The Hydrogen Atom
We finally plug the real hydrogen electric potential into the radial equation, grind through the algebra, and find R(r) — normalization and all.
(image unrelated)
OK so in the last few posts I kept yammering on about hydrogen, hydrogen, hydrogen — but every time the potential $V$ showed up I just wrote “$V$” and moved on. Today? Real hydrogen. For real this time!
Here’s the thing about the hydrogen atom:
It’s symmetric in $\theta$ and $\varphi$.
Which means — we only need to solve the equation in $r$. The Radial Equation. That’s it.
So the goal of this post:
Plug the actual electric potential of hydrogen into the $V$ in the $r$-equation, and find the function of $r$. Find $R(r)$, and while we’re at it, let’s do the normalization cleanly too. That’s the mission.
Alright, let’s go.
The equation we got by substituting $rR = u$ into the $r$-equation — written out again:
$$-\frac{\hbar^{2}}{2m}\frac{d^{2}u}{dr^{2}} \quad+\quad \left[ V \quad+\quad \frac{\hbar^{2}}{2m}\frac{\ell(\ell+1)}{r^{2}} \right] u \quad=\quad Eu$$Now let’s drop the hydrogen potential into that $V$.
Since
$$\nabla \times F \quad=\quad 0$$we can define a potential $V$ via
$$F \quad=\quad -\nabla V$$and for hydrogen, $V$ is
$$V \quad=\quad -\frac{1}{4\pi\epsilon_{0}}\frac{e^{2}}{r}$$(Hydrogen has 1 proton and 1 electron, so the charge product is $e^2$.)
$$-\frac{\hbar^{2}}{2m}\frac{d^{2}u}{dr^{2}} \quad+\quad \left[ -\frac{1}{4\pi\epsilon_{0}}\frac{e^{2}}{r} \quad+\quad \frac{\hbar^{2}}{2m}\frac{\ell(\ell+1)}{r^{2}} \right] u \quad=\quad Eu \\ \quad(\quad E \quad<\quad 0 \quad)$$($E<0$ = the electron is bound. If you want the classical-mechanics intuition, go look at Kepler’s laws.)
Now let’s mess with that equation.
$$-\frac{\hbar^{2}}{2m}\frac{d^{2}u}{dr^{2}} \quad+\quad \left[ -\frac{1}{4\pi\epsilon_{0}}\frac{e^{2}}{r} \quad+\quad \frac{\hbar^{2}}{2m}\frac{\ell(\ell+1)}{r^{2}} \right] u \quad=\quad Eu \\ -\frac{\hbar^{2}}{2mE}\frac{d^{2}u}{dr^{2}} \quad+\quad \left[ -\frac{1}{4\pi\epsilon_{0}}\frac{e^{2}}{r}\frac{1}{E} \quad+\quad \frac{\hbar^{2}}{2mE}\frac{\ell(\ell+1)}{r^{2}} \right] u \quad=\quad u \\ -\frac{\hbar^{2}}{2mE}\frac{d^{2}u}{dr^{2}} \quad+\quad \left[ -\frac{1}{4\pi\epsilon_{0}}\frac{e^{2}}{r}\frac{2m\hbar^{2}}{2m\hbar^{2}E} \quad+\quad \frac{\hbar^{2}}{2mE}\frac{\ell(\ell+1)}{r^{2}} \right] u \quad=\quad u \\ \text{OK let's swap the letters for something cleaner.} \\ K \quad=\quad \frac{\sqrt{-2mE}}{\hbar} \quad\cdot\cdot\cdot\cdot\cdot\cdot\quad E\text{ is negative, so }K\text{ is positive} \\ \frac{1}{K^{2}}\frac{d^{2}u}{dr^{2}} \quad+\quad \left[ \frac{1}{K^{2}}\frac{1}{4\pi\epsilon_{0}}\frac{e^{2}}{r}\frac{2m}{\hbar^{2}} \quad-\frac{1}{K^{2}}\frac{\ell(\ell+1)}{r^{2}} \right] u \quad=\quad u \\ \frac{1}{K^{2}}\frac{d^{2}u}{dr^{2}} \quad+\quad \left[ \frac{1}{K^{2}}\frac{1}{4\pi\epsilon_{0}}\frac{e^{2}}{r}\frac{2m}{\hbar^{2}} \quad-\frac{1}{K^{2}}\frac{\ell(\ell+1)}{r^{2}} \right] u \quad-u \quad=\quad 0 \\ \frac{1}{K^{2}}\frac{d^{2}u}{dr^{2}} \quad+\quad \left[ -1 \quad+\quad \frac{1}{K^{2}}\frac{1}{4\pi\epsilon_{0}}\frac{e^{2}}{r}\frac{2m}{\hbar^{2}} \quad-\frac{1}{K^{2}}\frac{\ell(\ell+1)}{r^{2}} \right] u \quad=\quad 0$$And now some tidying.
Here’s the final tidy:
$$\frac{1}{K^{2}}\frac{d^{2}u}{dr^{2}} \quad+\quad \left[ -1 \quad+\quad \frac{e^{2}m}{2\pi\epsilon_{0}\hbar^{2}K}\frac{1}{Kr} \quad-\frac{1}{K^{2}}\frac{\ell(\ell+1)}{r^{2}} \right] u \quad=\quad 0$$The reason I grouped things that way:
that pink expression — it’s just some constant blob, so let’s rename it:
$$\rho_{0}$$Boom. And then for the variable substitution, because of the blue thing:
$$\rho \quad=\quad Kr$$with that substitution, the coefficient in front of the second derivative vanishes!
Smoothing it out:
$$\frac{d^{2}u}{d\rho^{2}} \quad+\quad \left[ -1 \quad+\quad \rho_{0}\frac{1}{\rho} \quad-\frac{\ell(\ell+1)}{\rho^{2}} \right] u \quad=\quad 0 \\ \frac{d^{2}u}{d\rho^{2}} \quad=\quad \left[ 1 \quad-\quad \frac{\rho_{0}}{\rho} \quad+\frac{\ell(\ell+1)}{\rho^{2}} \right] u \\ \text{ta-da}~$$Well. We got something.
…But it’s another nasty differential equation we have no idea how to solve. (sigh)
Remember what we did for the algebraic solution of the harmonic oscillator? Same trick. Let’s start by hunting for the “asymptotes” of the solution.
$$\begin{cases}{\rho \quad\to\quad \infty}\\{\rho \quad\to\quad 0}\end{cases}$$Let’s look at each limit, one at a time.
$$\rho \quad\to\quad \infty \quad\text{case:} \\ \frac{d^{2}u}{d\rho^{2}} \quad=\quad \left[ 1 \quad-\quad \frac{\rho_{0}}{\rho} \quad+\frac{\ell(\ell+1)}{\rho^{2}} \right] u \quad\text{the purple guys go to 0} \\ \frac{d^{2}u}{d\rho^{2}} \quad\cong\quad u \quad\text{so the equation collapses to this.} \\ \text{and its solution is} \\ u(\rho) \quad=\quad Ae^{-\rho} \quad+\quad Be^{-\rho} \quad\text{but }\rho\to\infty\text{, so we need }B=0. \\ \therefore \quad u(\rho) \quad\cong\quad Ae^{-\rho} \quad(if..\quad \rho \quad\to\quad \infty)$$$$\rho \quad\to\quad 0 \quad\text{case:} \\ \frac{d^{2}u}{d\rho^{2}} \quad=\quad \left[ 1 \quad-\quad \frac{\rho_{0}}{\rho} \quad+\frac{\ell(\ell+1)}{\rho^{2}} \right] u \quad\text{the red guy dominates.} \\ \frac{d^{2}u}{d\rho^{2}} \quad\cong\quad \frac{\ell(\ell+1)}{\rho^{2}}u \quad\text{equation collapses to this.} \\ \text{solution:} \\ u(\rho) \quad=\quad C\rho^{\ell+1} \quad+\quad D\rho^{-\ell} \quad\text{but }\rho\to 0\text{, so we need }D=0. \\ \therefore \quad u(\rho) \quad\cong\quad C\rho^{\ell+1} \quad(if..\quad \rho \quad\to\quad 0)$$$$\rho \quad\to\quad \infty:\quad u(\rho) \quad=\quad Ae^{-\rho} \\ \rho \quad\to\quad 0:\quad u(\rho) \quad=\quad C\rho^{\ell+1} \\ \text{So those are the two limits. Now strip the approximations off and ask: what's the general }u(\rho)\text{?} \\ u(\rho) \quad=\quad G\rho^{\ell+1}\cdot e^{-\rho} \quad\text{something of this shape.} \\ \quad\text{Take the asymptotes again and you land back on the same two limits — consistent.} \\ \quad\text{Buuut — we can't really assume the prefactor is just a constant }G\text{.} \\ \quad\text{So let's give ourselves some room and call it a function }v(\rho)\text{ — a polynomial in }\rho\text{.} \\ \therefore \quad u(\rho) \quad=\quad v(\rho)\rho^{\ell+1}\cdot e^{-\rho}$$OK. So now what?
Now… we pray. lol
Everyone, pray to whatever deity you believe in, in your own way —
“Dear Lord, $u(\rho)$ was already painful to find. Please, I’m begging you, at least let $v(\rho)$ be findable… (sob) I want to find it so bad.”
We take our guessed-at “solution form” and plug it right back into the differential equation.
And there are some derivatives we’ll need —
$$u(\rho) \quad=\quad v(\rho)\rho^{\ell+1}\cdot e^{-\rho} \\ \frac{du}{d\rho} \quad=\quad \frac{dv}{d\rho}\rho^{\ell+1}\cdot e^{-\rho} \quad+\quad v(\ell+1)\rho^{\ell}e^{-\rho} \quad-\quad v\rho^{\ell+1}\cdot e^{-\rho} \\ \quad=\quad \rho^{\ell}e^{-\rho}\left\{(\ell+1-\rho)v \quad+\quad \rho\frac{dv}{d\rho} \right\} \\ \frac{d^{2}u}{d\rho^{2}} \quad=\quad \rho^{\ell}e^{-\rho}\left\{\left[ -2\ell-2+\rho+\frac{\ell(\ell+1)}{\rho} \right] v \quad+\quad 2(\ell+1-\rho)\frac{dv}{d\rho} \quad+\quad \rho\frac{d^{2}v}{d\rho^{2}} \right\}$$Stuff those into the differential equation from above:
$$\frac{d^{2}u}{d\rho^{2}} \quad=\quad \left[ 1 \quad-\quad \frac{\rho_{0}}{\rho} \quad+\frac{\ell(\ell+1)}{\rho^{2}} \right] u \\ \rho^{\ell}e^{-\rho}\left\{\left[ -2\ell-2+\rho+\frac{\ell(\ell+1)}{\rho} \right] v \quad+\quad 2(\ell+1-\rho)\frac{dv}{d\rho} \quad+\quad \rho\frac{d^{2}v}{d\rho^{2}} \right\} \quad=\quad \left[ 1 \quad-\quad \frac{\rho_{0}}{\rho} \quad+\frac{\ell(\ell+1)}{\rho^{2}} \right] v\rho^{\ell+1}\cdot e^{-\rho} \\ \text{clean clean clean} \\ \rho\frac{d^{2}v}{d\rho^{2}} \quad+\quad 2(\ell+1-\rho)\frac{dv}{d\rho} \quad+\quad \left[ \rho_{0}-2(\ell+1) \right] v \quad=\quad 0$$We guessed at the form of the solution and plugged it in. Now all we have to do is find the $v(\rho)$ that satisfies the equation above. How, though?
Same move as the harmonic oscillator’s algebraic solution — let’s just slam it with a power series and brute-force it.
⟨No matter what shape $v$ has, we should be able to express it as a polynomial!⟩
OK, direction’s clear. Let’s run.
$$v \quad=\quad c_{0} \quad+\quad c_{1}\rho \quad+\quad c_{2}\rho^{2} \quad+\quad \cdot\cdot\cdot\cdot \quad+\quad c_{n}\rho^{n} \quad+\quad \cdot\cdot\cdot\cdot$$Like that.
So:
$$\frac{dv}{d\rho} \quad=\quad c_{1} \quad+\quad 2c_{2}\rho \quad+\quad 3c_{3}\rho^{2} \quad+\quad \cdot\cdot\cdot\cdot \quad+\quad nc_{n}\rho^{n-1} \quad+\quad \cdot\cdot\cdot\cdot \\ \quad=\sum_{j=1}^{\infty}{jc_{j}\rho^{j-1}} \quad=\quad \sum_{j=0}^{\infty}{(j+1)c_{j+1}\rho^{j}} \\ \frac{d^{2}v}{d\rho^{2}} \quad=\quad \sum_{j=0}^{\infty}{j(j+1)c_{j+1}\rho^{j-1}}$$Ingredients prepped. Into the wok:
$$\rho\frac{d^{2}v}{d\rho^{2}} \quad+\quad 2(\ell+1-\rho)\frac{dv}{d\rho} \quad+\quad \left[ \rho_{0}-2(\ell+1) \right] v \quad=\quad 0 \\ \sum_{j=0}^{\infty}{j(j+1)c_{j+1}\rho^{j}} \quad+\quad 2(\ell+1)\sum_{j=0}^{\infty}{(j+1)c_{j+1}\rho^{j}} \quad-2\sum_{j=0}^{\infty}{(j+1)c_{j+1}\rho^{j+1}} \quad+\quad \left[ \rho_{0}-2(\ell+1) \right] \sum_{j=0}^{\infty}{c_{j}\rho^{j}} \quad=\quad 0 \\ \text{That }\rho^{j+1}\text{ is a little annoying — but it's fine to re-index like this; it's the same thing plus a }0\text{ at the start.} \\ \sum_{j=0}^{\infty}{j(j+1)c_{j+1}\rho^{j}} \quad+\quad 2(\ell+1)\sum_{j=0}^{\infty}{(j+1)c_{j+1}\rho^{j}} \quad-2\sum_{j=0}^{\infty}{jc_{j}\rho^{j}} \quad+\quad \left[ \rho_{0}-2(\ell+1) \right] \sum_{j=0}^{\infty}{c_{j}\rho^{j}} \quad=\quad 0$$No way $\rho = 0$ is the only solution here lol lol lol (the trivial one, obviously).
The real way for this to hold is for every coefficient to vanish, right? Right!!
Let me color the coefficients once and keep going:
$$\sum_{j=0}^{\infty}{j(j+1)c_{j+1}\rho^{j}} \quad+\quad 2(\ell+1)\sum_{j=0}^{\infty}{(j+1)c_{j+1}\rho^{j}} \quad-2\sum_{j=0}^{\infty}{jc_{j}\rho^{j}} \quad+\quad \left[ \rho_{0}-2(\ell+1) \right] \sum_{j=0}^{\infty}{c_{j}\rho^{j}} \quad=\quad 0 \\ j(j+1)c_{j+1} \quad+\quad 2(\ell+1)(j+1)c_{j+1} \quad-\quad 2jc_{j} \quad+\quad \left[ \rho_{0}-2(\ell+1) \right] c_{j} \quad=\quad 0 \\ \left\{j(j+1) \quad+\quad 2(\ell+1)(j+1) \right\} c_{j+1} \quad+\quad \left\{\rho_{0}-2(j+\ell+1) \right\} c_{j} \quad=\quad 0 \\ (j+1)(j \quad+\quad 2\ell+2) c_{j+1} \quad+\quad \left\{\rho_{0}-2(j+\ell+1) \right\} c_{j} \quad=\quad 0 \\ \therefore \quad c_{j+1} \quad=\quad \left\{\frac{2(j+\ell+1)-\rho_{0}}{(j+1)(j \quad+\quad 2\ell+2)} \right\} c_{j}$$So the condition on the coefficients of $v(\rho)$ that makes
$$u(\rho) \quad=\quad v(\rho)\rho^{\ell+1}\cdot e^{-\rho}$$an actual solution of the DE… is exactly that recurrence relation right above.
Let’s plug in from $j=0$ and see a few terms before moving on:
$$c_{0} \quad=\quad c_{0} \\ c_{1} \quad=\quad \frac{2(\ell+1)-\rho_{0}}{2(\ell+1)}c_{0} \\ c_{2} \quad=\quad \frac{2(2+\ell+1)-\rho_{0}}{2\cdot 2(\ell+1)}c_{1} \quad=\quad \frac{2(2+\ell+1)-\rho_{0}}{2\cdot 2(\ell+1)}\cdot\frac{2(\ell+1)-\rho_{0}}{2(\ell+1)}c_{0} \\ . \\ .. \\ ...$$So the $n$-th coefficient $c_n$ of $v(\rho)$ just keeps going like that.
And “keeps going in $\rho$” = “keeps going in $r$”, same thing.
Which means — BIG which means — $c_n$ has to become $0$ at some point.
“The wave function has to end somewhere.” — (because we need to be able to normalize it!!)
$$\text{At some }c_{j_{max}}\text{ the numerator hits }0 \\ \text{and from }j_{max}+1\text{ on, it's }0,\quad 0,\quad 0,\quad 0\text{ forever.} \\ \text{So from the }(j_{max}+1)\text{-th-degree term of }\rho\text{ onward, every coefficient is zero,} \\ \text{which means the top-degree term of }v(\rho)\text{ has degree }j_{max}. \\ \text{Such a }j_{max}\text{ has to exist!}$$So look at the recurrence again:
$$c_{j+1} \quad=\quad \left\{\frac{2(j+\ell+1)-\rho_{0}}{(j+1)(j \quad+\quad 2\ell+2)} \right\} \cdot c_{j} \\ c_{j_{max}} \quad=\quad \left\{\frac{2(j_{max}+\ell+1)-\rho_{0}}{(j_{max}+1)(j_{max} \quad+\quad 2\ell+2)} \right\} \cdot c_{j_{max}-1} \quad=\quad 0\cdot c_{j_{max}-1} \\ \quad (c_{j_{max}-1} \quad\neq\quad 0) \\ \text{i.e. } \quad 2(j_{max}+\ell+1)-\rho_{0} \quad=\quad 0 \\ \text{has to hold.} \\ 2(j_{max}+\ell+1)-\rho_{0} \quad=\quad 0 \quad\text{call whatever number satisfies this }n. \\ j_{max}+\ell+1 \quad\equiv\quad n$$And this $n$ — this is the principal quantum number! The one everyone knows!!
(Side note: $n - \ell$ has a minimum value of $1$. Right? Because $j_{max}$ can’t be negative. So we get $n > \ell$!)
$$\therefore \quad \rho_{0} \quad=\quad 2n$$Yesss. Done.
We have the Radial Equation in full:
$$u(\rho) \quad=\quad v(\rho)\rho^{\ell+1}\cdot e^{-\rho}$$Now let’s convert all of this back to $r$.
Since $u = rR$:
$$R \quad=\quad \frac{1}{r}v(\rho)\rho^{\ell+1}\cdot e^{-\rho}$$And we also need to swap $\rho$ for $r$. Recall $\rho = Kr$:
$$R \quad=\quad \frac{1}{r}v(Kr)(Kr)^{\ell+1}\cdot e^{-Kr}$$Uh… $K$? What was $K$ again?
$$K \quad=\quad \frac{\sqrt{-2mE}}{\hbar}$$Hold up — stop right here.
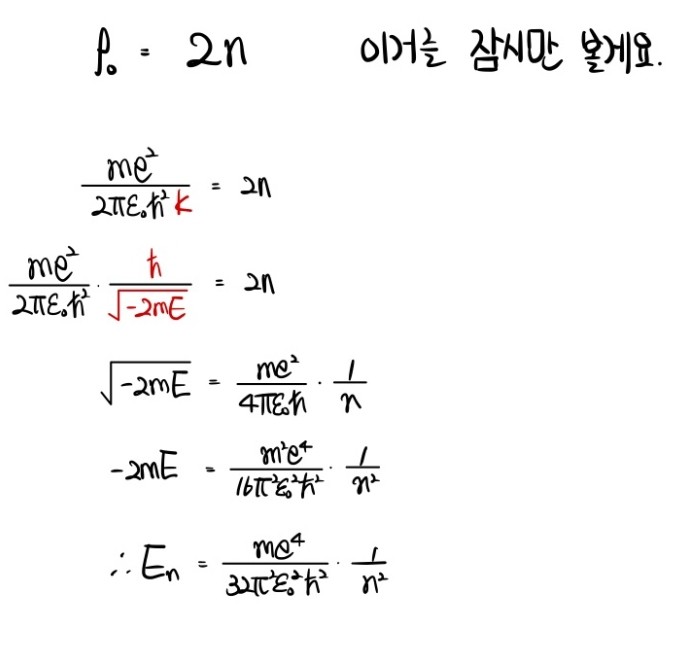
The energy of the hydrogen atom, as a function of $n$.
What’s the energy for $n=1$? That $-13.6$ eV you’ve seen a thousand times — the one basically everyone knows?
That’s where it comes from. Right here.
So anyway — $E$ is now tied to the principal quantum number $n$.
Energy is quantized. That’s what we just learned.
$$K \quad=\quad \frac{\sqrt{-2mE}}{\hbar}$$So this isn’t just $E$ anymore, it’s $E_n$:
$$K \quad=\quad \frac{\sqrt{-2mE_{n}}}{\hbar}$$Therefore:
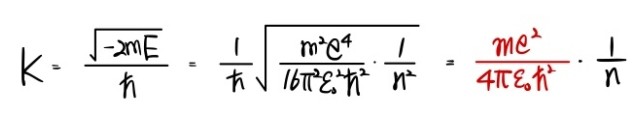
That red thing popping out at the very end —
the reciprocal of it —
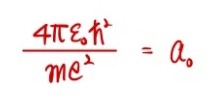
this guy, $a$ with subscript $0$, is called the Bohr radius.
⟨You can derive this pretty easily in a modern-physics course, so just peek over there.⟩
(And honestly — it’s crazy easy to prove using de Broglie’s matter waves. But when Bohr announced this, it was a full 20 years before de Broglie said anything about matter waves. Before the idea of matter waves even existed, Bohr just… somehow knew. Intuitively. Bohr is genuinely — I think he deserves the title “Pope of modern physics.” lol)
⟨Just a story I’m throwing in for fun⟩
(Apparently quantum mechanics only arrived in Korea around the 1960s–70s. By the time Korean physics students were finally learning this stuff, Bohr had already passed away. And supposedly a lot of them wanted to pay their respects somehow — so… a bunch of them named their sons “Bohr”. lol
If there’s any Kim Bohr / Park Bohr / Lee Bohr in your life — try asking them what their dad does for a living. hahahaha
I don’t personally know any Mr. Kim Bohr… heh.
This was a fun little anecdote I picked up in one of my professor’s lectures. Apparently he knows a bunch of Mr. Bohrs. lol)
Anyway, bottom line:
$$K \quad=\quad \frac{1}{a_{0}n}$$That’s it.
$$R \quad=\quad \frac{1}{r}v(Kr)(Kr)^{\ell+1}\cdot e^{-Kr}$$Now I can actually clean this up properly:
$$R \quad=\quad \frac{1}{r}\left(\frac{r}{a_{0}n}\right)^{\ell+1}\cdot e^{-\frac{r}{a_{0}n}}v\left(\frac{r}{a_{0}n}\right)$$One more little tweak —
$R(r)$ is a function that can only be pinned down once $n$ and $\ell$ are fixed, right?! Right!
So let’s flex that by slapping $n$ and $\ell$ on as subscripts, all cool:
$$R_{n,\ell} \quad=\quad \frac{1}{r}\left(\frac{r}{a_{0}n}\right)^{\ell+1}\cdot e^{-\frac{r}{a_{0}n}}v\left(\frac{r}{a_{0}n}\right)$$And just like that, the “completely finding the $r$-equation” game is over!!!
Now — normalization.
Well, as we covered way back, the normalization for the $r$-equation is
$$\int_{0}^{\infty}{R^{2}r^{2}dr} \quad=\quad 1$$— just that.
Hmm. Right now, sitting inside $R(r)$ is this $v(r)$ whose coefficients are defined by a recurrence relation!
That $v$ function —
mathematicians call it the (associated) Laguerre polynomial.
$$\text{Laguerre polynomial} \\ v\left(\frac{r}{na}\right) \quad=\quad L_{n-\ell-1}^{2\ell+1}\left(\frac{2r}{na}\right) \quad\leftarrow\quad \text{'associated Laguerre polynomial'} \\ L_{q-p}^{p}(x) \quad=\quad (-1)^{p}\left(\frac{d}{dx}\right)^{p}L_{q}(x) \\ L_{q}(x) \quad=\quad e^{x}\left(\frac{d}{dx}\right)^{q}\left(e^{-x}x^{q}\right) \quad\leftarrow\quad \text{Laguerre polynomial}$$Now that we know the Laguerre polynomial too —
they say you can actually do that integral by hand. And only now, finally, do we get to write down the fully normalized wave function of the hydrogen atom:
$$\psi_{n,l,m}(r,\theta,\varphi) \quad=\quad R_{n,l}(r)Y_{l,m}(\theta,\varphi)$$$$\psi_{n,l,m}(r,\theta,\varphi) \quad=\quad \sqrt{\left(\frac{2}{na}\right)^{3}\frac{(n-\ell-1)!}{2n[(n+\ell)!]^{3}}}e^{-\frac{r}{na}}\left(\frac{2r}{na}\right)^{\ell}v\left(\frac{2r}{na}\right)Y_{l,m}(\theta,\varphi) \\ \quad=\sqrt{\left(\frac{2}{na}\right)^{3}\frac{(n-\ell-1)!}{2n[(n+\ell)!]^{3}}}e^{-\frac{r}{na}}\left(\frac{2r}{na}\right)^{\ell}L_{n-\ell-1}^{2\ell+1}\left(\frac{2r}{na}\right)Y_{l,m}(\theta,\varphi)$$My goodness, deriving the wave function once is… exhausting………………………
When I’ve got time later, let’s grab a different textbook and try deriving this thing again… heh heh.
It’s absurdly complex, yeah — but apparently for us earthlings this is a very precious equation. There’s literally not a single other case that you can calculate this precisely the way you can with hydrogen. (sob)(sob)(sob)(sob) phew~
(image unrelated)
Originally written in Korean on my Naver blog (2015-08). Translated to English for gdpark.blog.