The Pauli Exclusion Principle
Why do metals get more resistive when hot — and why don't electrons freeze when cold? Turns out the Pauli exclusion principle is the whole hero here!!!!
So this is resistivity vs. temperature data for metals.
Stare at this for a sec and you start wondering:
Why does the resistivity of metals go up as temperature goes up? Why does the conductivity drop a bit?????
In other words — why doesn’t current flow as well when things get hot??
OK but this is the kind of thing we’ve all known since high school, right?!
“Because the atoms vibrate.”
Now “temperature going up” here — that’s not the room temperature around you, it’s the temperature of the object itself, yeah?
To say it in the slightly fancier way — when the temperature of an object rises, the energy it holds gets bigger. Thermodynamically we call that internal energy.
And if you try to imagine what that larger internal energy is in our metal above, you might jump to “oh, the electrons are just zipping around with more kinetic energy than before.” But no — the real star here is the lattice vibration energy of the atoms!!!!!
Which is exactly the high school line. The atoms are shaking like crazy, they don’t leave the electrons any room to slip through, and the flowing electrons keep smacking into these jittering atoms — so resistivity goes up and current doesn’t flow well.
And then we can just run that backwards.
Temperature drops → lattice energy drops → the atoms chill out and stop vibrating as much → electrons can cruise through more freely → resistivity shrinks, i.e. resistance shrinks..
But here’s the question I want to throw out:
“If the temperature drops, wouldn’t the electrons’ energy also drop? Shouldn’t the electrons themselves stop moving like they used to??”
“Shouldn’t the electrons freeze up too!!!!!”
Whoaaa… it’s getting a little deep now.
OK, here’s the answer I’ve heard: the reason that doesn’t happen is the Pauli exclusion principle.
Because electrons are fermions.
And fermions, fundamentally, follow Pauli:
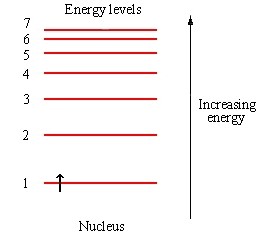
Once one electron plops into a level like this, the next electron
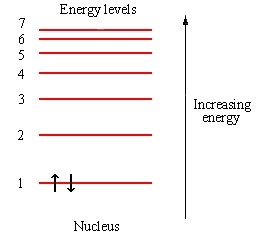
has no choice but to sit up here.
That is the Pauli exclusion principle, right?!?!!!
(This is a fermion thing specifically — worth flagging that bosons do not play by these rules.)
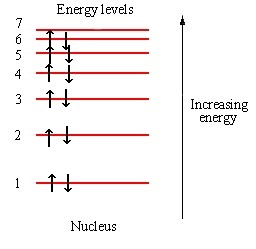
So no matter how far you crank the temperature down, because of Pauli, there are still electrons sitting at high energy levels.
And that’s how you get the result: temperature drops, resistivity drops.
PS.
What I just explained is about a metal, and I did the whole thing with little atoms and discrete energy levels. Which means if you look closely the picture doesn’t quite line up front to back — energy levels as lines aren’t really the right object here.
To do it properly you’d draw an energy band, mark in the Fermi level, and say “there are electrons sitting right at the Fermi level!!!!!” — that’d be cleaner…. heh heh.
But drawing discrete levels and shoving electrons up and down felt easier to picture, so I went with that.
This was actually the content from my March 11, 2016 materials physics lecture hahahaha
And the fundamental difference between fermions and bosons — whether or not they obey Pauli — I was genuinely shocked at myself for having forgotten it, and sprinted to the library right after class to write this up, go go go go.
If I do this now I am absolutely never forgetting it again hahahahahaha
Ah, and also…. there’s actually a pretty delicate piece I haven’t looked into:
The regime where the temperature drops really sharply.
If any of you reading this know what happens down at the extreme low end — like, what’s going on at 0 Kelvin — please teach me.
>_<
I’ll be waiting for your wisdom.
>>__« heh
Originally written in Korean on my Naver blog (2016-03). Translated to English for gdpark.blog.