Maxwell-Boltzmann Speed Distribution
We ditch direction entirely and dive into velocity space to figure out the Maxwell-Boltzmann speed distribution — because who cares which way the gas is flying lol.
Originally, I was going to write all of this content into the previous post,
as one single article, but if I did that, the scroll would be absurdly long as hell,
so reluctantly, I’ll write the speed distribution function anew.
However, the content continues from the previous post http://gdpresent.blog.me/220584222637 !!! Let us go ~ !
Alright, this time it’s ‘speed’.
Velocity is meaningless in our inquiry^^
(I mean, the gas is moving, but what does direction even mean^^ we can’t even see it lolol)
We first learn velocity easily, and based on that, we head toward ‘speed’ which is more meaningful to us!!!!
Without considering direction at all, we want to know the fraction(?), the distribution(?) of gases moving with only that speed.
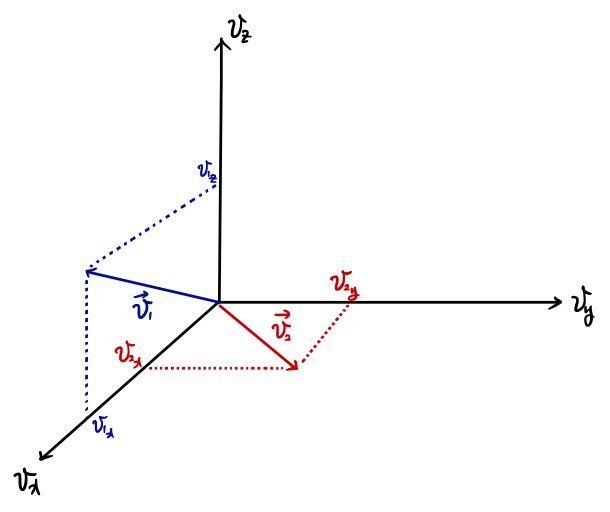
To resolve this curiosity, they say it’s more convenient to view things not in Real Space but in Velocity space.
Because, for example, if we want to know about gases moving with speed v = 2,
among the gases included here

That is, every single one of them
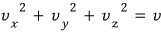
will have the same value, right?!?!?!
So we
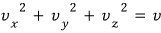
have to account for all such gases whose values are equal,
and to look at that, it’s much more convenient to view it in Velocity Space rather than Real Space.
Please listen carefully to what I’m saying!!
In a system at temperature T containing N gas particles,
among those with speed
there will be some with..
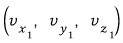
such ones.
Out of N, how many will there be?
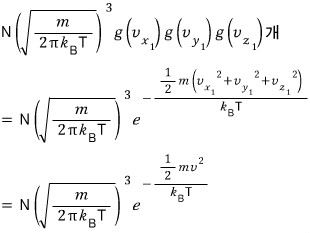
That many will exist. (The constant in front was covered in the previous post.)
In a system at temperature T containing N gas particles,
among those with speed
there will be some with..
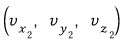
such ones.
Out of N, how many will there be?
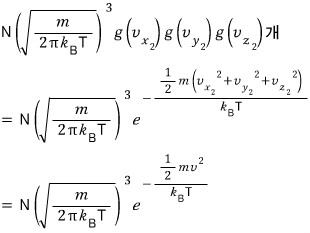
That many will exist.
In a system at temperature T containing N gas particles,
among those with speed
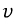
there will be some with..
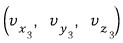
such ones.
Out of N, how many will there be?
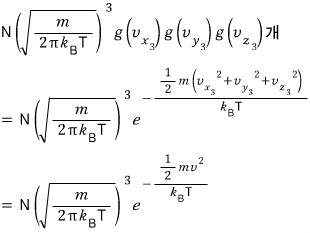
That many will exist.
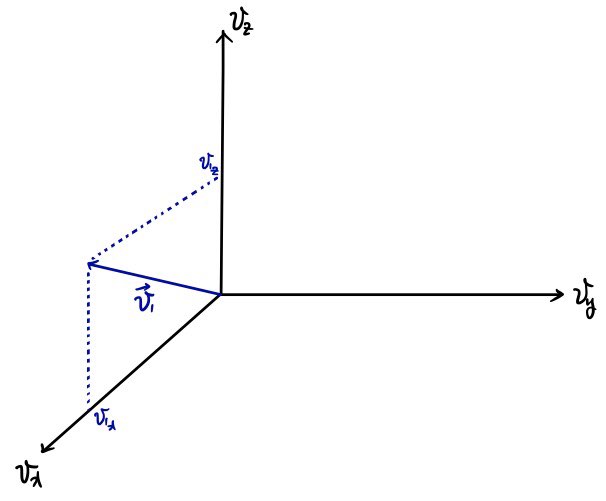
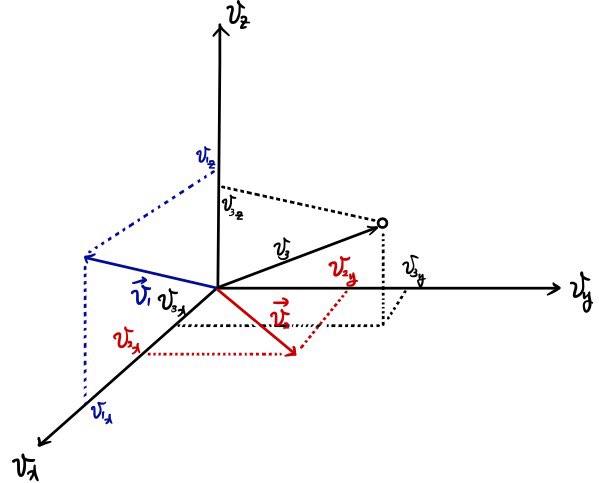
If we plot every case with speed
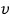
as dots in velocity space allllll over????
it will form a sphere.
In a system at temperature T containing N gas particles,
the number of those with speed
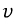
is
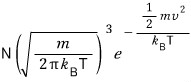
added up ‘by the sphere’, and that will be the true count.
Adding up by the sphere means we need to compute by the sphere’s surface area, right?
The radius of that sphere is
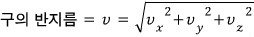
so,
the surface area of the sphere will be
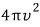
.
Adding up by the sphere’s surface area gives
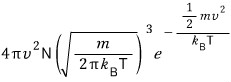
In a system at temperature T containing N gas particles,
this is the number of gases with speed
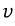
.
Okayokayokayokayokay refresh!!! Let’s wrap things up.
In a system at temperature T, the number with speed v is
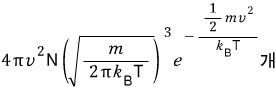
.
This now ends up meaning something like this.

If we add up all these counts~~~~~~~~~~~~~~~~~`
we should again get N, the number of gas particles in the system at temperature T, right!!!!???
We need to check it.
What we need to check is,
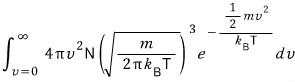
let’s check if this comes out to 1!!! (v is a continuous variable here too, so I’ve done the total sum via an integral!!)

The integral I used above, I’ll post separately.
It’s a waste of page space to write it here lololol
I guess I’ll write up the integral in the very next post and link it.
Alright, so
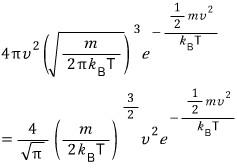
the Physical meaning of this is!!!!
distribution? fraction! is what we can call it!
Because it represents what (%) of N particles have speed v~ !!!!
This is called the Maxwell-Boltzmann speed distribution (Maxwell-Boltzmann Distribution).
Let me write this speed distribution simply, differently from what we wrote earlier as
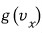
, as
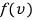
!!!!
Originally written in Korean on my Naver blog (2015-12). Translated to English for gdpark.blog.