Chapter 4 Practice Problems
Working through Chapter 4 problems on two-state systems, using the Boltzmann distribution and partition functions to nail expectation values and energy variance.
I think it’s absolutely essential to work through problems to get a grip on the concepts!
Example 4.3 A two-state system
A two-state system, one that has only two states.
One has energy 0, the other has energy ε (>0)
What is the expectation value of this system’s energy?
Earlier, we said that “the probability that an object at temperature T has energy ε” is

we said.
That is, here the two states are
(at temperature T) the probability of having energy 0
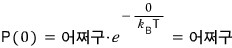
(at temperature T) the probability of having energy ε

We also have to do the normalization.
We can just do it like in quantum mechanics, I guess.
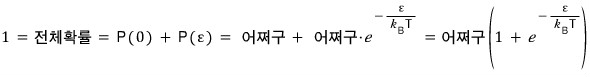
Ugh, this doesn’t work…..
This method doesn’t cut it.
So we’ll do the normalization like this

Let’s build it out.

By expressing the probabilities as a partition function like this, normalization is done, and we can also compute the average.
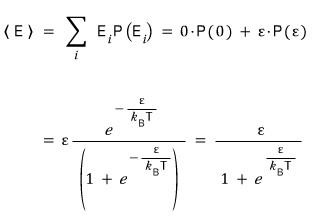
Prob 4.2 For the two-state system described in Example 4.3,
derive an expression for the variance of the energy.
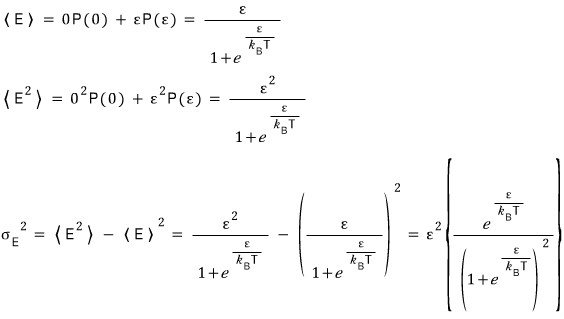
EX 4.4 : Isothermal atmosphere (air)
Roughly estimate the number of molecules in an isothermal atmosphere as a function of height.
For a system at temperature T, the probability of having energy E is
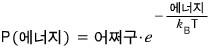
Let’s assume 1 gas molecule. And let’s say the energy here is only the potential energy mgz.
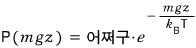
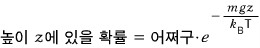
Now let’s say there are many molecules.
Time to cancel the subscription on our “1 molecule” assumption, go go

If we plug in z=0, it should equal n(0).
So I’ll rewrite the constant like this!!!
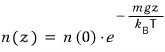
Done.
The higher you go, the more it drops off — whoosh~~~ it drops off~ — at the rate of the Boltzmann factor
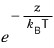
, that’s what it’s saying.
(Actually, since the real atmosphere isn’t isothermal, they say it’s a bit different, and it’s covered in detail in Chapter 12!)
Prob 4.3
A certain system consists of N states, and each state has energy 0 or Δ.
Show that the number of arrangements Ω(E) such that the whole system has energy E = rΔ (r is an integer) is given by the following expression
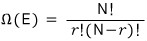
Each of the N is either 0 or Δ, and total energy being rΔ means that r of them are in the energy-Δ state.
That is, the number of ways of choosing r out of N is the number of microstates for E = rΔ!
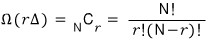
From this system, remove a small energy sΔ. (The relation s«r is satisfied.)
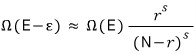
Show this.
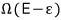
means that out of N, (r-s) are in the energy state Δ.
That is,
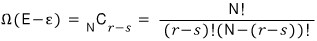
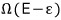
We need to find an approximate expression for this, so we’ll use Stirling’s approximation.
Taking ln of both sides first,
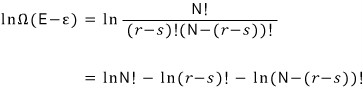
Now let’s approximate the right-hand side.

The temperature T of the system is
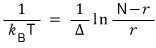
Show this.
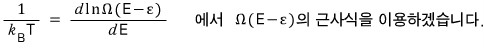
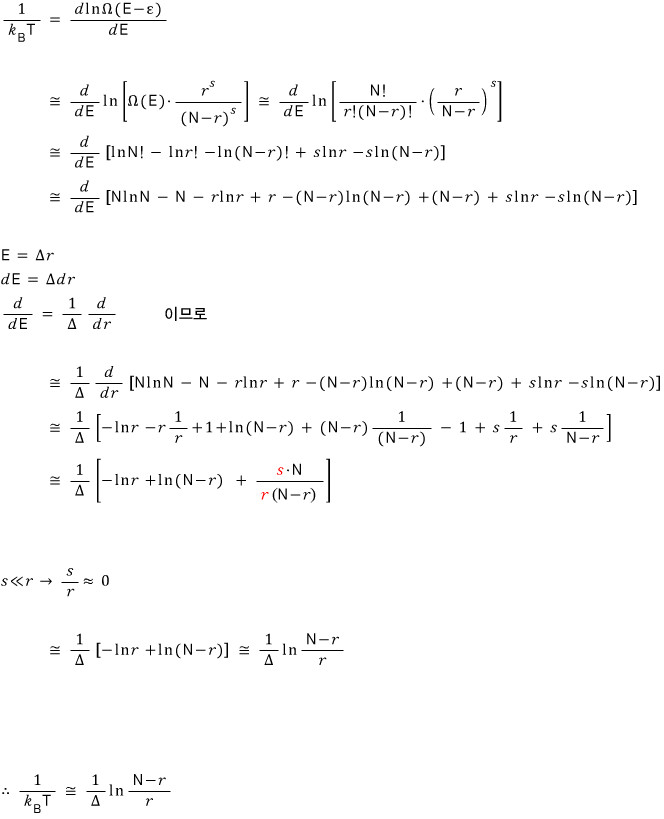
Originally written in Korean on my Naver blog (2015-12). Translated to English for gdpark.blog.