Graham's Law of Effusion
We derive Graham's Law of effusion from first principles using flux and the Maxwell-Boltzmann distribution — turns out it's way easier than it sounds!
Already Chapter 7. Molecular Effusion. We’ve already sprinted to chapter 7!
This time we learn effusion speed!
But this is super super easy.
Because we learned it in high school chemistry class!
The chem teachr probably threw a proportionality formula at you.
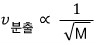
That the effusion speed is inversely proportional to the square root of mass!!!
Or you might have learned it like this
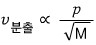
Anyway, we’re going to find out why this proportionality formula makes sense
In chemistry class you probably learned this law as “Graham’s law (of effusion)”.
Actually, it’s understandable that the chemistry teacher would just throw the above proportionality at you without deriving it.
Because, first of all, the calculation is hard for a high schooler to derive
And second, this law was originally asserted based on experiments
But, since we’re physics majors, let’s work through the cause and effect one by one!!!
No need to talk so grandly though.
The materials needed to derive this are all things we’ve used before at some point… hehe, lots of copy-paste incoming? hehe
Let’s take a look.
Somewhere, molecules are effusing…
We need to count how many of these pass through per unit time~
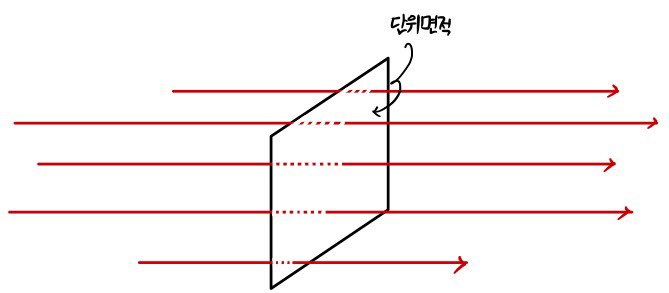
To count “the ones passing through”, I’ll introduce the concept of Flux.
It’s really nothing.
Flux is
When the thing passing through is an electric field, or a magnetic field
Just as we derived Maxwell’s Equations with this in electromagnetism
I’ll calculate the passing “molecules” as a Flux.
The number of things passing through a unit area per unit time is????????
Huh???????????????????? We just did this right before, didn’t we?
We did “the number of things hitting a unit area per unit time”
If we say that hitting means the number passing through an empty unit area,
wouldn’t it be same-same?????

We did this here. (http://gdpresent.blog.me/220584695823)
My study of thermal/statistical mechanics #11. Ideal gas equation of state, pressure
Now here we’re going to enter ‘Chapter 6. Pressure’. Rather than saying we’re learning about pressure, it’s just PV=nRT, the ideal gas equation of state…
blog.naver.com
If we count all molecules passing through for every θ and every v,
likewise, we just need to sum them all up with an integral.
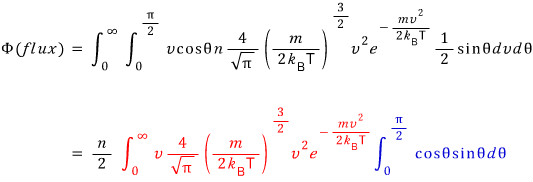


(link - http://gdpresent.blog.me/220584329950)

My study of thermal/statistical mechanics #10. Expected value of ideal gas speed, expected value of energy, ideal gas speed…
Alright, now let’s stop talking about distributions, now we’ll look at averages, so let’s look at speed again…
blog.naver.com

We didn’t compute the effusion speed.
We ended up computing the effusion rate.
We looked at Graham’s law, which says the effusion rate is inversely proportional to the square root of mass hehe
Originally written in Korean on my Naver blog (2016-01). Translated to English for gdpark.blog.