Ideal Gas Equation of State and Pressure
Deriving PV=nRT from a microscopic view — but first we have to survive solid angles, which are exactly as weird as they sound.
OK, time to dive into Chapter 6. Pressure.
Although honestly… calling it “pressure” is being generous — really the whole chapter is just $PV=nRT$.
The goal is to derive the ideal gas equation of state from a microscopic point of view. That’s it. That’s the chapter.
Before we go in, let me copy down the textbook’s “definition of pressure” word-for-word, and then we’ll dive in.
Pressure $p$ is defined as the ratio of the normal contact force to the contact area.
So the unit is force (N) over area (m²), and we call it Pa (pascal). $[\text{Pa} = \text{N}/\text{m}^2]$
The direction pressure acts in is perpendicular to the surface (and it’s always acting on it).
That word “perpendicular” feels important. OK, let’s go in.
Solid angle first
The first thing we need to arm ourselves with is the solid angle.
I picked this up a little bit in mathematical physics, so I’ll just zip through it.
A solid angle is… this kind of weird angle. Like a — I don’t know, a little ice cream scoop angle? lol
Anyway, here’s how it’s defined.
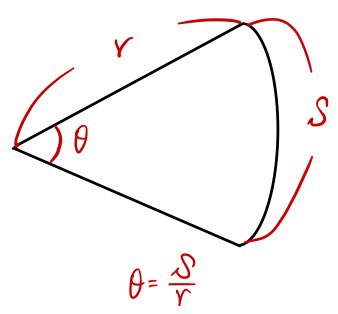
so
$$\theta = \frac{S}{r}$$In the same way we say that,
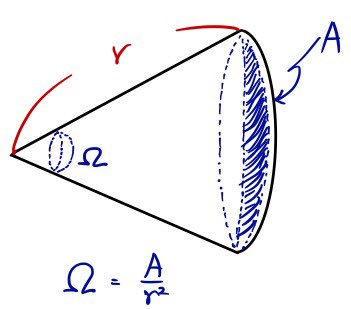
— θ takes whatever it’s looking at and divides by the radius —
Ω takes the curved patch it’s looking at and divides by the radius squared.
Now then. We need to think about this next part.
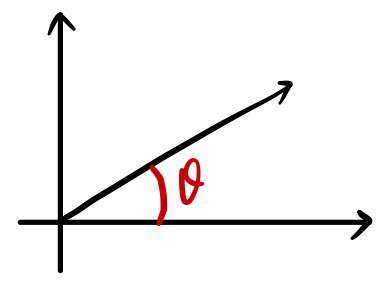
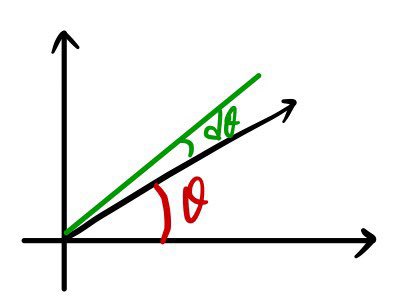
We wrote down $d\theta$ relative to $\theta$ like this, no problem at all. Easy.
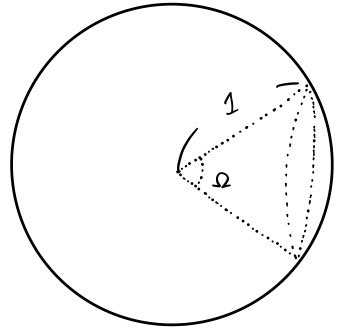
So what about $d\Omega$ relative to $\Omega$?!
At first I couldn’t quite picture it, but if you stare at it for a second —
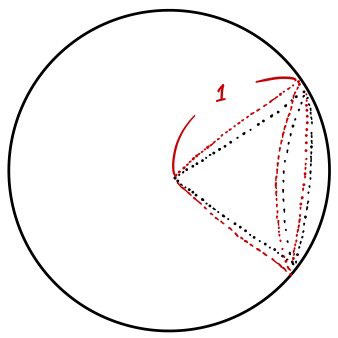
— that red bit there is $d\Omega$, right???
(Drawing pictures is really… hard…)
In that picture, the solid angle of the space sandwiched between the outer cone and the inner cone is $d\Omega$. Right?!
(Side note: “cone” is the wrong word — the base isn’t a circle, so technically it’s not a cone. But there’s no cleaner word for it… shudder.)
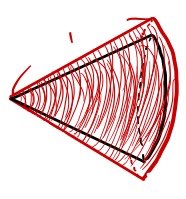
OK, so to express $d\Omega$ in terms of radius and area, we need to take the area that $d\Omega$ is looking at, and divide by the radius squared.
So let’s figure out that area!
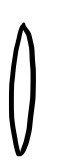
&&&

Left picture — the circumference of the circle is
$$2\pi \left( 1\cdot \sin\theta \right)$$Right picture — multiply by the thickness too
$$2\pi \left( 1\cdot \sin\theta \right) d\theta$$So the area the solid angle is looking at is
$$2\pi \left( 1\cdot \sin\theta \right) d\theta$$And since the solid angle is that area divided by the radius squared,
$$d\Omega = 2\pi \left( 1\cdot \sin\theta \right) d\theta / 1^{2} = 2\pi \left( 1\cdot \sin\theta \right) d\theta$$Whew. “Why are we even learning this?”
OK, let me explain why we need the solid angle, and then use it as our weapon to derive pressure.
Setting up the sphere
Picture this.
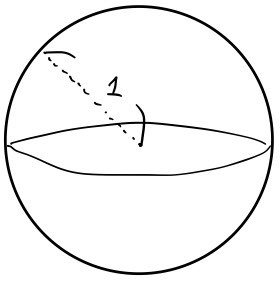
A sphere of radius 1 — that’s our system. Temperature $T$.
Inside, there are an absolutely enormous number of gas molecules, going in tens of thousands of directions with tens of thousands of speeds.
Say there are $N$ of them in total.
Out of those $N$, let’s pluck out only the molecules whose speed is exactly
$$v$$How many do we pluck out?
Yep, that’s right.
$$N\left( \frac{4}{\sqrt{\pi}}\left( \frac{m}{2k_{B}T} \right)^{\frac{3}{2}}v^{2}e^{-\frac{mv^{2}}{2k_{B}T}} \right) \text{ (particles)}$$That many.
These guys all have the same speed, but each one is moving in a different direction.
So here’s what we’re going to do — just with these plucked-out ones —
we drop them into another identical sphere of radius 1,
BUT!!!!!!!!!!
we rearrange them so that each molecule’s direction of motion lines up with the radius vector pointing out to where it sits.
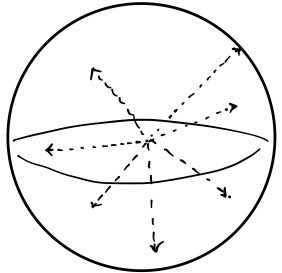
Yeah… I’m not sure what the smartest way to draw this is… (sob)
Just imagine the gases have been rearranged so each one’s direction of motion = the radius vector at its location.
The reason we learned the solid angle is so we can use $d\Omega$.
Why?!
Because all the gas molecules sitting inside one $d\Omega$ — after the rearrangement — have something in common!
Specifically: for all the molecules inside that $d\Omega$, the value of $\cos\theta$ is the same!
(Let’s call that direction “rightward.”)
Their rightward component of velocity is the same. That’s what this means!
So out of the ones we plucked out,
$$N\left( \frac{4}{\sqrt{\pi}}\left( \frac{m}{2k_{B}T} \right)^{\frac{3}{2}}v^{2}e^{-\frac{mv^{2}}{2k_{B}T}} \right) \text{ (particles)}$$how many have the same speed component in a specific direction (→) — i.e., the same value of $\cos\theta$? Let’s count them!
$$N\left( \frac{4}{\sqrt{\pi}}\left( \frac{m}{2k_{B}T} \right)^{\frac{3}{2}}v^{2}e^{-\frac{mv^{2}}{2k_{B}T}} \right) \text{ (particles)} \times \frac{d\Omega}{\text{total solid angle}} \\ \text{The total solid angle is } 4\pi. \\ = N\left( \frac{4}{\sqrt{\pi}}\left( \frac{m}{2k_{B}T} \right)^{\frac{3}{2}}v^{2}e^{-\frac{mv^{2}}{2k_{B}T}} \right) \text{ (particles)} \times \frac{2\pi\sin\theta\, d\theta}{4\pi}\\ = N\left( \frac{4}{\sqrt{\pi}}\left( \frac{m}{2k_{B}T} \right)^{\frac{3}{2}}v^{2}e^{-\frac{mv^{2}}{2k_{B}T}} \right) \frac{1}{2}\sin\theta\, d\theta \text{ (particles)}$$OK now let’s go back to before the plucking-out.
Gases tangled up with all sorts of jumbled speeds and directions —
we’re back to the spherical system that’s a total lawless wasteland.
If the molecules in that sphere have no preferred speed or direction, then per unit volume of sphere, the number of molecules with speed $v$ and the same speed component in the (→ rightward) direction is:
$$\frac{N\left( \frac{4}{\sqrt{\pi}}\left( \frac{m}{2k_{B}T} \right)^{\frac{3}{2}}v^{2}e^{-\frac{mv^{2}}{2k_{B}T}} \right) \frac{1}{2}\sin\theta\, d\theta \text{ (particles)}}{V} = n\left( \frac{4}{\sqrt{\pi}}\left( \frac{m}{2k_{B}T} \right)^{\frac{3}{2}}v^{2}e^{-\frac{mv^{2}}{2k_{B}T}} \right) \frac{1}{2}\sin\theta\, d\theta \text{ (particles)}$$(I just set $n = N/V$ ~)
Stick a plate in there
Now let’s drop a plate of area $A$ (unit area) somewhere inside the sphere.
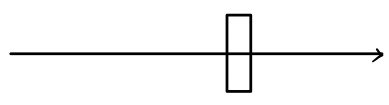
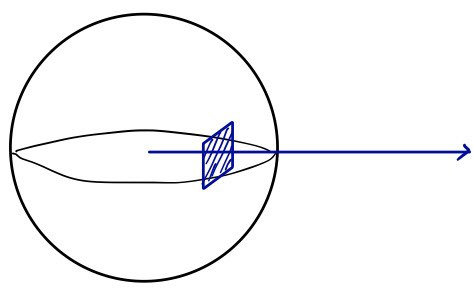
And let’s count how many molecules slam into that plate.
<That’s why we just counted — per unit volume — the molecules with speed $v$ and the same velocity component in the → direction.>
The plate’s getting hammered by molecules with tens of thousands of speeds in tens of thousands of directions.
Question time:
“How many gas molecules hit a plate of unit area $A$ during unit time $dt$?”
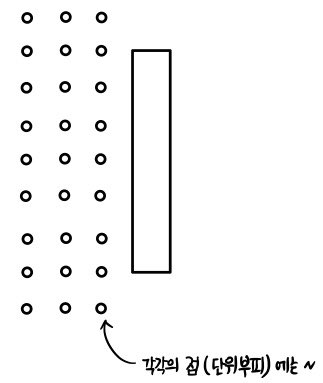
At each point (each unit volume), the number of molecules with speed
$$v_{1}$$and (→) component (with respect to the plate)
$$v_{1}\cos\theta_{1}$$is
$$n\left( \frac{4}{\sqrt{\pi}}\left( \frac{m}{2k_{B}T} \right)^{\frac{3}{2}}v_{1}^{2}e^{-\frac{mv_{1}^{2}}{2k_{B}T}} \right) \frac{1}{2}\sin\theta_{1}\,d\theta \text{ (particles)}$$each. And the molecules that collide during $dt$ — we get them by bundling those points together.

That is,
$$\text{volume} \times \frac{\text{(count)}}{\text{volume}} = \text{(count)}$$That’s how we count.
So “the number of gas molecules hitting a plate of unit area $A$ with speed $v_1$ during unit time $dt$” is
$$\left( Av_{1}\cos\theta_{1} \right) \left( n\left( \frac{4}{\sqrt{\pi}}\left( \frac{m}{2k_{B}T} \right)^{\frac{3}{2}}v_{1}^{2}e^{-\frac{mv_{1}^{2}}{2k_{B}T}} \right) \frac{1}{2}\sin\theta_{1}\,d\theta \right) \text{ (particles)}$$But — what’s actually hitting the plate isn’t just $v_1$. It’s tens of thousands of different velocities.
And it’s not just $\theta_1$ either — they’re flying in at dozens of different angles.
So for all those numbers, for all~~~ the cases like $v_1, v_2, v_3$, and all~~~~~ the angles — we add them all~~~~ up, and that’s the true number of gas molecules hitting the plate during $dt$. Right???
And now… pressure
Now we can find pressure per unit time per unit area.
Integrate over all angles $\theta$ and all speeds $v$ to get the count, and for each of those gas molecules, multiply by the change in momentum from its collision and add it all up — and we get something with the units of force.
<Ideal gas assumption → perfectly elastic collisions, so the change in momentum per collision is $2mv\cos\theta$. We multiply by that and sum over all~ cases. Change in momentum = impulse = $F\cdot dt$.>
$$\begin{aligned} \text{pressure}\quad p &= \iint \left( \text{change in momentum} \right) \times \text{(count)} \\ &= \iint \left( 2mv\cos\theta \right) \times \left( v\cos\theta \cdot n\frac{4}{\sqrt{\pi}}\left( \frac{m}{2k_{B}T} \right)^{\frac{3}{2}}v^{2}e^{-\frac{mv^{2}}{2k_{B}T}}\frac{1}{2}\sin\theta\, d\theta \right) d\theta\, dv \\ &= mn\int_{v=0}^{\infty}\int_{\theta=0}^{\frac{\pi}{2}} \left( 2v\cos\theta \right) \times \left( v\cos\theta \frac{4}{\sqrt{\pi}}\left( \frac{m}{2k_{B}T} \right)^{\frac{3}{2}}v^{2}e^{-\frac{mv^{2}}{2k_{B}T}}\frac{1}{2}\sin\theta\, d\theta \right) d\theta\, dv \end{aligned}$$Since $dt = 1$ and $A = 1$, just summing all of this up does the job.
Why does $\theta$ only go from $0$ to $\pi/2$? Because going past that means counting molecules that don’t even hit the plate. Only up to $\pi/2$ is correct!
And lucky for us, nothing inside that integral is tangled up — we can pull it apart into separate integrals.
$$mn\int_{v=0}^{\infty}\int_{\theta=0}^{\frac{\pi}{2}} \left( 2v\cos\theta \right) \times \left( v\cos\theta \frac{4}{\sqrt{\pi}}\left( \frac{m}{2k_{B}T} \right)^{\frac{3}{2}}v^{2}e^{-\frac{mv^{2}}{2k_{B}T}}\frac{1}{2}\sin\theta\, d\theta \right) d\theta\, dv\\ = mn\int_{v=0}^{\infty} v^{2}\frac{4}{\sqrt{\pi}}\left( \frac{m}{2k_{B}T} \right)^{\frac{3}{2}}v^{2}e^{-\frac{mv^{2}}{2k_{B}T}}dv \int_{\theta=0}^{\frac{\pi}{2}} \cos^{2}\theta\sin\theta\, d\theta$$Hold up!! Before we actually do the integral — in the expression above, the blue chunk is $\langle v^{2} \rangle$ and the red chunk evaluates to $1/3$, so
$$p = \frac{1}{3}nm\langle v^{2} \rangle$$Let’s just tuck that in our pocket and keep going~~ heh.
OK, the actual integral comes out like this.

So pressure depends only on the number of molecules per volume $n$, and on the temperature $T$!
Rewriting with $n = N/V$:
$$p = \frac{N}{V}k_{B}T\\ pV = Nk_{B}T$$Wait, what?!?!? In chemistry class we learned $pV = nRT$?!?!?!
OK so the $n$ in the chem-class ideal gas equation is not the same $n$ we’ve been using here.
Our $n$ has been “number of molecules per unit volume.”
The $n$ in the chem-class ideal gas equation should really be written
$$n_{m}$$Why?! Because in chem class it wasn’t a count — it was a number of moles.
I mean, sure, a mole is also a count of sorts. But the unit is different!
In $n_m$, the subscript $m$ is not mass — it’s mol! That is,
$$n_{m} = \frac{N}{N_{A}}\\ N_{A} : \text{Avogadro's number} = 6.022\times 10^{23}/\text{mol}$$The unit of “count” just got swapped out for the unit “mol.”
Plug that in and clean up:
$$pV = Nk_{B}T = n_{m}N_{A}k_{B}T$$Ugh……… still no $pV = nRT$. (annoyed face)
The gas constant $R$ from chem class was actually two constants holding hands the whole time — Avogadro’s number and Boltzmann’s constant!
That is, $R$ is defined as Avogadro’s number times Boltzmann’s constant.
$$pV = n_{m}RT$$I’m changing my major.
I cannot make a living doing this.
Originally written in Korean on my Naver blog (2016-01). Translated to English for gdpark.blog.