
The essence is matter (object, particle),
but can we think of these as equivalent waves(?)~~~~
As for the equation first… its momentum is
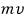
and,
“haaa~~ you are a particle alright, but what if you were a wave..T_T.T_TT_T~~”
if we go crazy and think that way,
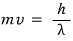
would it be okay to write it like this~~~~????
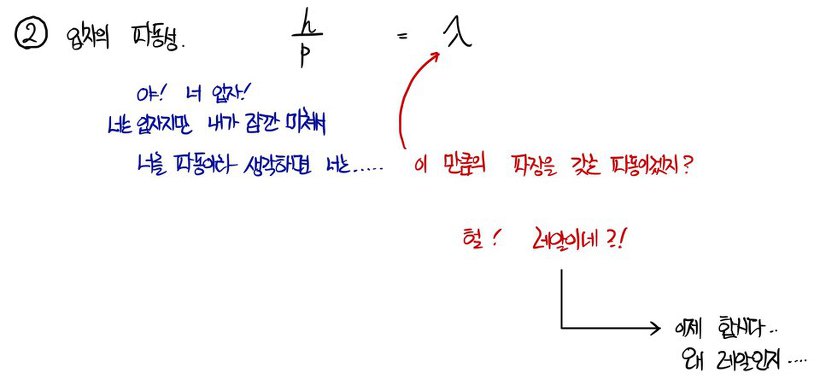
When we think of it as a wave, the wavelength of that equivalent wave
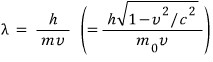
this wave’s wavelength is called the De broglie wavelength, the de Broglie wavelength!!!~!!!!
Now that we’re looking at it as a wave, it definitely has to show diffraction and interference, but basically for waves with too short a wavelength,
you can’t really see whether diffraction or interference is happening.
So
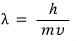
for this to be meaningful, since h is a constant let’s set it aside, mv has to be ridiculously small,
but Planck’s constant h is about 10 to the minus 34th power, right???????
Ah then!!!??!?! for the wavelength to be visible?!?!?! how small does mv have to be~~~~???
If the wavelength is at least 10 to the minus 10th, we can observe X-ray diffraction.
So to see it the way we saw X-ray diffraction, mv needs to be on the order of about 10 to the minus 24th power for observation to be possible………..
whoa… does such a thing even exist???????????????????????
(cf. regarding duality: as was the case with electromagnetic waves, the particle-ness and wave-ness of an object are never observed simultaneously.
Therefore, the question of which is the ‘correct’ description is meaningless, and all we can say is that a moving object looks like a wave in some cases and like a particle in other cases.)
Scientists thought about such waves of matter, and they wrote this ‘matter wave’ of matter as ψ(r,t),
and Schrödinger also found an equation that this matter wave psi(r,t) satisfies.
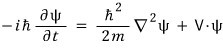
He just found an equation that satisfies this,
but we have no idea what the physical meaning of ψ(r,t) is!!!!
At that moment, appearing like a hero, the genius Max Born said this
“ψ(r,t), as you can see in the equation above, is just a complex number. But the important thing is that when you square that ψ(r,t), it does have a physical meaning!!
That is, it’s the probability of finding that particle at position r at time t~~~~”
Now the revolution has begun.
So can we think that we already know the wavelength of the wave ψ(r,t)?
As the de Broglie wavelength????
Then, forget the wavelength, about the wave~~~~
How fast is the wave ψ(r,t), which we can neither actually measure nor see with our eyes, moving??
Is it the same as the speed of the actual object?????
Let’s first set off to try to answer this question.
First, using de Broglie’s matter wave relation, let’s find the speed of that wave.
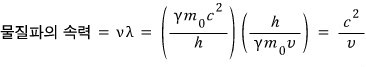
Now now now now, but whatever we don’t know, the object’s speed v should be less than c, right.
That means, we end up with the conclusion that the speed of the matter wave is greater than the speed of light………..whoa………
Then do we have to break Einstein’s postulate and say that the speed of an object can be faster than the speed of light~~?
No No No., that’s not it; there are a few cases in nature where something can be faster than the speed of light.
So people thought this.
“The matter wave must be a wave group~~~~~~~”
So from now on what I’ll talk about
is the story of phase velocity & Group velocity.
If we study this, we can find out the cases where it can be faster.
In general, the classical wave equation is
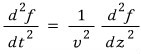
(For the derivation of this equation, please refer to the electromagnetic wave post in electromagnetism. http://gdpresent.blog.me/220435098229)
Electromagnetic Wave - first, ‘Wave’ [ Electromagnetism I studied #28 ]
Before thinking about electromagnetic waves…. which you’ve probably heard a lot about, let’s first get to know what ‘waves’ are…
gdpresent.blog.me
A function that satisfies the differential equation like above is what represents a wave,
and if that function is a sin function,
that is, if we make the assumption that the wave is a ‘sine wave’~~,
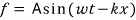
it’s derived like this.
Let me write k like this.
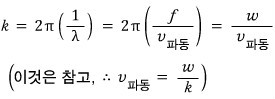
Now then, the matter wave must be a Wave Group~~!!!!~~~!~~!!!!!!
The idea will look something like this~~~~
that’s what we think.
This kind of wave group has what’s called beats in our reality, and I’ll explain the wave group(?) by looking at that.
First let’s make beats into an equation.
Ah ah ah, first of all what beats are, is ‘a phenomenon that appears when two waves with the same amplitude and sliiiiightly different frequencies combine’~!
To give an example with a sound wave, there’s the ‘Divine Bell of King Seongdeok’.

<Actually this is the Emille Bell. It’s called the Emille Bell because the bell sound you hear sounds like emil~~~le, but this nickname was apparently made up by Japanese people during the Japanese colonial period who were secretly super amazed by it and wanted to belittle this bell. Let’s all call it the Divine Bell of King Seongdeok!!!!
(If you compare this bell’s sound with bells from around the world of that era, it’s definitely almost revolutionary-level bell sound!!!! But what’s even more amazing is that it’s not just bell sounds from the same era — even compared to aaaalllll bell sounds in the whole world, this is truly a revolution. How were our ancestors able to think of such a thing? lollollollollolcheck it out yourself — compared to bells from all over the world, the Divine Bell of King Seongdeok is a revolution. Revolution!!!!)>
( The scientists of the Silla era who made that bell had already understood beats through experience, and it’s true that they cleverly dug out the ground underneath and designed it so that the sound made by ringing the bell and the combined two sound waves would produce the beat phenomenon. Daebak!!!!
Another thing! During the Park Chung-hee regime, they tried to move the location of the Divine Bell of King Seongdeok, but the bell hanger that was holding up that several-ton bell —
(what do you call a bell hanger in pure Korean… I forgooot..T_TT_TT_T) they tried to make a new one with high-tech modern science and technology and swap it out,
but apparently they couldn’t make that black-colored thick rod with the latest technology of the 70s at that time……. the science and technology of that time couldn’t make a bell-hanger rod thin enough to fit into the red hole while still being able to bear that bell’s weight…..so…… only that bell hanger…. they couldn’t swap out with the original one… apparently.lol.lol.lollollollolfascinating)
The reason the Divine Bell of King Seongdeok is mystical is
when you strike the bell ding~~~~
woooongwoooooooongwooooooo~
the sound rings out like this, and that’s the mystical reason!!!!
Well, why does it sound like that~~~
Apparently it’s because of beats.
Now let’s simply make beats.
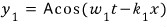
To make beats on this kind of wave,
let’s make a wave with the same amplitude and a sliiightly different frequency and combine the two.
(Frequency being sliightly different means w is sliiightly different, and that means k is sliiiightly different.)
So the wave to combine with is
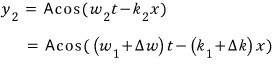
combine, let’s go go go
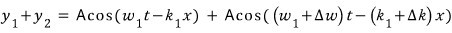
Did you learn “cos-plus-cos two-cos-cos” in advanced high school calculus? lollollollollol I don’t know it hehehe
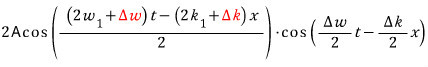
Now that red part is too small a value compared to what’s being added in front, so let’s ignore it!
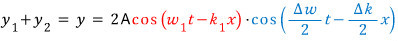
Let’s look at one point (fixing x) at the position of the medium (y) over time t, with the red and blue separately.
Compared to the red wave, the blue wave’s medium position will move soooo soooo slowly.
Since Δw is soooo small compared to w, even if t passes a fair amount, the blue wave barely…..
Now, as we saw earlier,
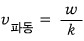
so~
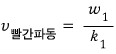
But, hey…. w and k are related to the frequency and wavelength lambda,
and that frequency and wavelength would have come from the equivalent wave matter wave of the moving object, wouldn’t they~~~~?

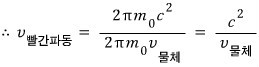
oho~~~ something very familiar came out first~
Riding the momentum, let’s also do the blue wave’s speed, yeah???!?!~?!!

yahoo~~ now I know!!!!!
The conclusion that the wave’s speed was greater than the speed of light c wasn’t strange~~~
Conclusion: for a moving object, we can define a matter wave, and that matter wave must be a wave group.
But the speed of that wave group is the same as the speed of the moving object!!!!
The speed of the red wave is called the phase velocity of the wave,
and the speed of the blue wave is called the group velocity of the wave.
So we thought that this coming out faster than the speed of light was a contradiction,
but we’ve confirmed that it’s not a contradiction!!!
Now let’s take a look back.
(time for self-reflection, let’s go)
De Broglie’s matter wave
is nothing but the momentum of an electromagnetic wave (photon)
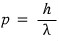
shoved into it.
So how, the wave-ness of a particle… if it’s a wave, it has to show the characteristic of refraction or diffraction,
have we confirmed that~~?
Did (particle) electrons’ motion direction actually change when we shot them at a tiny slit????~?!?
That’s not the case T_TT_TT_TT_TT_T
Here too, we can’t drill a hole the size of an electron
we can’t drill a hole this tiny,
and even if we could drill one, we wouldn’t be able to make the electron score a goal in through that hole
But it’s not that we haven’t confirmed it. So what I’m about to describe is the content of Davisson & Germer’s experiment.
<England’s J.J Thomson apparently also did a different experiment on the same topic in the same era, but this one is said to be easier than that.>
What’s the content of that experiment~?~?!?!?!?
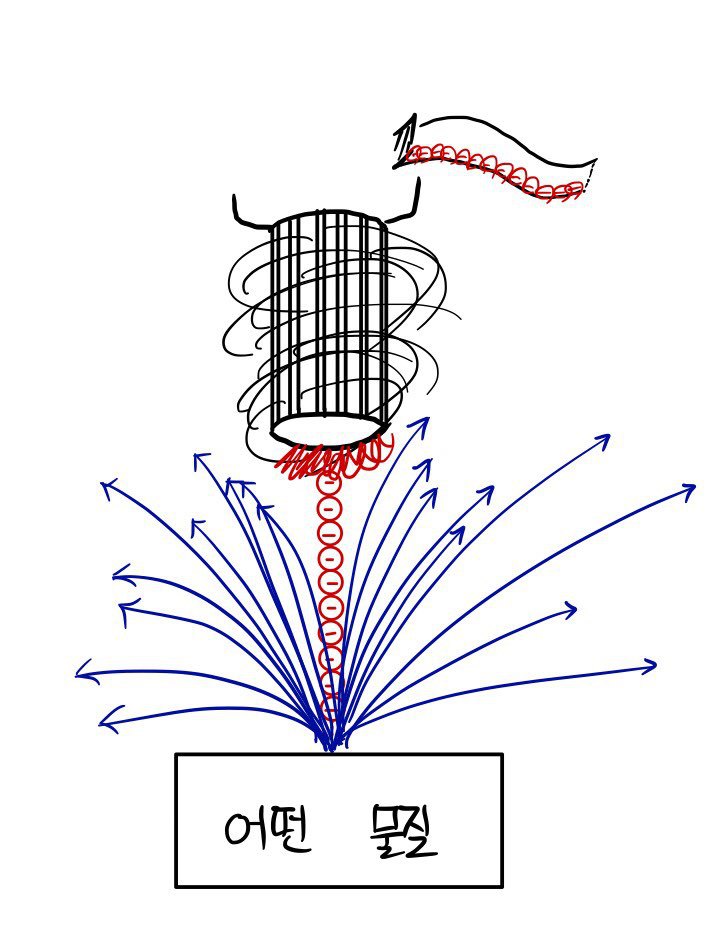
Take some material, and shot the hell out of them~~!~!!!! with an electron gun (electron Gatling gun) firing bullets (electrons) lol
“Clearly the electrons must scatter in all directions like crazy.”
it should be, but the fascinating thing about the experimental result is that “as we varied the energy of the electrons, at some particular energy, there was an angle at which particularly many electrons bounced off.”
Precisely when the electron’s energy was 54eV, the number of electrons bouncing out at 50 degrees was noticeably larger!!!!
Then “so what?” one could reasonably ask, but now I’ll explain that this is related to the de Broglie matter wave!!!!
The incident angle and reflection angle with respect to the Bragg plane group in this experiment were each 65 degrees, apparently — let’s see.
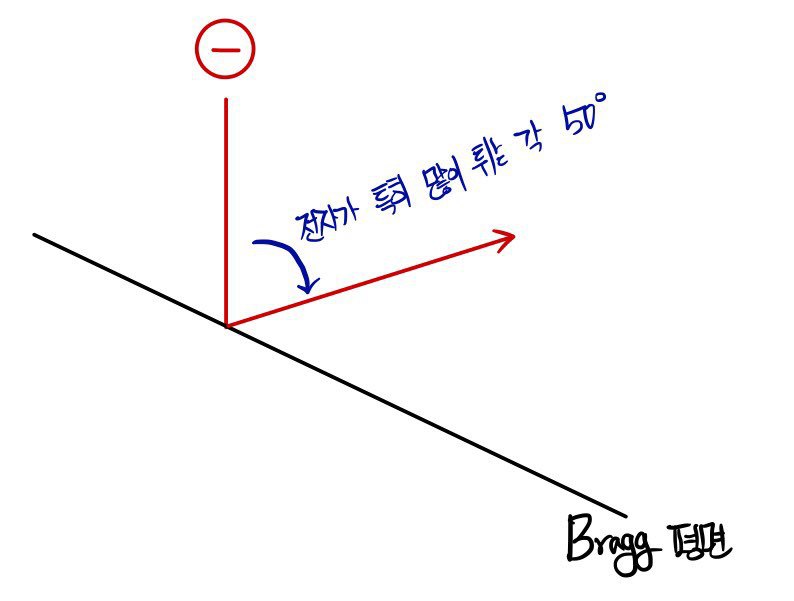
What? Isn’t it obvious that the angle where electrons particularly bounce a lot is 50 degrees~~?!?!?!?!?
you might think, but don’t — in a game of marbles, where is there such a thing as an angle where marbles particularly bounce a lot…
That is, particles don’t have such a property!! But!!!! the experimental result is like that!!!!!! whoa!!
what the heck?!?!?!?!?!?
ha….. “Now I’m going to go crazy and look at you not as a particle but as a wave!!!” if we have a slightly crazy mind like that,
now at a 50-degree angle, it’s not that you bounced a lot,
but I’ll think that what I saw was one fringe of constructive interference….
Calculating that way and backtracking to reveal — what is your wavelength~?!!?~~!?!?~~~~~~
if we ask that,
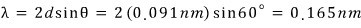
(everyone get ready for chills)
Could you….possibly…..???? …… let me calculate your wavelength using the de Broglie wavelength…..
Since the electron’s 54eV energy is much smaller than its rest mass energy of 511keV,
so there’s no need to calculate with relativistic momentum, let’s do a classical calculation.
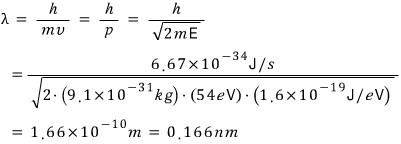
chiiiiiiiiiiiiiiiiiiilllllllllllllllllllllllllllllllsssssssssssssssssssss
Let me summarize one more time!
Clearly electrons are particles so it’s obvious they bounce off in every direction when they hit something!
But, when we shoot electrons of a particular energy, there appears to be an angle at which they bounce in one direction.
I’m going to lose my mind…. with the feeling of just going crazy together,
when we thought of the particle as a wave and shoved it into the equation,,,, the explanation became possible….. through ‘constructive interference’
eek
And just to be extra careful, let me emphasize one more time,
matter having wave-ness
does NOT mean that tiny particles like atoms actually travel wobbly like waves!!!!!!!
“If anyone here has seen an atom directly with their own eyes, come out!!!”
Nobody anywhere has ever seen that an atom is round-shaped…
There are cases where we need to think of the atom as a round particle to get an answer,
and there are just cases where we need to think of the atom as a wave to get an answer…..
Originally written in Korean on my Naver blog (2015-08). Translated to English for gdpark.blog.