Atomic Models and the Bohr Model
We dig into why the old orbital atomic model is totally wrong — spinning electrons radiate energy and collapse — and set the stage for Bohr's fix.
Now we’re on Chapter 4. Atomic Structure,
if you read the Rutherford scattering experiment post ( http://gdpresent.blog.me/220347903153 ) first,
please think of this post as the content that unfolds next.
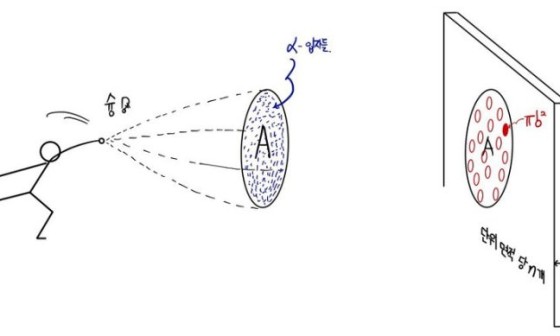
Rutherford scattering, Rutherford scattering formula [ The modern physics I studied. #0 ]
Ah, today while studying I was so sleepy that I thought I’d post about the Rutherford scattering formula I just studied, piping hot. …
gdpresent.blog.me
Start
Typically, up through middle school science and high school Chemistry 1, they teach the atomic model like this.
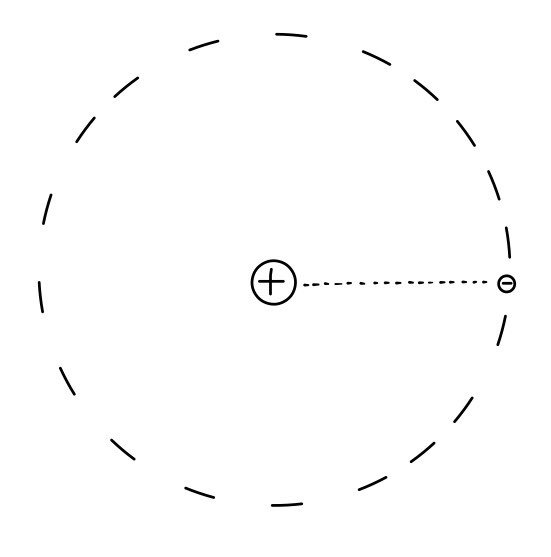
Wouldn’t it be strange to teach what an atom is from the very start using orbitals?????
Let’s go over once again the orbital model of the atom that we learned when we were young.
Through Rutherford, the existence of a nucleus with positive charge was firmly established.
So I think that at that time, there probably wasn’t any other way besides this orbital model.
(Even though Rutherford himself knew the orbital model was a wrong model)
Anyway, let’s look at it with the wrong model, the orbital model, for now.
Hmm, first of all, in that model, the electron can’t just sit still. Because the nucleus inside pulls it in!!!!
Then the electron??? “It has to orbit.” Only then is it balanced.
The centripetal force is
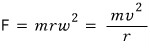
And that force will be the electric force between the positive charge and the negative charge separated by distance r acting as the centripetal force, so,
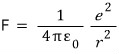
Therefore,
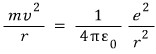
That is, the speed of the electron when it rotates is
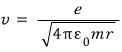
Since it has speed, it’s obvious it has Kinetic Energy,
and it also has electric potential energy~!~!!!!
The total energy is
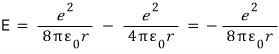
But~~~~(ah, finally saying it) the orbital model of the atom is wrong.!!!
The reason is simple. Because according to the electromagnetic theory fully established by Maxwell, the fact that an electron is moving means the existence of a current,
and if a current is produced, then by the current a magnetic field, and again by the magnetic field an electric field, and again by the electric field a magnetic field…. in this way an electromagnetic wave will be radiated
and an electromagnetic wave being radiated means the existence of a Poynting vector,
that is, energy is being carried away.(If the electron revolves around the atom, energy gets carried away.)
The atom is already tiny, so how much energy does it even have,
but once it starts spinning, electromagnetic waves are radiated, and so the electron, while rotating, drops energy bit by bit by bit
and this means the rotation radius gradually gradually gradually shrinks.
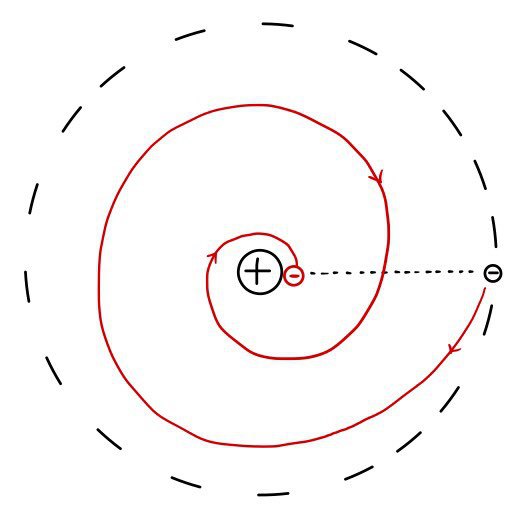
This means the atom collapses, meaning the atom didn’t exist,
and it becomes a statement that all things must disappear as soon as they are born T_T T_T T_T
Now then, let me talk about something else. Namely ‘Niels Bohr’
This man is a Danish physicist, and he’s said to have played as an amateur soccer player, position goalkeeper.
But at age 26 he was suddenly captivated by electron theory, and is said to have left his fiancée briefly in his hometown and boarded a ship bound for England…..
In order to go to ‘Thomson’ of Cambridge, the top figure in this field at the time,
but Bohr was supposedly very brash. To Thomson, the top authority,
he’d say things like “This part here is wrong.” ~
Thomson, the top authority, was reportedly displeased.
Bohr, who had this bad relationship with Thomson, one day met Rutherford.
After meeting and having a conversation, he supposedly decided to spend the remaining few months of his doctorate under Rutherford.
Bohr supposedly had a thought around that time.
He
thought, “The fact that the atom maintains a stable state could mean that there exists a stable arrangement in which the electron can revolve around the nucleus?”
But at this point he hadn’t yet made it to completion, and while spending about a year absorbed only in research on that,
one day (February 1913), while talking with ‘Hans Hansen,’ a young physicist from the German University of Göttingen who had been studying atomic spectra,
he is said to have encountered an odd formula called the ‘Balmer series’,……..
The fuse was lit,,,,,,,,,,,,,
So, to talk a bit about the Balmer series,
this is a research result from 1885, about 30 years before that time,
and it’s a study in which the Swiss mathematician ‘Johann Jakob Balmer’ discovered a certain regularity in the positions (frequency or wavelength) of the hydrogen atom spectrum.
Balmer’s formula is said to have been generalized three years later by the Swedish physicist Johannes Rydberg,
but until Bohr heard this news, this was just a matter of fitting numbers,
and nobody knew the physical meaning……
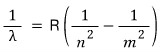
This here, this one. This is the research result carried out by Rydberg up to this point.
But our genius Mr. Bohr, after that,
is said to have accurately grasped the physical source of m and n.
“When an electron orbiting in a high-energy orbit jumps to a lower-energy orbit, E is emitted in the form of radiation” — like this.
Hmm, the Balmer series is a series of spectra of electrons that fell to the n=2 energy orbit, and the Paschen series, discovered in 1908, is the case of falling to n=3,
and Bohr found them all — n=1, n=4, 5, 6,,,
and Bohr also succeeded in showing that the Rydberg constant R can be expressed in terms of already-known physical quantities — Planck’s constant, the mass and charge of the electron, etc.
Also, there was a person called Edward Charles Pickering, an American physicist, who was studying a different kind of emission spectrum,
and that series uniquely did not work with integers but with half-integers.
When Bohr’s idea was applied to that series for He, which has two protons, it was reportedly splen~~~didly reduced to integers.
And so he named the integer that characterizes orbital electrons
the quantum number, and the jump between two orbits the quantum jump,
this is the person who gave them those names lol lol lol
Now I have to explain this, and I’m going to do it using ‘matter waves’????~~~
Actually, de Broglie explained the wave nature of particles about 10 years after this achievement of Bohr’s,
but since this idea of Bohr’s is easily derived from the de Broglie matter wave, let’s just explain it with the de Broglie matter wave concept.
First, de Broglie threw away the idea that the electron is a ‘particle (lump of energy)’.
And then he said this.
“We firmly believe that the electron is the archetype of an energy lump (particle), but there is no guarantee that this is the truth.
If the electron’s energy is spread over a wide region and most of its value is concentrated in a narrow region, it could look like a particle.
Since we know very little about the electron, we cannot guarantee that either side is correct.
However, if we allow this possibility, then an electron bound in an atom should be thought of not as confined within the limited region of the atom but as spread out over all of space.
Also, since the electron cannot be split into smaller pieces any further, it must be regarded as a single unit!”
Well, this seems to be the introduction — I don’t know if it’s because de Broglie was actually of noble descent, but he’s said to have had quite an ear for music.
To talk about instruments, all string instruments and wind instruments make their characteristic pitch by vibrating a string or air, and the wave generated at that time has the common feature of being a standing wave (stationarty wave) fixed at both ends.
De Broglie tied the physics principle hidden in music to the ‘quantum number’.
“For an electron to maintain a stable state inside an atom, the electron’s wave must take the form of a standing wave along the orbit, like a string of a string instrument.”
Now what differs from an instrument is that here the orbit is circular,
and to produce a standing wave on a circular orbit, the length of the orbit must be an integer of the electron’s wavelength!
<If it isn’t in the standing wave state, it won’t be sustained due to destructive interference, so the condition ‘it is a standing wave’ is attached>
Electron’s wavelength???? OK OK
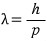
The electron’s speed is
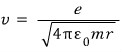
was what we had, so
if we compute the de Broglie wavelength,
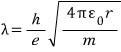
An integer multiple of this wavelength must be the length of the orbit, so,
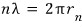
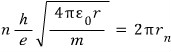
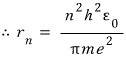
Everything except n is a constant, so, just
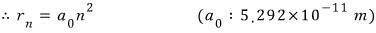
Here, a-zero can be thought of as the number that comes out when you put 1 into n,
and it’s the same as having computed the radius of the atom when n=1,
and this is called the ‘Bohr radius’.
Now the revolution has begun.
“The electron cannot orbit in ju~st any orbit.”
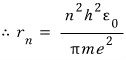
It can only orbit in orbits like this….
What this means now is,
the electron cannot orbit in any orbit → “the electron cannot have just any (arbitrary) energy.”
Earlier when we computed the electron’s total energy,
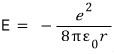
we had this, and here if we replace r also with r sub n,
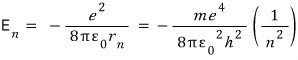
Then if we compute E sub 1………..we get -13.6 eV.
This was also something treated importantly in quantum mechanics~~~?!?!?
< Wait!!!! I keep saying orbiting,
but here nobody has ever seen the electron orbiting!!!!!
It’s just an expression; whether it’s orbiting or not, nobody knows! >
cf.) The state n=1 is called the ground state,
and all states with n≠1 collectively are called excited states….lol
As everyone knows, by this principle the secret of the line spectrum was unraveled.
(For atoms with small mass…. but once you get to 2 protons, there’s the downside that it now becomes hard to compute by hand lol)
Anyway, the energy levels of the atom are formed as you see above, n=1, 2, 3, 4 ~ like this,
and when energy is received the electron’s energy level goes up, and when by some event the electron’s energy level falls, energy is emitted by that much~
we could come to interpret it like this.
Starting with the Balmer series in this way, the secrets of the Lyman, Paschen, Brackett series, etc. were revealed,
and the reason the Balmer series was discovered first is probably…. because it’s the visible-light range, visible to the eye, right?!?!??!?!~~!!?!?!?
Aaah and I said Bohr described the Rydberg constant in terms of physical quantities,
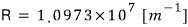
let’s check this.
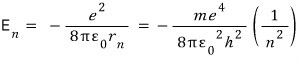
from
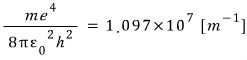
Amazing…. try punching it into a calculator yourself lol
Aaah aaah aaah anyway Bohr,
the fact that he did all of this without even introducing de Broglie’s matter-wave wavelength concept,
means he’s definitely a real genius…lol lol lol heh heh heh heh heh heh
Originally written in Korean on my Naver blog (2015-08). Translated to English for gdpark.blog.