Mean Free Path Between Molecules
A casual, self-reconstructed walkthrough of mean free path derivation — expect some controversy, but the author breaks down molecular collisions and relative velocity in their own way.
This posting is probably going to stir up a lot of controversy.
I just couldn’t accept the book’s explanation, so I reconstructed it in my own way using ideas from the internet and the book.
It might not even make sense, so please do point things out.
The content of Chapter 8 is — collisions between gases!
But rather than acknowledging the effect of those collisions and then gently gently lifting the ideal gas assumption,
the goal is to find “the time between collisions” along with “the free distance between collisions.”
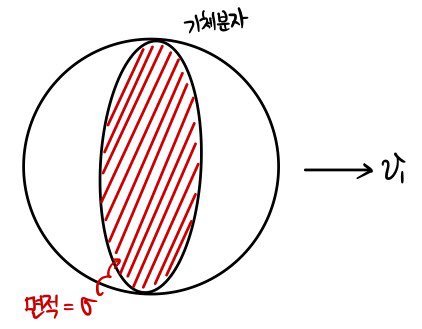
One spherical gas molecule with cross section σ
is flying along at speed v1.
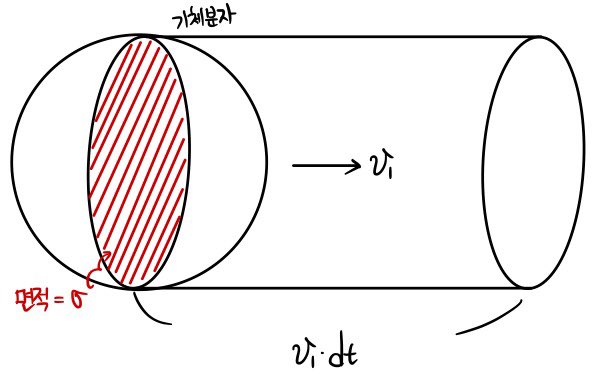
During time dt this guy..
will sweep through a volume of σv1dt.
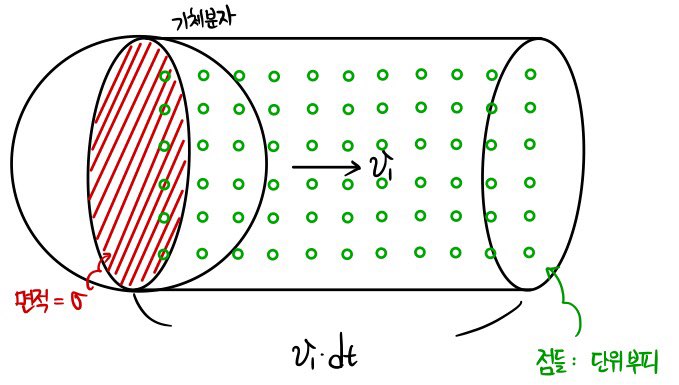
Let’s say the number of molecules in unit volume (the dots) inside that cylinder is n.
That’s the basic setting done.
The length of the cylinder that the gas sweeps through during those dt seconds is v1dt.
Let me reinterpret that length v1dt in a different way.
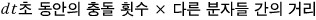
I think we could write it like this.
Now now now now, but the other molecules are also moving around as they please, so this is a bit confusing.
So we introduce the concept of relative velocity and
treat those as
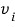
being at rest, and instead have the moving gas molecule
move with
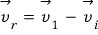
.
Then let’s count the ’number of collisions during dt seconds’ again.
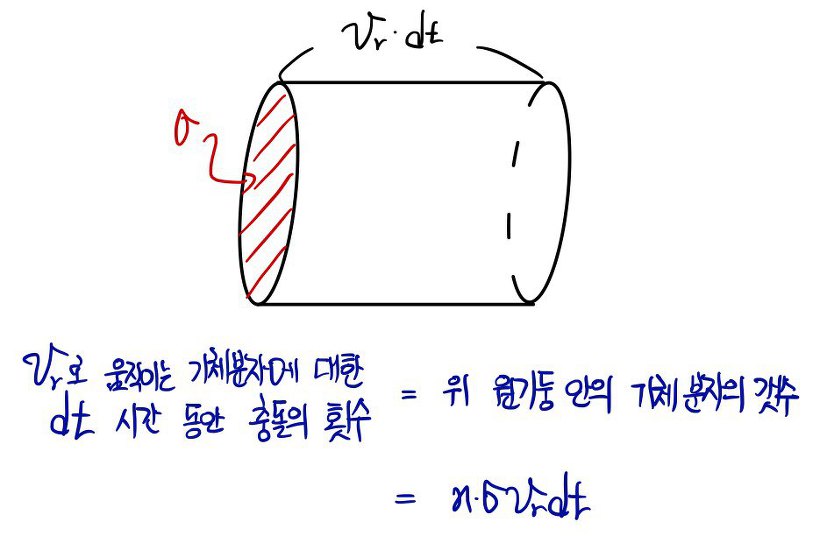
So let’s look at the equation above again.

Ah but, the distances between molecules aren’t all the same, so let me rewrite it like this.


Ah damn it, what we need is
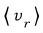
this,
in other words,
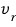
’s mean is what we need, but root-mean-square came out instead.
Earlier

Again,


Now… let me reveal my trick.
The reason we were able to get here was,
it just so happened that,
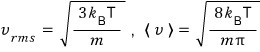
because of this.
It was because the rms and mean values just happened to be related by a constant factor.
The reason they were related by a constant factor like that is because the gases followed the Maxwell-Boltzmann distribution!!!!!!
If it had been a different distribution, that constant-factor relation might not have heldd~
So we might not have arrived at our conclusion~
That is, we juuust quietly, sneakily slipped in the assumption that they follow the Maxwell-Boltzmann distribution….hehe
Ex 8.1
At room temperature and atmospheric pressure
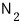
calculate the mean free path of the gas
(In the case of
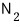
, use a molecular diameter of d=0.37nm.)

Prob 8.1
With pressure
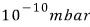
and temperature at room temperature,
what is the mean free path of nitrogen molecules inside an ultra-high vacuum chamber? What is the average collision time?
The diameter of the chamber is 0.5m.
Compared to collisions between molecules, at what ratio do molecules collide with the chamber walls?
If the pressure were suddenly increased to
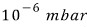
, how do these results change?



Originally written in Korean on my Naver blog (2016-01). Translated to English for gdpark.blog.