Chapter 7 Practice Problems
Working through Chapter 7 problems on particle flux for nitrogen at STP and the average kinetic energy of molecules escaping through a tiny hole.
Ex 7.3
Find the particle flux of nitrogen gas in the STP (standard Temperature & Pressure) state.
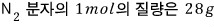
The way we computed the flux was, the number of gas molecules hitting a unit area per unit time
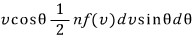
and we let it be that,
thinking of that number as being equal to the number passing through a unit area per unit time, if we add up~~ all of them for all v and all θ


Ex 7.5
What is the average kinetic energy of the gas molecules being ejected through a small hole?
We have to solve the problem with the same idea as in Chapter 6.
Multiply the gases at each v and θ by a weight KE, and add them all up,
then just divide it all by the total count, and we can call that the average energy.
The number of molecules hitting the wall per unit area, per unit time = the number of molecules going out per unit area, per unit time, since they’re the same,
the number of molecules going out per unit area, per unit time =
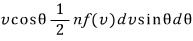
That is,

Because ejection preferentially selects molecules with higher energy,
it has a value a bit larger than the average of the total kinetic energy,
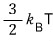
they say.
Prob 7.1
Inside a vacuum chamber designed for surface science experiments, in order to keep the surface clean, the pressure of the remaining gas is made as low as possible.
Covering the surface with just a single layer requires 10^19 atoms per square meter.
What pressure is needed so that, from the remaining gas, per hour (/h) less than one layer is covered?
You may assume that once a molecule strikes the surface, it sticks to it.

Prob 7.3
A closed vessel is partially filled with liquid mercury, and above the liquid surface there is a spherical surface of area 10^-7 square meters.
This vessel is placed in a high-vacuum region at a temperature of 273K, and after 30 days have passed it was found to have lost 2.5x10^-5kg in weight.
Estimate roughly the vapor pressure of mercury at 273K (the relative molecular mass of mercury is 200.59).


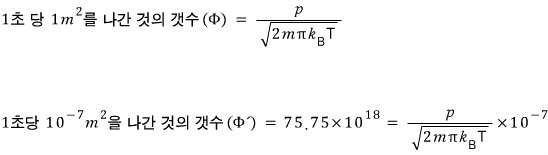
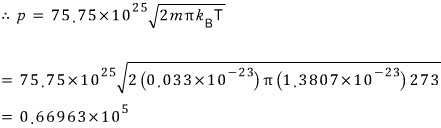
Prob 7.5
Through a small hole of area A, gas is being ejected into a vacuum.
These particles are adjusted nicely so that they pass through a very small circular hole of radius a on a screen located a distance d away from the first hole.
Show that the rate at which particles come out from the second hole is
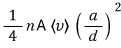
(Assume that after the gas has passed through the second hole no collisions occur, and that the relation d»a holds.)

So we have to count the number of gases that go through A and then through a like this,
meaning we need to count the gases passing through A at basically
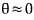
, but we don’t know that θ value…..
Let’s set it as an unknown and go-go-go (it’ll soon turn out θ = a/d, no worries no worries)

Let’s count all~~~ the kinds of this count.
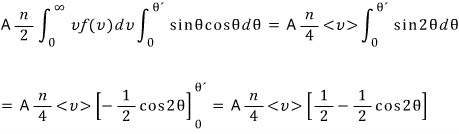

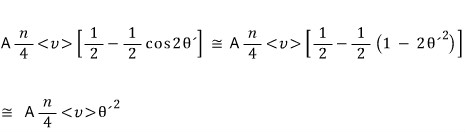

Originally written in Korean on my Naver blog (2016-01). Translated to English for gdpark.blog.