Chapter 6 Practice Problems
Working through Chapter 6 practice problems on ideal gas volume at STP, kinetic energy density, and what happens to the air in your room when you turn up the heat.
Ex 6.2
What volume does 1 mol of ideal gas occupy at STP (standard temperature & pressure)?
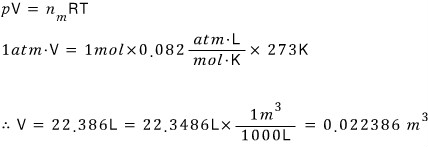
Ex 6.3
What is the relation between pressure and kinetic energy density?
The sum of the kinetic energies of n particles contained in unit volume can be called the kinetic energy density (u).
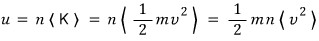
When we derived pressure p (in the previous post http://gdpresent.blog.me/220584695823)
My study notes on thermal/statistical mechanics #11. Ideal gas equation of state, pressure
Here we’re going into ‘Chapter 6. Pressure’. Rather than saying we’re learning pressure, it’s just the PV=nRT ideal gas state…
blog.naver.com

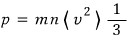
we established this.
Therefore,
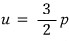
Prob 6.2
Calculate the kinetic energy density (u) for air at atmospheric pressure.

Prob 6.3
Mr. Fourier is sitting in a living room at 18 degrees Celsius.
He thinks it’s a bit cold, so he adjusts the heating so the temperature becomes 25 degrees Celsius.
What happens to the total energy of the air in the living room? (hint: what controls the pressure in the room?)

Prob 6.5
a) If the number of molecules hitting per unit area, per unit time, with the angle made with the direction perpendicular to the surface between θ ~ θ+dθ and the speed between v~v+dv is given by the equation below,
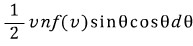
show that the mean value of cosθ for these molecules is 2/3.
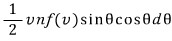
was the number, the number.
Let’s multiply each of those numbers by the cosθ for each of their respective angles (think of it as a weight), sum them all, and then divide by the total number of hits to get the average.

b) Using the result above again, show that for a gas satisfying the Maxwell distribution, the average energy of the entire molecules is
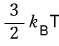
but,
the average energy of molecules hitting the surface is
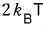
.
The average of E for the ones that hit…… hmm
Multiply each of those numbers by the KE for each of their respective motions (think of it as multiplying by a weight), sum them all,
and divide it by the total number of hits.
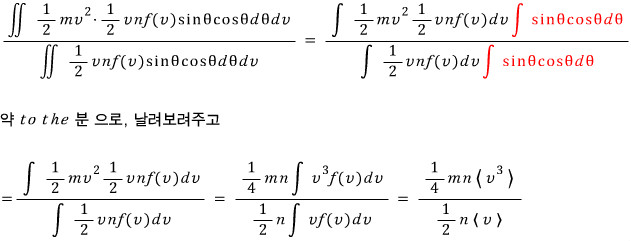

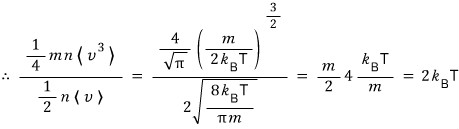
(proof of integration formula http://gdpresent.blog.me/220584291592)

My study notes on thermal/statistical mechanics #9. Proofs of integrals often used in thermal/statistical mechanics (link-only)
I won’t derive this one! Because it’s easy, so we’ll skip it, and taking the derivative of this result equation with respect to α on both sides, thermal·statistical…
blog.naver.com
( proof of

My study notes on thermal/statistical mechanics #10. Expected value of ideal gas speed, expected value of energy, ideal gas’s…
Alright, now I’m done talking about distributions, from now on we’ll look at averages, so back to looking at velocity again…
blog.naver.com
Prob 6.6
Molecules within some gas move with different velocities from one another.
A specific molecule moves with velocity v, with speed |v|, at an angle θ with respect to some fixed axis.
In some gas, it has already been shown that the number of molecules whose speed is between v and v+dv and whose angle with the specified axis is between θ and θ+dθ is
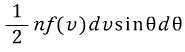
.
Here n is the number per unit volume, and is a function of f(v).
(f(v) could be the Maxwell distribution given above, but do not assume so and calculate for the general case.)
Integrate and obtain the following. (u is one rectangular coordinate component of v.)
Let me interpret the problem first.
Our
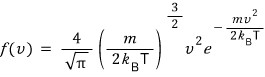
how this was derived,

something like this was how we derived it,
but here it’s saying to also consider the case when it’s not the Maxwell-Boltzmann distribution…..
f(v)
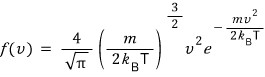
it’s saying not to rewrite it as this exponential term, but just go with f(v) as is.
And the idea for solving the problem, we have to use the idea from Prob 6.5!!!!
So let’s solve the problem gogo
a) = 0
Since u was said to be one of the rectangular coordinate components, we can just flat-out set u = vcosθ!
Because
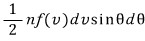
the number that this represents is
→ the number of things with the same cosθ value for the direction, so we can think of that direction as one axis of the coordinate axes!!!
Then the average of vcosθ will be obtained by multiplying the weight vcosθ to each number having v and θ, adding them all~~~ up,
and then dividing by the total number again to get the average!
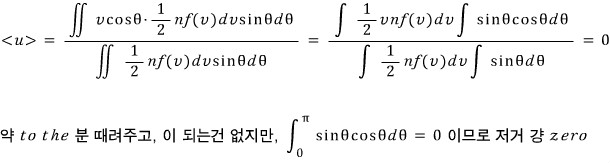
An obvious result. For a component in any one direction, it’s distributed evenly in all directions,
so it’s obvious that the average in that component direction comes out to 0!!!
b)
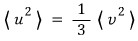
Let’s also take a look at the square of that component.
Calculating the average by the same principle,
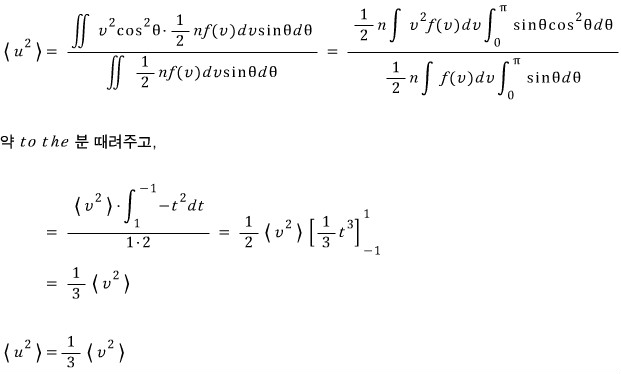

Originally written in Korean on my Naver blog (2016-01). Translated to English for gdpark.blog.