Isothermal Expansion and Contraction, Adiabatic Expansion and Contraction
We revisit isothermal and adiabatic processes under the reversible + ideal gas assumptions, and get clear on what 'reversible' actually means before the real fun begins!
In high school Physics II we learned about isothermal and adiabatic processes, and we’re going to verify these once again using the tools we’ve learned.
And the assumption we’re going to make is!!! we’ll assume it’s ‘reversible’.
So just like we did before, we can write δW = -pdV.
Then we can also just write δQ as dQ~
The content of this Chapter 12 is important when we deal with Chapter 14. Entropy, so they say we have to get it down properly!!!!
Alright then, first let me walk through one more time what ‘reversible’ actually means.
The word “gayeokjeok” is a translation of the English word reversible.
That is, it means reverse-able…
It means the situation in the reverse direction also makes sense!!!!
So, at a pool hall, you run at a stationary red ball with a white ball and collide with it.
And then let’s say the white ball and the red ball each start moving.
For the reverse-direction situation, by sending the white ball and the red ball along,
can’t you have them collide at the same point where they collided before, so that the red ball comes to rest and only the white ball continues on?????
Yes, it’s reversible!!!!! (Of course, due to losses in sound energy and heat energy, we might not be able to call it reversible,
but please ignore that and think of it ideally. Right now I just want to explain what reversible means hehhh hehehe)
But, for events like an egg being dropped and broken,
or the legendary sage Gang Tae-gong (Jiang Ziya) splashing water on his ex-wife — the classic “spilled water cannot be gathered” tale,
or rubbing your hands so it becomes heat energy, etc.,
the reverse-direction events — the pieces of the egg all stick back together and rise up into the sky,
or the water molecules that are on the ground merge back into one and go back into the cup,
or the heats gather together and make my hand move??
you can’t say these things are possible…. right??? Wouldn’t they absolutely be impossible??????
If that were possible, the saying “it’s water already spilled” wouldn’t exist.
Like this, in the natural world there are way way way more irreversible things.
*quasistatic
But here’s how we think about these things.
Even for an irreversible phenomenon, if you take like a million photos per second of that … event that’s happening snap-snap-snap-snap-snap-snap-snap-snap-snap-snap-SNAP,
and then print out and look at the photos, the process from the k-th photo to the k+1-th photo can be said to be a reversible phenomenon.
Since this process maintains static equilibrium, it is static in a sense,
but in fact it isn’t really static, so they call it quasistatic.
Alright, now onto the real main topic.
*I’m going to explain isothermal processes and adiabatic processes.
From here on out, since we have the ultra-special assumption of an ideal gas,
together with the ultra-ultra-ultra-ultra-special assumption of a reversible process, it’s not hard!!!
Alright, then, start — gogogogo
First up is the isothermal expansion of an ideal gas (with reversible change).
As the words ‘isothermal’ and ’expansion’ suggest,
let’s think of it as: the temperature T stays the same while at the same time the volume V gets bigger.
Thanks to the ultra-special assumption of an ideal gas, the internal energy U does not depend on the volume V, and depends only on the temperature T.
That is, dU = 0.

Through this equation we can answer the question “with temperature held constant at T, if V changes, what is ΔQ?”
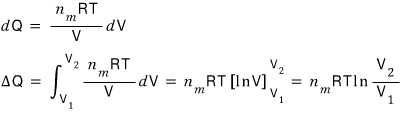
Done.
As we anticipated earlier, with all other conditions being the same, if V1 < V2,
that is, if the volume gets bigger, ΔQ > 0.
Conversely, for the compression case V1 > V2,
if the volume gets smaller, ΔQ < 0.
The fact that the volume gets bigger with temperature constant means heat energy has flowed in,
and if the volume gets smaller it means heat energy is being released~~~~?!?!
Now let’s look at ‘adiabatic’ expansion.
Thinking along the same lines, the word adiabatic means the change in heat energy δQ = 0,
and the word expansion means we’re looking at the process in which the volume gets bigger?!!!

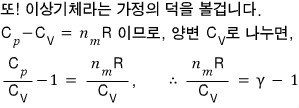





You might not understand at all what this is saying.
I also thought “what on earth am I doing right now”,
and I definitely!! recommend you work through the exercises diligently.
Only then does the understanding start creeping in little by little.
Originally written in Korean on my Naver blog (2016-01). Translated to English for gdpark.blog.