Two-Particle Systems and the Exchange Force
Finally on Chapter 5 — in QM you literally can't tell two electrons apart, and that one fact changes everything about how we write the wave function.
Welllll… finally. Chapter 5.
Up to now, we basically wrapped up everything about the hydrogen atom. (Which took ridiculously long.)
Now that we know how to handle hydrogen, let’s move past it.
Time to go to helium — 2 protons.
I mean the helium atom — 2 protons, 2 electrons~!
(Full disclosure: we still don’t actually know hydrogen. lol lol lol lol)
(And as you’ll find out while studying it — we can’t do it. lol The one thing that’s certain is: no undergrad is handling helium in any cool way. lol lol lol)
Anyway, before we jump to the 2-particle case, since we’re no longer at “1,” there’s something we need to learn first.
That’s Chapter 5. Identical Particles.
Once you’ve got 2 or more electrons, what’s the new thing we have to think about?!?!!?!
It’s exactly this.
Let me draw it.
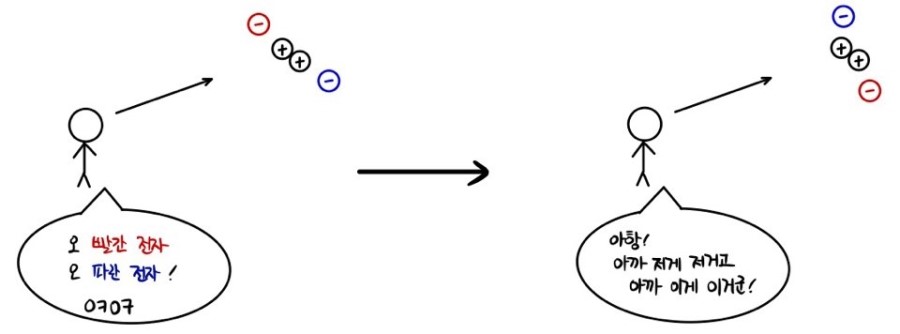
This seems kind of obvious at first — but in quantum mechanics, you can’t do that.
As in —
the stick figure over there is saying this.
In quantum mechanics (unlike classical mechanics), you cannot tell them apart.
That’s why the whole concept of “Identical Particles” got introduced.
Alright~~~ in we go!!!

Say we have a system of two particles.
Particle a is in
$$\psi_{a}\left( r_{1} \right)$$Particle b is in
$$\psi_{b}\left( r_{2} \right)$$(Hold up!!! The fact that I used lowercase psi means we are NOT considering spin yet!!!
So we’re only thinking about the spatial part?!?!
The full state function — spin included — will get an uppercase Psi later. Please mind your Greek: lowercase vs uppercase!!
Sad thing is Naver’s equation editor barely shows a difference between uppercase and lowercase psi. T_T)
The wave function for a system of two particles, each in those states, is
$$\psi \left( r_{1},\quad r_{2} \right) \quad =\quad \psi_{a}\left( r_{1} \right) \psi_{b}\left( r_{2} \right)$$(They call this the product form.)
Ahhh but wait — we just said we don’t know which is which, right?
So we also have to write it like this:
$$\psi_{b}\left( r_{1} \right) \psi_{a}\left( r_{2} \right) \\ \quad :\quad r_{1}\text{ position, }b\text{ particle},\quad r_{2}\quad a\text{ particle}$$Then $\psi \left( r_{1},\quad r_{2} \right)$ is either this one or that one — so call it 1/2 probability each.
Meaning:
$$\psi \left( r_{1},\quad r_{2} \right) \quad =\quad \frac {1}{\sqrt {2}}\left( \psi_{a}\left( r_{1} \right) \psi_{b}\left( r_{2} \right) \quad +\quad \psi_{b}\left( r_{1} \right) \psi_{a}\left( r_{2} \right) \right)$$Ah well — since we absolutely cannot distinguish them, this is all we can do. T_T
But if this really holds, what’s it actually saying?
It’s saying: even if a and b swap spots, the whole system is like, whatever, doesn’t matter~~.
In equation form, that’s
$$\psi \left( r_{1},\quad r_{2} \right) \quad =\quad \psi \left( r_{2},\quad r_{1} \right)$$Except — quantum mechanics is never that nice.
Apparently there are also times when
$$\psi \left( r_{1},\quad r_{2} \right) \quad =\quad -\quad \psi \left( r_{2},\quad r_{1} \right)$$which means
$$\psi \left( r_{1},\quad r_{2} \right) \quad =\quad \frac {1}{\sqrt {2}}\left( \psi_{a}\left( r_{1} \right) \psi_{b}\left( r_{2} \right) \quad -\quad \psi_{b}\left( r_{1} \right) \psi_{a}\left( r_{2} \right) \right)$$Like that…… heh heh heh…… oh my.
(You probably don’t get what this means yet… I didn’t either when I first saw it. But after I ground all the way through to the end and came back to the start — then it finally clicked. heh heh)
So if we bundle them together:
$$\psi \left( r_{1},\quad r_{2} \right) \quad =\quad \begin{cases}{\quad \psi \left( r_{2},\quad r_{1} \right)}\\{-\quad \psi \left( r_{2},\quad r_{1} \right)}\end{cases}\quad$$The top one: symmetric (eigenvalue = +1, symmetry).
The bottom one: antisymmetric (eigenvalue = -1, antisymmetry).
$$\psi \left( r_{1},\quad r_{2} \right) \quad =\quad \begin{cases}{\quad \psi \left( r_{2},\quad r_{1} \right) \quad =\quad \frac {1}{\sqrt {2}}\left( \psi_{a}\left( r_{1} \right) \psi_{b}\left( r_{2} \right) \quad +\quad \psi_{b}\left( r_{1} \right) \psi_{a}\left( r_{2} \right) \right)}\\{-\quad \psi \left( r_{2},\quad r_{1} \right) \quad =\quad \frac {1}{\sqrt {2}}\left( \psi_{a}\left( r_{1} \right) \psi_{b}\left( r_{2} \right) \quad -\quad \psi_{b}\left( r_{1} \right) \psi_{a}\left( r_{2} \right) \right)}\end{cases}\quad$$OK, don’t let your heart stop yet — here’s the punchline.
For 2 bosons (integer S), the state function is symmetric.
For 2 fermions (half-integer S), the state function is antisymmetric.
(Just take it as an experimental fact. But we’re going to use this fact below.
So keep it in mind!!)
Up to here was the spatial story.
Now let’s factor in spin.
Hmm~ we’ll work with two electrons, which are fermions.
(Why? Simple… you’ll see once you keep reading.)
The state function for a “system” made of two electrons is
$$\text{state function for space}\quad \times \quad \text{state function for spin}$$Writing it that way is the finishing touch.
We learned just before that there are 4 total spin states for two electrons.
<We are looking at 2 electrons.>
$$\uparrow \uparrow ,\quad \downarrow \downarrow ,\quad \frac {1}{\sqrt {2}}\left( \uparrow \downarrow \quad +\quad \downarrow \uparrow \right) \quad //\quad \frac {1}{\sqrt {2}}\left( \uparrow \downarrow \quad -\quad \downarrow \uparrow \right)$$4 spin states like this (weren’t they eigenvectors?!).
The reason I went out of my way to color them red, blue, and stick a slash between them is:
Red — those three stay the same even if you swap electron 1 and electron 2!!!!!
So: symmetric.
Blue — that single one. Swap the positions, it picks up a (-1).
“The overall state function” —
(Since we’re dealing with fermions, it has to be antisymmetric.)
(Doing this one by photo. My handwriting tells the story cleaner.)

(The subscript under psi is S for Symmetry, a for antisymmetry ^^)
And one more thing — a ridiculously powerful theorem falls out right here.
The “Pauli exclusion principle” — you’ve heard of it.
Question: “What if two electrons are at the same position?????”
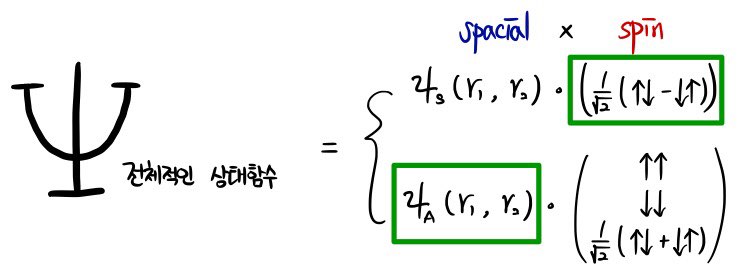
Because of that boxed part, the whole state function goes to zero.
And what does that mean?
It means two electrons can’t be at the same position~.
Which is exactly the Pauli exclusion principle, right?? That’s literally it!! heh heh
OK — let’s get a taste of helium.
This is where we start seeing why helium is kind of a hopeless case.
The Hamiltonian for an atom is
$$H\quad =\quad \sum _{i}^{\quad}{\left( -\frac {\hbar^{2}}{2m}\nabla_{i}^{2}\quad -\quad \frac {1}{4\pi \epsilon_{0}}\frac {Ze^{2}}{r_{i}} \right)}$$Kinetic energy, plus the electric potential with the protons.
If that were all there was, helium and everything past it wouldn’t be hopeless.
The world doesn’t do us that favor. /// Is life just like this?
We have to add this term:
$$+\frac {1}{2}\sum _{i\neq j}^{\quad}{\frac {1}{4\pi \epsilon_{0}}\frac {e^{2}}{\left| r_{i}\quad -\quad r_{j} \right|}}$$It’s the electron-electron potential term!!! And apparently, in most cases, this term straight up can’t be solved.
So we ignore it and solve. (Classic case where ignoring it is fine: silicon.)
But there are also cases where this term matters so much you absolutely cannot ignore it!!!! (Classic case where you cannot ignore it: superconductors.)
So apparently there are lots of ways to approximate it.
But that’s not the point right now!!!!
(Chapters 6, 7, 8, 9 are basically all approximation, so no worries. lol lol lol lol We are going to approximate until we puke.)
OK let’s go!!! You’ll get it once you see it.
With that Hamiltonian, we hit the
(time-independent) Schrödinger Equation
$$H\psi \quad =\quad E\psi$$(The reason we can use the time-independent version is precisely because V is a function of r only.)
We already did this for hydrogen (i = 1). Now let’s think about He.
He’s Hamiltonian is
$$-\frac {\hbar^{2}}{2m}\nabla_{1}^{2}\quad -\quad \frac {\hbar^{2}}{2m}\nabla_{2}^{2}\quad -\quad \frac {1}{4\pi \epsilon_{0}}\frac {2e^{2}}{r_{1}}\quad -\quad \frac {1}{4\pi \epsilon_{0}}\frac {2e^{2}}{r_{2}}\quad +\quad \frac {1}{4\pi \epsilon_{0}}\frac {e^{2}}{\left| r_{1}\quad -\quad r_{2} \right|}$$But since that red thing can’t be solved —
if we just ignore it and push on,
the moment it drops, suddenly it looks like: “Huh?! That’s just two hydrogen atoms?!”
$$-\frac {\hbar^{2}}{2m}\nabla_{1}^{2}\quad -\quad \frac {\hbar^{2}}{2m}\nabla_{2}^{2}\quad -\quad \frac {1}{4\pi \epsilon_{0}}\frac {2e^{2}}{r_{1}}\quad -\quad \frac {1}{4\pi \epsilon_{0}}\frac {2e^{2}}{r_{2}}$$and then
$$\psi (r_{1},\quad r_{2})\quad =\quad \psi_{n,l,m}(r_{1})\cdot \psi_{n,l,m}(r_{2})$$We can write it exactly like that!!!
(Once the red is gone, the Hamiltonian is just electron 1’s Hamiltonian + electron 2’s Hamiltonian, right?)
(It’s some absurd kind of hydrogen. Is it 2 hydrogens sitting at the same spot? Or a hydrogen atom with 2 protons? lol lol lol
Absurd-hydrogen..? What do we even call this thing?)
Back in Chapter 4, the energy of a normal hydrogen atom was
$$E_{n}\quad =\quad -\left\{\frac {m}{2\hbar^{2}}\left( \frac {e^{2}}{4\pi \epsilon_{0}} \right)^{2} \right\} \frac {1}{n^{2}}\quad =\quad \frac {E_{1}}{n^{2}}$$The 2 absurd-hydrogens we’re assuming now have 4× that energy (ground state).
$$E_{1}\quad =\quad -\left\{\frac {m}{2\hbar^{2}}\left( \frac {2e^{2}}{4\pi \epsilon_{0}} \right)^{2} \right\} \frac {1}{n^{2}}\quad =\quad 4\frac {E_{1}}{1^{2}}$$So the energy becomes 4× the old one.
So the ground-state energy of helium, with e-e interaction ignored, is
E = 4 (−13.6 − 13.6) eV ≈ −109 eV
But,,,,!! the actual measured value is about −78.975 eV..
That error is… kinda huge…… 30%?
The only place the gap can be coming from is that red term we threw out…
OK!!! We’re already done here!!!!
Conclusion: “no answer.”
But there’s one more factor that makes helium even more of a no-answer case.
Depending on the situation, that repulsion term can get bigger — or smaller.
And the reason is the exchange force.
What’s the exchange force??????
I’m going to introduce orthohelium and parahelium first, and then circle back to the exchange force.
Let’s look at the ground state of helium.
“Spatially in the ground state” means
$$\psi_{g}(r_{1},\quad r_{2})\quad =\quad \psi_{1,0,0}(r_{1})\cdot \psi_{1,0,0}(r_{2})$$We’ll write it like that.
<Again: we’re ignoring the electron-electron repulsion.>
With Chapter 4 helping us out, this becomes
$$\quad =\quad \frac {8}{\pi a^{3}}e^{-\slash {2\left( r_{1}+r_{2} \right)}{a}}$$The spatial part is symmetric…
Ah — then for the overall state function, the spin part has to be antisymmetric.
Which means the spin state must be the singlet:
$$\Psi \quad =\quad \psi_{1,0,0}(r_{1})\cdot \psi_{1,0,0}(r_{2})\cdot \frac {1}{\sqrt {2}}\left( \uparrow \downarrow \quad -\quad \downarrow \uparrow \right)$$Helium with a singlet spin part like this is called parahelium.
Why specifically parahelium? Because there’s also something called orthohelium.
You’ll see why once we look at the excited state.
Say that in an excited state of helium, one electron is in (1, 0, 0) and the other is in some (n’, l’, m’).
(Apparently if both are in excited states, weird stuff happens, and even then it relaxes to the situation above — one ground, one excited.)
Then the spatial state can be symmetric, or it can be antisymmetric.
Both are allowed.

Both can happen!!!! Because the spin part just needs to make the overall state function antisymmetric on its own!!!!!!
And here too — the one where spin is singlet is called parahelium.
But the one where spin is triplet is called orthohelium!!!
Ah — so why bother splitting it into parahelium vs orthohelium??????
Because apparently there’s an energy difference between them, even though it’s the “same” helium.
They say parahelium’s energy > orthohelium’s energy.
Why????????????????????????????????
Because of the exchange force.
The exchange force is something you can derive through a pretty simple integral calculation on p.197.
Let me write down the definition of exchange force.
Exchange force:
An energy difference between symmetric and antisymmetric states, caused by the exchange force.
symmetry spacial state brings electron closer
That is — a symmetric spatial state pulls the electrons closer, so the potential between them (the one we ignored) gets bigger, so the Hamiltonian gets bigger —
and that’s why parahelium’s energy is higher~
<L e t ’ s g o o v e r t h i s o n e m o r e t i m e.>
Symmetric spatial part ⇒ singlet spin.
Singlet spin ⇒ parahelium.
symmetry spacial state brings electron closer
That is — symmetric means the electrons are pulled closer,
so the potential between them (the one we ignored) gets bigger, so the Hamiltonian gets bigger,
- The one with the bigger Hamiltonian is parahelium.
OK so if we forget about spin and just talk big picture: boson–boson pairs have symmetric spatial parts, so bosons keep trying to get closer to each other because of the exchange force.
To our eyes that looks like attraction.
And fermion–fermion pairs have antisymmetric spatial parts,
so they try to get farther apart, and to our eyes that looks like repulsion!!!
That’s where I’m closing Chapter 5.
This is a part a lot of professors at other schools don’t even lecture on — but
our prof wanted us to hear this chapter at least once, so he covered it briefly
and said he’d focus on Chapters 6, 7, 8, 9. T_T
It kind of feels like Chapter 5 just got hand-waved past…… I’m a little…… T_T (sniff sniff)
Alright — onward to Chapter 6: perturbation theory.
Originally written in Korean on my Naver blog (2015-12). Translated to English for gdpark.blog.