The First Law of Thermodynamics, State Functions, and Exact vs. Inexact Differentials
We dig into what heat actually IS, why state functions don't care about the path you took, and the difference between exact and inexact differentials — hehe.
From here on it’s Chapter 11. Energy.
Chapters 9 and 10 were skipped in the school curriculum.
Alright then, let’s get into Chapter 11, Energy…
Now we have to throw out another fundamental question…
Sigh… “What IS heat???????” lolllllllllllllllll
I remember this as one of the things that really mental-bombed me earlier too. Heat is ’energy'.
Because with heat you can create energy that does work,
or energy that gets lost can be lost as heat,
we have no choice but to think of heat as energy….hehehe ………..
So, to make it even clearer that heat is energy, we’re going to build an equation.
To do that, for now we need to learn the concept of a ‘function of state’.
A function of state is apparently “a physical property that takes a well-defined value for the equilibrium state of a system.”
You might recall the concept of ‘macrostate’ we dealt with earlier. Right?
You could think of it as being in the same vein. But, it’s more special than a macrostate.
That’s because we write this as a function. So then, let me write down the emphasized characteristics of a function of state.
A function of state has no dependence on time.
Why! Because we commonsensically know, and were taught, that such a macrostate will be constant once thermal equilibrium is reached.
Things like volume, pressure, temperature, internal energy, and so on.
Also! A function of state is something that has nothing to do with “how you arrived at that state.”
If we said this in coordinate-axis terms, we would say “it doesn’t depend on the path.”
That is, since the initial state and the final state — just those — are what matter,
we can express it like this.
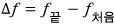
we can express it as,
and if we write this more mathematically,
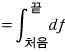
What I want to say here is,
since a function of state doesn’t depend on path, “it has df. = it has an exact differential.” That’s what this is.
This might seem like I’ve just been saying obvious stuff,
but it’s not…….. hehe
The opposite of exact differential is something called an inexact differential.
For this, ‘how you got from the initial state to the final state’ affects the value.
Let the initial state be g(initial),
the final state be g(final),
and say the function g is not a function of state.
Now, if there are several different ways to go from the initial state to the final state,
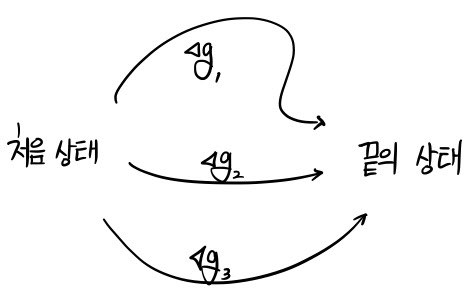
If g were a function of state,
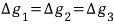
we could have said this,
but since it’s not a function of state, we can’t unconditionally say they’re the same.
Such an inexact differential
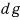
doesn’t have this, so we can’t write it like that, and
to denote an inexact differential
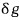
we apparently have to write it like this!!!
I’ve organized the content up to this point like this.

(Since I’m not a math major, I can’t say for sure.)
Lastly, functions of state can be classified into two kinds.

I’ll gloss over this. It’s something we dealt with before!!!
The ideal gas equation of state, which we’ve used so often and will keep using,
pV = nRT — you now have to look at it as “aha!! it’s a single equation connecting each function of state~!”
(Otherwise, things will get awkward when we do the Maxwell relations later lolllllllllllll)
Alright, now it seems it’s time to introduce the first law of thermodynamics.
The concept is simple.
“Energy is neither newly created nor destroyed” — that’s all.
That is, the first law of thermodynamics is exactly what the law of conservation of energy says.
(From here on, in all our discussions, the system has ‘gas’ in it.)
Earlier, as the answer to “Why is heat energy?” I said,
“Because with heat you can create energy that does work, or energy that gets lost can be lost as heat.”
Inside that statement, the entirety of the first law of thermodynamics is actually contained.
Let’s think about it simply.
When some object gets hotter (think of an object (system) containing gas) — as it gets hotter by that much, the gases inside make the volume get bigger, right?!
Huh!!!? The volume gets bigger?!
Yes yes yes, it did work. So like this, we can convert heat into work.
Work??
As with the work-energy theorem, having done work means “having consumed energy.”
Anyway, we can see the relationship heat ~ energy.
So now, let’s say that gas-containing object currently has some amount of energy.
The total sum of all the energies inside that system, all lumped-lumped-lumped together, is called the internal energy (internal Energy),
and we denote internal energy as U.
Now, we don’t look at the internal energy U itself,
but rather we look at the change in internal energy U. This is better for your mental health.
How can we gauge the change in internal energy U?
If we base it on the text above? The change in heat, and how much work was done —
we can say that by that much we can gauge the change in internal energy U.
That is,
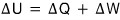
this is what I was saying heheheh.
Now let’s consider ΔW.
For a system containing gas, ‘how much work was done’ can be gauged by ΔW = -pdV.
As for why pV is ‘work’, it’s because FΔx = ΔW, so
three-dimensionally, pdV can be expressed as work.
And, since the volume decreases when work is received, we attached a (-) sign.
Hmm… are you sure???
Speaking like the above is wrong.
So, it’s only in a certain case that saying it like the above holds.
That ‘certain case’ is “no friction, no resistance, not moving too fast…. etc.”
That kind of case. (This case is the condition for a reversible change. We’ll talk about it again below.)
In that ‘certain case’ it can be written as ΔW = -pdV, but
if there’s friction, resistance, moving at super super speed, then ΔW = -pdV does not hold.
Let me explain what I just said.
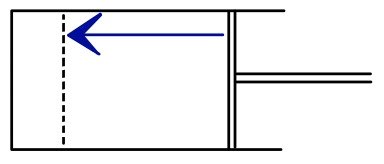
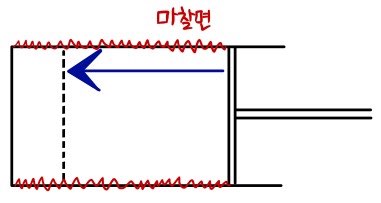
Let’s think about two cases.
Both go from V1 to V2 the same way, but say one side has friction.
For the same amount of volume change, would the same amount of energy be required?
That’s no no — in the case on the right, more energy would be needed, right?
What I wanted to say was that ΔW is not dW, but δW.
In the same vein, ΔQ also should be written as δQ, not dQ!!!!
And, I said the case where we can write dW = -pdV is in the situation of “no friction, no resistance, not too fast…..” —
such a situation is called a ‘reversible change’.
What ‘reversible’ means, I’ll cover a liiiiittle bit more in my next posting.
And as for this too, why such a situation is called a ‘reversible change’ will gradually become understandable, and
complete understanding will come around chapter 14. Entropy, so there’s no need to be impatient.
So then, the first law of thermodynamics,
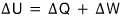
this, I’ll write it like this.
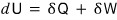
(It makes no sense for U not to be a function of state. If U weren’t a function of state, the law of conservation of energy would be meaningless.)
This too — complete understanding will come around chapter 14. Entropy, so there’s no need to be impatient.
Especially, in the case of a reversible change,
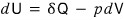
I organized up to the point of writing it like this.
Actually, in a reversible change, the change in heat Q is also unique.
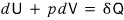
couldn’t we write it like this?
The right-hand side is unique, so it seems there’s no need to go out of our way to denote the change in Q as δQ.
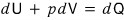
I’ll denote it like this.
Since we’ve gotten a new definition of dQ, what we had previously learned only as a concept —

— we can express in terms of functions of state!!!!
Let me organize this and move on.
For that, the first thing we’re going to do is express dU as a function of T and V.
Not U, but dU (the change dU in U) is what we’re going to express.
In math class, some function z —
and if that function z’s variables are x and y, then the change in z(x, y) is
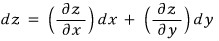
we expressed it like this, right!!!!?
This is when it’s a two-variable function, and in the case where it has more variables but we express it only in terms of x and y,
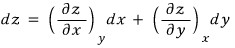
That is, to express dU in terms of the variables V and T,
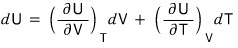
we can express it like this.
Substituting this into
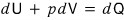
and expressing it,
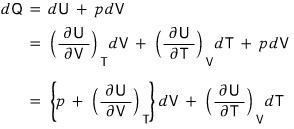
Substituting this organized dQ into

the defining expressions,
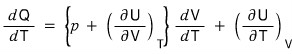
Now, once holding V constant and once holding p constant, let me express the heat capacity at constant volume and the heat capacity at constant pressure.
- Holding V constant in the above equation.. => dV = 0

This is how the book derived it, but there seems to be an inconsistency T_T
So for C_v at least, I’ll derive it from the first law of thermodynamics.
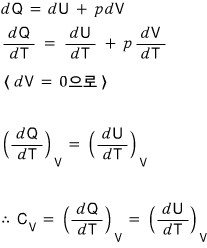
- Holding p constant in the above equation, => dp = 0
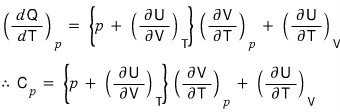
Huh, it just ended.

Let me forcibly construct the relation between them.
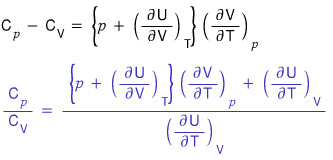
Especially, I’ll define the blue part as gamma.
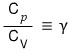
(This gets used later on~~~~)
Up to this point, everything was derived under the assumption of ‘reversible change’?!! You agree, right?
On top of this, if we throw in the assumption of an ideal gas~~~
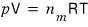
using this relationship, we can express it a bit more simply.
And and and and and, with the assumption of an ideal gas, the energy inside the system
is only the ‘only kinetic energy’ of the gas, so we can say

Once we plug in the ideal gas assumption, the internal energy U of an ideal gas depends only on T, and has nothing to do with the volume V.
That is,
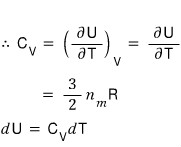
well, we can organize it like this, and

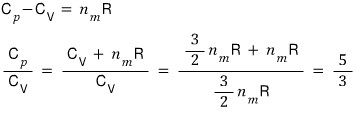
Once we assume an ideal gas,
the equation simplifies like this!!!!!!!!!
My writing is kinda super….. all over the place. Sorry T_T
P.S.
Did you know?
I’ve converted all my blog posts into PDFs
and I’m selling them as PDF materials :-)
https://blog.naver.com/gdpresent/222243102313

Blog post PDFs (ver.2.0) for sale (My Studies in Physics, My Studies in Finance)
Purchase info is below ~Hello! If there are parts of the blog posts that aren’t satisfactory, too much…
blog.naver.com
Originally written in Korean on my Naver blog (2016-01). Translated to English for gdpark.blog.