Velocity Distribution Function of Gas Molecules
We finally get to handle something real — gases! Starting from a simple ideal gas model, we derive the velocity distribution using the Boltzmann distribution.
Up until now we haven’t concretely dealt with anything, we just played around with numbers.
The meaning was just… probability? That’s all we did…haha
But from now on we’re going to handle something!!!
Namely, we’re going to handle gases!!
Alright, so to discuss gases, we need to assume a model for the gas we’ll be dealing with.
So we’ll assume a Simple Model.
I’ll just list them out.
All gas molecules are assumed to be monatomic.
For the energies of those gas molecules, rotation and vibration are ignored for now.
The size of the molecule is fucking tiny. Just a point, a point.
So let’s say there are almost no collisions.
Let’s say there are no interactions (universal gravitation, electric force, etc.).
Let’s say the system is closed (we learned canonical, right~)
(oh my, oh my, is this how I say that there’s only kinetic energy?)
The gases inside the Model built with these assumptions will be applied to the ‘Boltzmann distribution’,
and that Boltzmann distribution was about energy, right???
Since we’ve decided that the energy of the gas we’ll apply to the Boltzmann distribution is Only Kinetic energy, that Kinetic Energy is
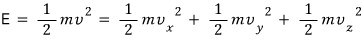
it’ll be like this?!
For now let’s look at just one component!
Let’s look at one component at a time, and then later lump them all together.
Here we go!!!
In a system at temperature T like this, the probability that * a particle has velocity in the x direction of
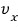
is
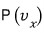
we write it like this and
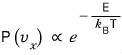
it’ll have this proportionality relationship with energy!!!!
The probability that the x-direction component of velocity is vx!
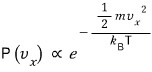
Then if we put N gas molecules into that system?
Out of N, the ratio of those whose x-direction component velocity is
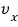
is
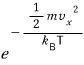
so this says,
out of N, exactly how many have
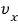
as their velocity!??!?!?!!!
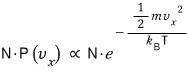
!!!!!!!!!!!!!!!!!!!!!!
So,
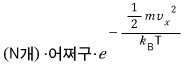
we can say it’s this many, right?!?!!!
(I don’t know what to set the proportionality constant as, so as usual let’s just call the proportionality constant ‘something’.)
That is,
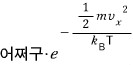
this thing,
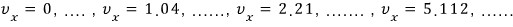
over sooo~~ many cases the sum of those probabilities must be 1, so
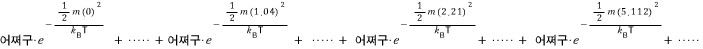
the sum of aaa~~~ll these must be 1, so
ugh, let’s not write it like above.
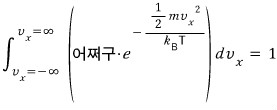
How do we do this integral????
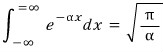
We can do it through the Gaussian integral.
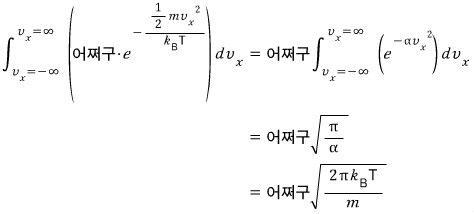
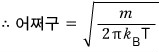
yay~~~ we don’t have to write the veloci’ty’ distribution function as
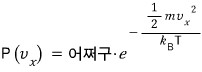
anymore!!
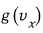
we can just write it like this!!!
Let’s call this veloci’ty’ distribution function
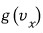
.
As we did before, out of N gas molecules in a closed system, the number with x-direction
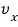
velocity is
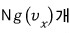
we can say.
Alright, so let’s now wrap up the exploration of the x-direction component and
let’s also explore y and z.
Ugh but, from what we did before,
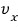
becoming
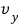
or
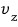
aside, everything~~is the same, so the conclusion will be exactly the same too.
So what we’re going to do is,
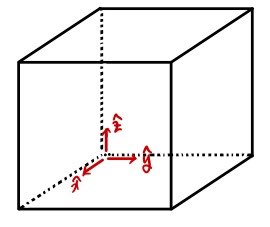
out of N gases inside a system at temperature T, how many gases have
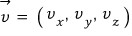
Here's how we'll count them.
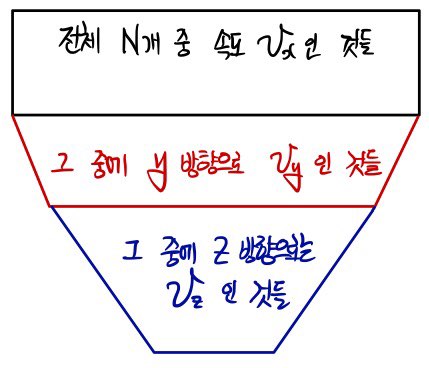
If we sift through like this, out of N in the system, the gases with velocity
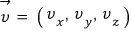
we can count without missing a single one.
If we calculate in the order above,

Does that make sense? T_T T_T T_T T_T T_T whimper
Then by the exact~ same logic as in the 1D case,
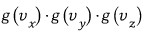
this thing is like a 'ratio' that divides up the whole N.
The sum of those ratios
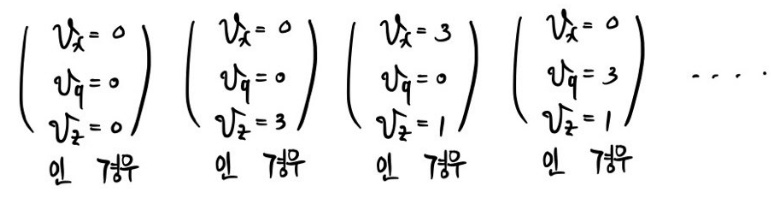
over all these cases, the sum of the ratios?!?!?! must be 1.
If we represent it as a sum, it doesn't work, we can't represent it as a sum,,,,
(because, velocity is a continuous variable!!!!) let's write it as an integral.
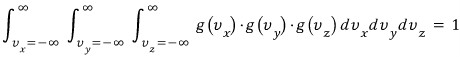
writing it like this should be right~~~hi hi
---
*Originally written in Korean on my [Naver blog](https://blog.naver.com/gdpresent/220584222637) (2015-12). Translated to English for gdpark.blog.*