The Delta-Function Potential
Time for the delta-function potential — we sort out bound vs. scattering states, meet the Dirac delta, and motivate why V(x) = −αδ(x) is a solid model for electrons in a material.
We’ve been grinding through the time-independent Schrödinger equation case by case, depending on V.
And now — the long-awaited 4th potential: the delta-function potential.
(The finite square well is exaaactly the same discussion as the delta-function well later on!!!!)
Before we actually get started, a teeeeny bit of terminology: bound state vs. scattering state.
1. Bound state
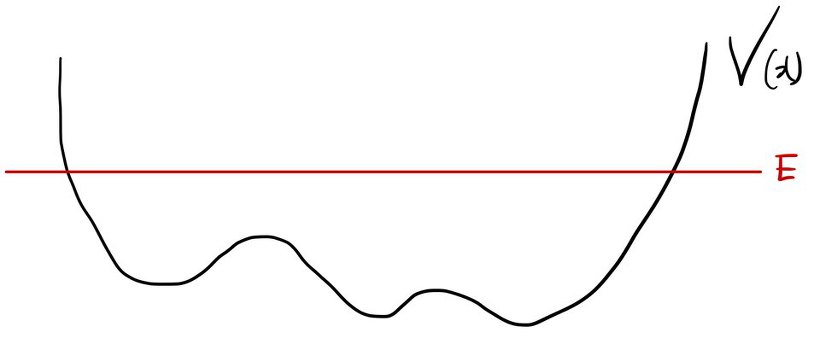
Bound state. As the name hints:
$$E < |V(\pm\infty)|$$…or roughly, just $E < 0$.
Most(?) of what we’ve done so far has been bound states. The infinite square well, the harmonic oscillator — all bound.
Why “bound”? Because the particle’s E is smaller than V(x) out at the edges, so it literally cannot escape past the wall. It’s stuck. Hence — bound. heh.
That’s the picture, right?! (Is it really stuck though? hehe~)
2. Scattering state
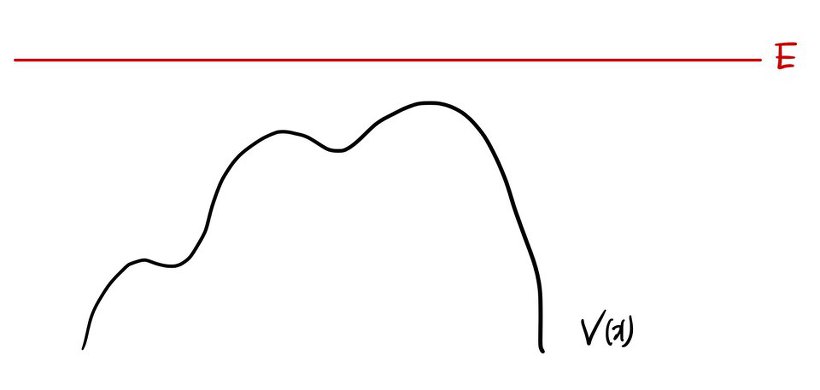
Scattering state is the exact opposite.
$$E > |V(\pm\infty)|$$And of course, if $V=0$, then $E>0$ says the same thing.
From what we’ve already done, the free particle was a scattering state. heh
Now — does E have to be bigger than V everywhere to count as scattering?

What about something like this?
Yep, this counts as a scattering state too!
OK, so. Delta function.
Definition:
$$\delta(x) = \begin{cases} \infty \quad (x=0) \\ 0 \quad (x \neq 0) \end{cases}$$Pointy! Only at one specific spot, and flat-out zero everywhere else!
(Like — imagine a person who doesn’t react to anyyything. Except money. Money sends them into orbit. If you wrote that person as a delta function:)
$$\delta(x - \text{money}) = \begin{cases} \infty \quad (x = \text{money}) \\ 0 \quad (x \neq \text{money}) \end{cases}$$So, it’s this function people cook up whenever they need that kind of “spike at one point, nothing else” behavior.
But then Dirac came along and made it meaningful:
“Hey — come on. Where in nature is there actually something that reacts infinitely to one specific thing? It’d just be a really high value, not infinity~~~”
$$\int_{-\infty}^{\infty} \delta(x)\,dx = 1$$The $\delta(x)$ that satisfies that — that’s the Dirac delta function.
And it means:
$$\int_{-\infty}^{\infty} f(x)\,\delta(x-a)\,dx = f(a)$$OK. Before we dive in for real, let me quickly motivate why we care about a potential shaped like $\delta(x)$.
Two pictures, keep it simple:

Imagine an electron moving around inside a material.
If we sketch that as an energy graph —
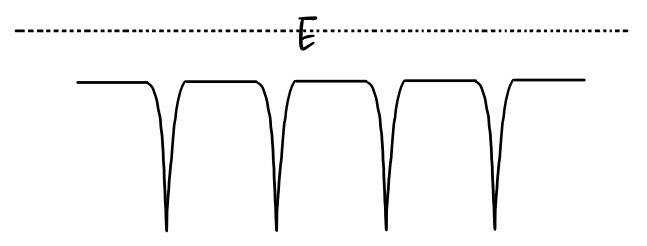
Ta-daaa~~~
Now we really start.
In the Schrödinger equation we need to plug something in for V(x). But instead of just $\delta(x)$, let’s go a little more general:
$$V(x) = -\alpha\,\delta(x)$$You can think of $\alpha$ as a kind of “property of the material.” It captures the fact that the potential depth in, say, uranium isn’t the same as in hydrogen.
The minus sign just means the delta spike points down — a downward-buuulging delta.
So the time-independent Schrödinger equation is
$$-\frac{\hbar^2}{2m}\frac{d^2\psi}{dx^2} - \alpha\,\delta(x)\psi = E\psi$$We’ll look at $E>0$ and $E<0$. Starting with $E<0$.
What does the bound electron look like?
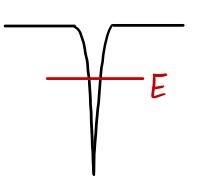
Something like this, right?!
OK so just like with the infinite square well, split by intervals and gogo.
$$x<0,\quad V=0 \\ -\frac{\hbar^2}{2m}\frac{d^2\psi}{dx^2} = E\psi \\ \frac{d^2\psi}{dx^2} = -\frac{2mE}{\hbar^2}\psi = K^2\psi \quad \text{(E is negative, so factor out the minus and call it } K\text{.)} \\ (D-K)(D+K)\psi = 0 \\ \therefore\quad \psi(x) = Ae^{Kx} + Be^{-Kx} \\ \text{But } B=0 \text{ — otherwise it blows up at } x=-\infty. \\ \therefore\quad \psi(x) = Ae^{Kx}$$Now $x>0$!!!!
$$\therefore\quad \psi(x) = Fe^{-Kx}$$Same logic — to keep it from diverging at $+\infty$, only that term survives.
And now the boundary! At $x=0$, $\psi(x)$ has to be continuous —
$$\psi(0_-) = \psi(0_+) \\ A = F$$So:
$$\therefore\,\psi(x) = \begin{cases} Ae^{Kx} \quad (x<0) \\ Ae^{-Kx} \quad (x>0) \end{cases}$$Sketch it:
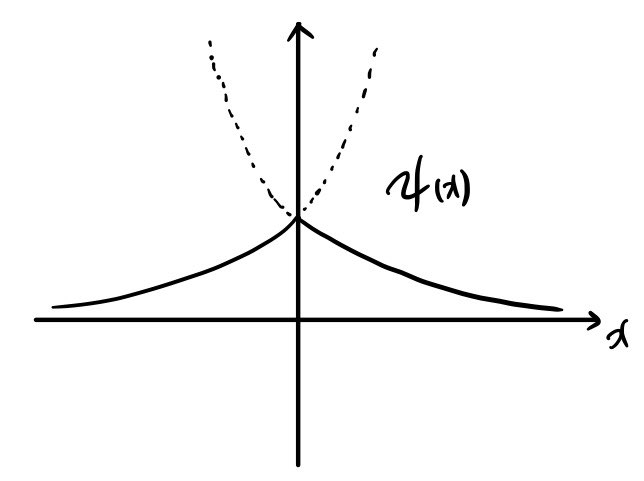
Looks like this. But… the sharpness of the kink in the middle — that should depend on the potential, shouldn’t it?! huh?
I mean, how pointy that cusp is — that’s gotta be tied to $\alpha$…. (cries)
But how do we actually express that?!
OK so —
$$-\frac{\hbar^2}{2m}\frac{d^2\psi}{dx^2} - \alpha\,\delta(x)\psi = E\psi$$We’re going to integrate both sides of this from $-0$ to $+0$…
But writing “$-0$ to $+0$” feels too high-school. Let’s be grown-ups:
$$\int_{-\epsilon}^{\epsilon} \cdots$$No problem, right?! Just think of $\epsilon$ as $0.000000000000000000000001$.
Then gogo, integrate both sides:
$$-\int_{-\epsilon}^{\epsilon}\frac{d^2\psi}{dx^2}dx - \frac{2m\alpha}{\hbar^2}\int_{-\epsilon}^{\epsilon}\delta(x)\psi\,dx = \frac{2mE}{\hbar^2}\!\left(\int_{-\epsilon}^{\epsilon}\psi\,dx\right) \\ \text{The red one is Zero! because } \psi(x) \text{ is continuous}$$$$-\int_{-\epsilon}^{\epsilon}\frac{d^2\psi}{dx^2}dx - \frac{2m\alpha}{\hbar^2}\int_{-\epsilon}^{\epsilon}\delta(x)\psi\,dx = 0 \\ \Delta\!\left(\frac{d\psi}{dx}\right) + \frac{2m\alpha}{\hbar^2}\int_{-\epsilon}^{\epsilon}\delta(x)\psi\,dx = 0 \\ \Delta\!\left(\frac{d\psi}{dx}\right) = -\frac{2m\alpha}{\hbar^2}\psi(0) \\ \left.\frac{d\psi}{dx}\right|_{+\epsilon} - \left.\frac{d\psi}{dx}\right|_{-\epsilon} = -\frac{2m\alpha}{\hbar^2}\psi(0)$$And those two colored guys on the left — we can totally find them!
$$\left.\frac{d\psi}{dx}\right|_{+\epsilon} = -KAe^{-K\cdot 0} = -KA$$$$\left.\frac{d\psi}{dx}\right|_{-\epsilon} = KAe^{K\cdot 0} = KA$$$$-2KA = -\frac{2m\alpha}{\hbar^2}\psi(0) = -\frac{2m\alpha}{\hbar^2}A \\ \therefore\quad K = \frac{m\alpha}{\hbar^2}$$Originally K was defined as $K = \frac{\sqrt{-2mE}}{\hbar}$, and now we’ve also written K in terms of $\alpha$ — which means we can write E in terms of $\alpha$:
$$\frac{\sqrt{-2mE}}{\hbar} = \frac{m\alpha}{\hbar^2} \\ \therefore\,E = -\frac{m\alpha^2}{2\hbar^2}$$Finding K means finding E, which means finding $\psi(x)$. OK, let’s close it out with normalization — so $|\psi|^2$ actually means a probability.
$$\therefore\,\psi(x) = \begin{cases} Ae^{\frac{m\alpha}{\hbar^2}x} \quad (x<0) \\ Ae^{-\frac{m\alpha}{\hbar^2}x} \quad (x>0) \end{cases}$$$$1 = \int_{-\infty}^{\infty}\psi^*(x)\psi(x)\,dx = \int_{-\infty}^{0}A^2 e^{\frac{2m\alpha}{\hbar^2}x}dx + \int_0^{\infty}A^2 e^{-\frac{2m\alpha}{\hbar^2}x}dx \\ = A^2\!\left[\frac{\hbar^2}{2m\alpha}e^{\frac{2m\alpha}{\hbar^2}x}\right]_{-\infty}^{0} - A^2\left[\frac{\hbar^2}{2m\alpha}e^{-\frac{2m\alpha}{\hbar^2}x}\right]_0^{\infty} = 2\frac{A^2\hbar^2}{2m\alpha} = 1 \\ \therefore\quad A^2 = \frac{m\alpha}{\hbar^2},\quad A = \frac{\sqrt{m\alpha}}{\hbar}$$Fully normalized:
$$\psi(x) = \frac{\sqrt{m\alpha}}{\hbar}e^{-\frac{m\alpha}{\hbar^2}|x|} \\ E = -\frac{m\alpha^2}{2\hbar^2}$$Unlike the infinite square well, the answer doesn’t come out as some expression in $n$!?!?!
Which means there’s only one state — one E — that exists.
And the probability of finding the electron is (naturally) highest right next to the atom, right?!
(Why? Lots of stuff stuck to the atom, I guess?) hahahaha idk
Originally written in Korean on my Naver blog (2015-08). Translated to English for gdpark.blog.