Chapter 11 Practice Problems
Working through Chapter 11 thermo problems — isothermal expansion of a monatomic ideal gas and showing those Cv/Cp identities with γ, step by step.
Prob 11. 1
1 mole of monatomic ideal gas is trapped in a cylinder by a piston, and is kept at constant temperature T0 by being in contact with a heat reservoir.
This gas expands slowly while being kept at the same temperature T0, and its volume changes from V1 to V2.
Why does the internal energy of the gas not change? Calculate the work done by the gas and the heat that flowed into the gas.



Prob 11.2
Show that for an ideal gas, the following holds.

For an ideal gas, in the previous post I summarized it like this.
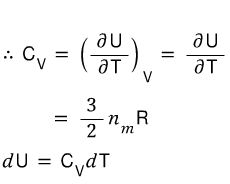


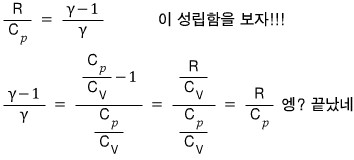
Prob 11.3
Consider the following differential.



Huh, it doesn’t depend on the path.
Prob 11.4


Originally written in Korean on my Naver blog (2016-01). Translated to English for gdpark.blog.