Chapter 3 Practice Problems
Stumbling through heat, heat capacity, and all their flavors (constant-volume, constant-pressure...) with way too many question marks before tiptoeing into probability.
As I said in #1, now I’ll redefine concepts.
*Heat (Q)
What is heat~ to that question… I… really don’t know.
But, heat seems to be something we should accept as simply ’energy in motion'.
Because,, if you rub your hands, heat is generated in your hands.
The energy from rubbing got converted into thermal energy, right???
Other than rubbing hands, heat generation is accompanied by work,
or you can do work with heat??? I should just keep my mouth shut….T_TT_TT_T
There’s a passage in the book that I was glad to find, so I’ll copy it down and move on.
“Since heat is only meaningful when it is ‘in motion’,
there is no measuring device that can read the amount of heat a body contains, nor can it be read.”
(Later we will learn that a body contains energy.
Therefore, in principle, it is possible to measure the amount of energy a body contains.)
I was glad to read this, accepting that ‘we don’t actually know what heat is to begin with??’ sort of thing.
This is the content of the book, and when I read it I felt even more puzzled looking at this,
but around Chapter 4 I understood a li~~ttle bit of what it was saying.
For now I think I should just move on.
- Heat capacity (C)
Since heat was defined earlier as ’energy in motion’, the commonly used ‘heat capacity’ is said to be a slightly illogical term.
‘Energy capacity’ would be a more correct expression, but historically that word has been used a lot (even while knowing it’s wrong), so let’s just keep using it,
and that heat capacity C is
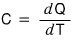
what it means is “how much heat dQ is needed to raise the temperature of an object by dT?”
And let’s also carry along the advice that we should actually look at ’energy’.
However, since this is missing information about ’the amount of the body’,
therefore
we can classify heat capacity as per unit mass, per unit volume, etc.
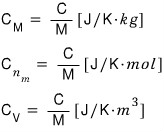
And and and and and there’s this too.
We can also classify heat capacity as constant-volume heat capacity or constant-pressure heat capacity!
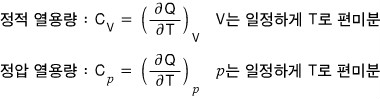
I won’t treat this in detail here.
For now, just note ‘oh, such a thing exists~’ and move on, and later it will be covered in great detail. no worries nope nope nope
And about Chapter 3. Probability
Before starting quantum mechanics, in Chapter 1 of the quantum book I talked a bit about the probability/statistics needed, and
here it’s just exactly the same. So I’m just going to skip it.
But, this is something you learn in high school, and since you’ve probably forgotten, I’ll summarize and move on. (I had forgotten,)
Binomial distribution
Later when we solve problems, I’ll prove it. For now I’ll just throw the results out there.
A discrete probability distribution giving k successes from n Bernoulli trials (ex. coin flips) is P(n, k)!
The mean of this distribution is
The variance is sigma(k).^2 = np(1-p)
There’s also something I didn’t know, but I’ll pass over it as common knowledge.
- The kth moments about the mean
- First moment about the mean
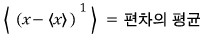
- Second moment about the mean

- Third moment about the mean - skewness
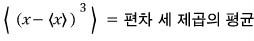
- Fourth moment about the mean - kurtosis
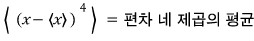
Ex 1.3
Consider a system made of 10 atoms.
These atoms exist in one of two states: energy 0 units or energy 1 unit.
This energy ‘unit’ is called an energy quantum.
In the quantum state with energy 10 and
In the quantum state with energy 4
how many possible quantum arrangements can the system have?
- Don’t ask, don’t argue
- Don’t ask, don’t bicker
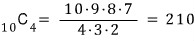
Ex 1.4


Ex 2.1
A 1kW electric heater is turned on for 10 minutes. How much heat is generated?
W (watt) : electrical energy consumed in 1 second : 1W = 1J/s
An electric heater consuming 1kW of power consumes 1000J of energy per second
For 10 minutes, that’s 600 seconds
Over 600 seconds it will have consumed a total of 6000000J.
‘Heat’ is energy…. heat Q = 600kJ
Ex 2.2
At room temperature, the heat capacity of 0.125 kg of water is 523J/K.
Calculate the heat capacity of water a) per unit mass b) per unit volume.
At room temperature, the heat capacity of 0.125kg of water is C = 523J/K
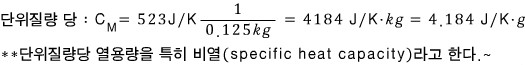

Prob 3.3
There is a discrete probability distribution known as the Poisson distribution.
Let x be a discrete random variable taking values 0, 1, 2, ….
If P(x), the probability, is given as follows, x is said to follow a Poisson distribution.
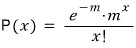


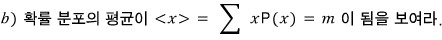

c) The Poisson distribution is useful for describing very rare events that are independent and whose rate of occurrence does not change in our region of interest. For example, the occurrence of congenital disorders measured each year, traffic accidents that happen each year at a particular intersection, typos occurring on a certain page, the number of activations of a Geiger counter occurring per minute, etc. fall into this category.
In fact, the first recorded measurement of the Poisson distribution, motivated by Poisson himself,
was related to a very rare accident such as someone kicked to death by a horse in the Prussian army.
The number of soldiers kicked to death by horses in the Prussian army was recorded over 20 years from 1875 to 1894 in 10 corps, and is as follows.

First, let’s find the probabilities.
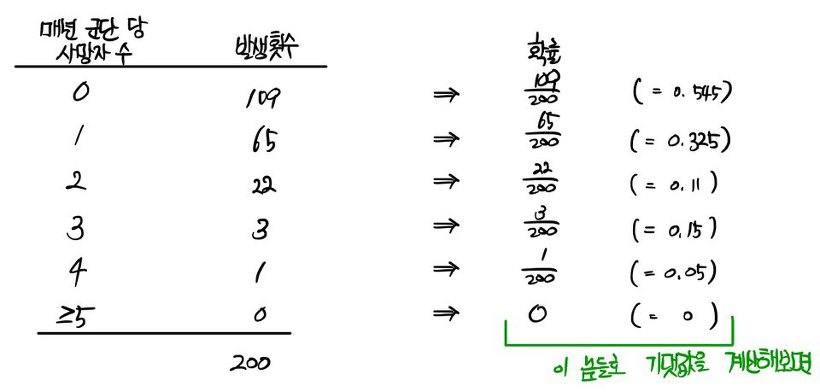

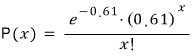
we can say
Let’s check!

Prob 3.4
This problem is about a continuous probability distribution known as the exponential distribution.
Let x be
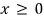
a continuous random variable taking arbitrary values
if
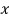
and
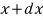
the probability between them is given as follows, this distribution is called the exponential distribution.


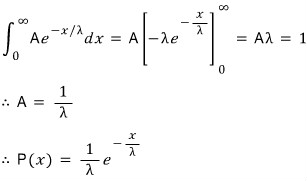
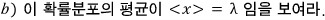
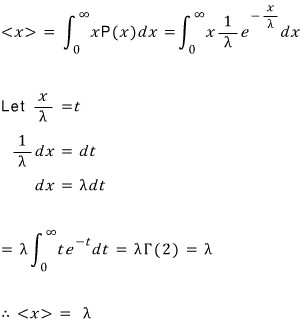
'

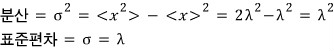
Prob 3.5
θ is a continuous random variable, uniformly distributed between 0 and π.
Now, write the expression for P(θ). And write the values of the following means.

Prob 3.7
For the case where n»1 but np is small, show that the binomial distribution is approximated by the Poisson distribution
(this case represents a situation where p«1, so success is very rare.)
Start!
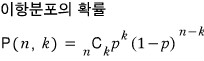
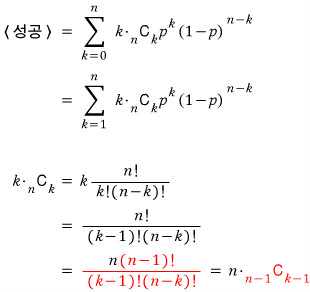


Therefore,

Real start!



Originally written in Korean on my Naver blog (2015-12). Translated to English for gdpark.blog.