Microstates and Macrostates
We kick things off with the Zeroth Law of Thermodynamics — what thermal equilibrium really means, why heat only flows one way, and how that leads us to thermometers!
From here on out, I’m not going to breeze through things!!!!!
I’ll try my best to give explanations that don’t leave you with questions.
If questions do come up, I really, really hope we can discuss them together!
Alright, let’s get started.
The Zeroth Law of Thermodynamics
Say there are two objects at different temperatures.
Now, if the two objects are in thermal contact such that they can exchange energy, our experience will give us the answer.
“(Unless some additional work is done on this system from the outside) ‘heat’ will flow from the hotter place to the colder place~” — is what it tells us.
We already know this, right?
cf.) In the middle of winter, when you leave the door open, the cold air comes into the room.
No — it’s the hot air in the room going out the window.
That’s the correct way to say it. Coldness is merely the ‘absence of heat’ — there’s no such thing as coldness existing on its own.
Alright~ and our experience gives us another answer too.
“The temperatures of the two will become equal~~~!” — is what it tells us.
After a bit of time has passed in this state of thermal contact,
the net heat flow between the two objects becomes zero,
and we say they are in thermal equilibrium.
(To describe it a leeetle more concretely, we can say that the macroscopic properties of two objects in thermal equilibrium no longer change with time.)
And this series of processes going from thermal contact to thermal equilibrium is called thermalization.
To our eyes the thermalization process seems ‘irreversible’ as a matter of common sense,
because we’ve never seen the reverse process, and we can’t even imagine it…
In the end, in chapter 34, this kind of thermal process is apparently used to define the ‘arrow of time’.
Anyway, what we need to squeeze out of this here is: “things in thermal equilibrium → have the same temperature”
This is now summarized as the ‘Zeroth Law of Thermodynamics’.

In the early days of thermodynamics, the content of the 0th law was so obvious that it wasn’t mentioned separately,
but after all the fuss had played out, and since they wanted to mention this to give legitimacy to everything,
they apparently named this — the most fundamental one — the 0th law, giving it a number at the very end.
Thermometer
Temperature, heat — it’s true that these can’t be measured directly,
but having been granted legitimacy by the Zeroth Law of Thermodynamics, we can devise a device to measure them indirectly.
When we want to measure the temperature of something, we bring some ‘somethin’’ that can measure temperature up against it (thermal contact),
wait for thermal equilibrium, and then we can measure the temperature!!!
That somethin’ that lets us measure temperature is called a thermometer.
(Of the many, many thermometers out there, one is the mercury thermometer!!)
When measuring the temperature of an object, for the act of measuring not to change the object’s temperature???
The heat capacity of the thermometer has to be
waaa~!!!!!!y smaller than the heat capacity of the object~~~??? haha
Galileo apparently made a water thermometer in 1593 based on this principle of the Zeroth Law of Thermodynamics.
And the scale we use today was made by Fahrenheit,
and then more refined, upgraded by Celsius.
Ah but, even here we still don’t have an answer to ‘what the heck IS temperature!!!!’…. hahaha ;_;
We need an absolute definition of temperature grounded in physics,
and that definition of temperature will come a bit later. Let’s wait just a little!
Microstate vs Macrostate
To distinguish between the microstate and the macrostate, let’s use an example with five coins to understand.
Let’s say we put these 5 coins into some container.
And we shake the crap out of it.
After we’ve shaken the crap out of it, when we pop! open the lid, what states can exist?

There will be a total of 6 possible states like this. (This is the macrostate)
There are a total of 32 states. (This is the microstate)
What I’m trying to say right now is,
when we pop! open the box with 5 coins in it, the probability of that one exact case — where exactly one comes up tails — coming up is 1/32.
But, the cases where exactly one is tails,
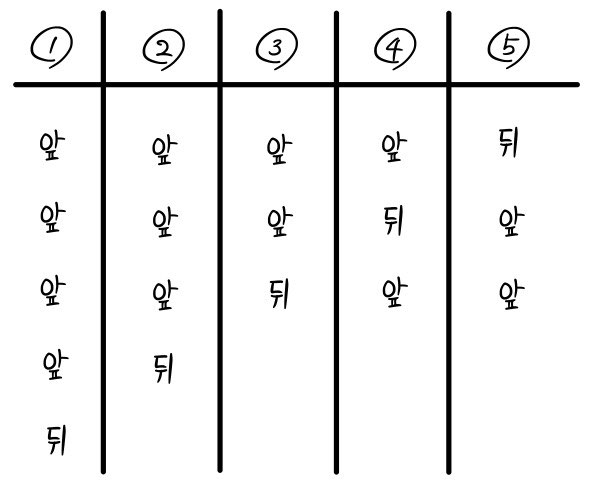
would be one of these 5.
Now,, instead of thinking of them as coins, let’s think of it as a system containing gas molecules.
After shaking the system, the (different) states where only one of the 5 gas molecules is ’tails’ — would their properties or characteristics be different????
nn, they’d be the same.
That is, the probability of this kind of state coming up was 5/32.
In other words, a microstate means assigning numbers to molecules,
and distinguishing them down to the level of molecule #1, molecule #2~~ — that’s the microstate,
while the macrostate is looking at the ‘state’ that appears. (The wording is tough.)
Assigning numbers to each individual molecule is just utterly impossible, and also meaningless…

To add more — macrostates are ’not equally likely.’
Let me toss out a definition of temperature.
Temperature is ‘one of many macrostates’.
The content after this I think is even more important than this, so I’ll push it to the next post.
Originally written in Korean on my Naver blog (2015-12). Translated to English for gdpark.blog.