Expected Speed, Expected Energy, and Statistical Analysis of Ideal Gas Speed
We ditch talking distributions and actually calculate averages — finding the expected speed and energy of an ideal gas molecule using integrals and odd-function tricks.
Alright, so let’s stop talking about distributions now. Now we’re going to look at averages,
So let’s look at velocity again.
This time, let’s again put ‘one’ gas molecule into a system at temperature T.
The probability that this one molecule has velocity
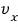
is
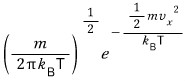
.
This was organized already.
(from. http://gdpresent.blog.me/220584222637)

What I studied in thermo/statistical mechanics #7. Gas velocity distribution function, velocity…
Up till now I haven’t dealt with anything concretely, just played a numbers game. The meaning is well, probability? Just that much…
blog.naver.com
Now we’re going to Measurement one molecule in a system at temperature T!
Let’s find the expectation value for that measurement.
That expectation value is
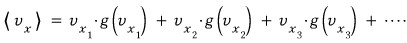
you just sum them all up like this!!!!!!
But again, since it’s a continuous variable, let’s find the total sum via an integral.

Aha, so the average of velocity
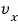
is 0
makes sense…. hehehe
Ah damn!!! This isn’t what I was curious about! Let’s find the average of ‘speed’
This thing, this one
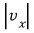
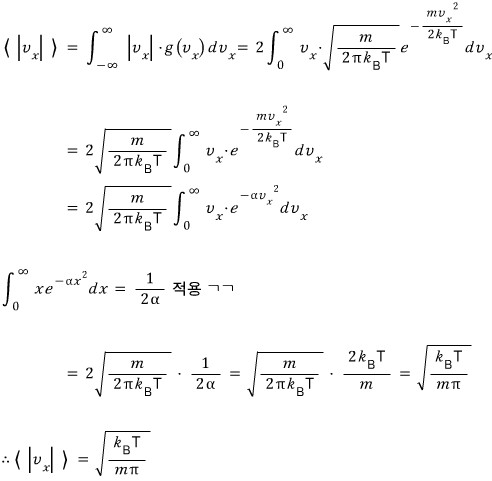
We integrate the earlier function from 0 to infinity,
and multiply by 2
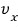
we can call this the average of ‘speed’!!!!!!
Let’s also get the average of speed squared!!!!!!

So like this,
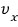
I’ve done everyyyything for this, but this was just practice ^^
It was practice for what we’re about to do.
What’s more meaningful for us is finding the expectation values of each kind for the direction-independent speed.
Earlier it was
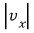
this, and now we’re doing it for direction-doesn’t-matter
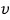
this speed.
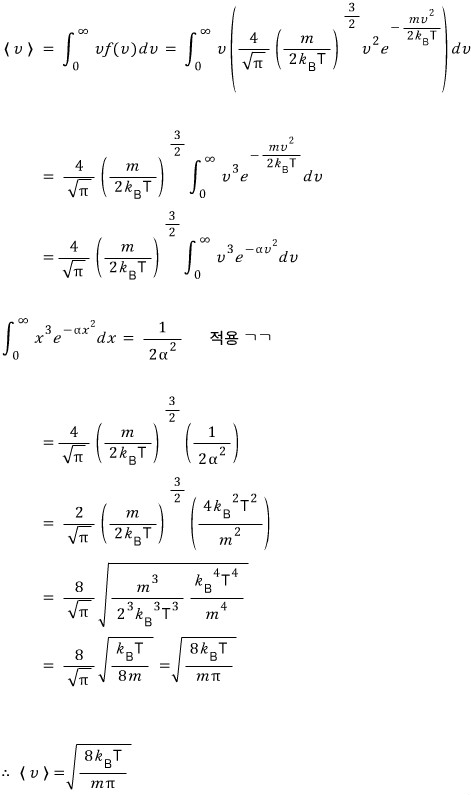
It looks somewhat complicated, but since you just need to do a little cancel-to-the-ation, it’s not hard.
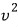
Let’s also find the average of
.
Since we have a kinda-formula-kinda-not-formula, there’s nothing to be scared of.

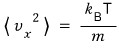
Do you remember this came out?!?!?!?!?!!!!
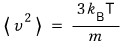
the fact that this came out means
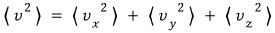
this was the case!!!
And to the mean of the square, to kill off the weight of the squaring, if you “put a square root on it…”
that’s exactly the root-mean-square(rms) value!!!
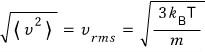
They say to pay attention to the fact that the rms of speed is proportional to the 1/2 power of mass!!!
(This is something we learned as Graham’s law of effusion in chemistry class, and it’s going to come up later for us ^^
Since it’s content that’ll appear then, I think that’s why they told us to pay attention.)
Let’s also find the average of kinetic energy
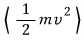
!!!!
This is a calculation we’ve already done above~~~kekeke
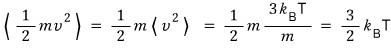
I learned this in Physics 2 class!!!!
The kinetic energy of a gas is dependent ONLY on ’temperature’!! is what we learned! hehe
The integrals needed in the ‘apply go go’ in the above integrations are at
http://gdpresent.blog.me/220584291592

What I studied in thermo/statistical mechanics #9. Proofs of integrals frequently used in thermo/statistical mechanics (link-only)
I’m not going to derive this one! Because it’s easy — skipping this, and by taking the derivative with respect to α on both sides of the resulting equation, thermo/stat…
blog.naver.com
Don’t forget.
The assumption of an ideal gas came along,
and the assumption of following the Boltzmann distribution came along.
Originally written in Korean on my Naver blog (2015-12). Translated to English for gdpark.blog.