Chapter 12 Practice Problems
Chapter 12 practice problems working through adiabatic expansion, ideal gas state equations, and partial derivative relations for heat capacity — some surprisingly simple!
Prob 12. 1
In adiabatic expansion of an ideal gas,
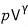
is a constant.
Show that the following equation holds.
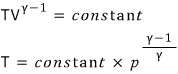

Prob 12. 2
Suppose a gas behaves according to the law given by pV = f(T).
Here f(T) is a function of temperature. From this, show the following equation.
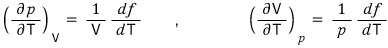

Also show that the following equation holds.
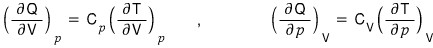

In an adiabatic change, the following equation is satisfied.
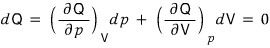
Therefore,
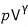
show that it is a constant.
It’s the equation where Q is thought of as a function defined by the two variables p and V.
Earlier we
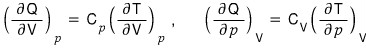
proved this,
so I’ll substitute it into the above equation.
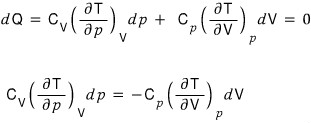
And I’ll substitute in what was previously proved,
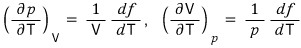

Prob 12. 3
Explain why the following can be written as such.
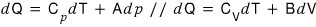
Here A and B are constants.
From these equations,
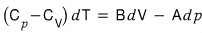
show that this holds.


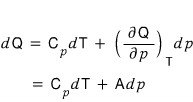


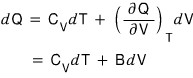

When the temperature is a constant, show that the following is satisfied.
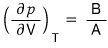
Above we had
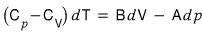
but T being constant means dT = 0
i.e.

Show that in an adiabatic change the following equation holds.
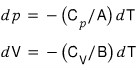
This one is easy.

The one below too
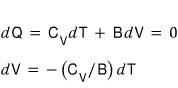
Therefore show that in an adiabatic change the following equation is satisfied.
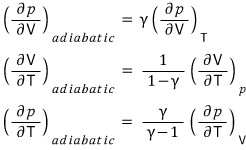
First,
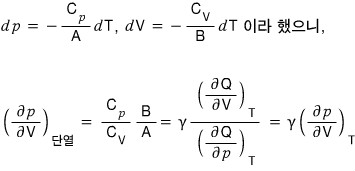
Diving into the second.

Let’s cook up the last equation too.

Prob 12. 4
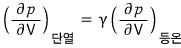
Using this,
explain the slopes of adiabatic and isothermal on the p-V graph in relation to each other.

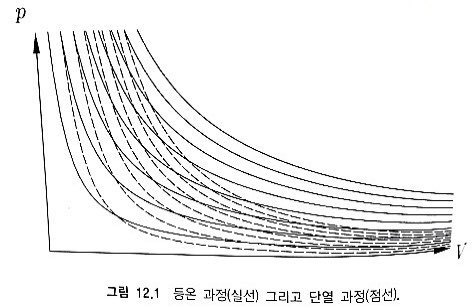
Prob 12. 5
Two thermally insulated cylinders A and B of equal volume are connected by a valve each having a piston.
Initially A has its piston completely pulled out and contains a monatomic molecular gas inside the cylinder at temperature T,
B has its piston pushed completely in, and the valve is closed.
After the operation described below is carried out, calculate the final temperature of the gas in each case.
In each operation the initial arrangement starts from the same state, and the heat capacity of the cylinder is neglected.
a) Fully open the valve and pull B’s piston all the way out so that the gas slowly enters into B.
A’s piston is fixed so that it doesn’t move.

Since it was said to be completely thermally insulated, δQ = 0
The reason why it was emphasized that it moved slowly is a reversible change
i.e., it will mean that we can express it as δW = -pdV.

Originally written in Korean on my Naver blog (2016-01). Translated to English for gdpark.blog.