The Second Law of Thermodynamics and the Clausius Inequality
We dive into the second law of thermodynamics, unpacking how Clausius and Kelvin's seemingly different statements are actually the same thing — and why the Carnot engine is the key!
What we’ll cover in this Chapter 13. is
the second law of thermodynamics, and there’s nothing really to set up beforehand, so I think we can just dive right in.
Clausius’s statement of the second law of thermodynamics:
A process whose (sole result) is the flow of heat from a colder body to a hotter body is impossible.
Kelvin’s statement of the second law of thermodynamics:
A process whose (sole result) is the complete conversion of heat into work is impossible.
The two physicists arrived at these statements after much deliberation, and these statements are going to turn out to be the same statement in about 60 seconds or so.
Clausius seems to be saying that heat flow has a direction,
and Kelvin seems to be talking about the relationship between heat and work — how on earth are these the same statement!?
To explain this, the Carnot engine is introduced.
First off, the definition of the “engine” that we all know as common sense is,
an engine is defined as “a system that operates as a cyclic process that converts heat into work,”
and that engine has to be a cyclic process that operates continuously and well~, performing a regular function.
Wait then what on earth is a Carnot engine!!!
A Carnot engine is an engine that follows the ‘Carnot Cycle,’ and such an engine is called a Carnot engine.
Wait, then are there engines that don’t follow the Carnot cycle??
When you learn thermodynamics in high school or in college general physics class, they always teach you about the Carnot engine, right?????
The Carnot engine seems so obvious,…
but in fact, the engines from before Sadi Carnot showed his genius
probably didn’t follow the Carnot cyclic process, right? They say such engines exist.
And of course they don’t cycle through the Carnot cycle either.
What they looked like is
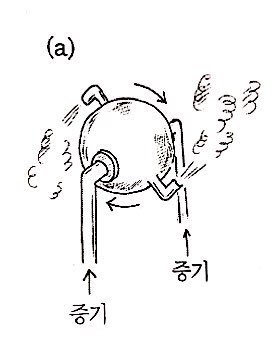
they say this is a schematic diagram of an engine made by Hero of Alexandria in the 1st century.
The principle doesn’t really need explanation I think,
but apparently you couldn’t do any big work with this, it was just at the level of a toy.
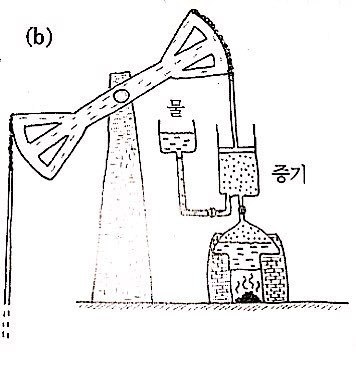
This one was designed by Thomas Newcomen (1664~1729).
This was used to pump water up from mines, apparently?!
The part labeled ‘steam’ heats up and goes up, then cools and comes back down,
then heats up and goes up again, and comes back down again.. you can roughly see the mechanism, right?!!
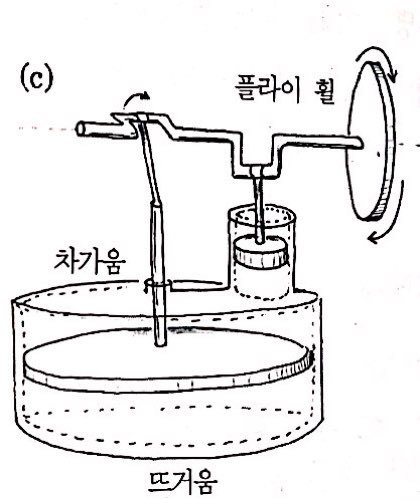
And this one was designed by a minister named Stirling.
So it’s called the Stirling engine (stirling’s engine).
Two pistons move in opposite phases and are made to move by the temperature difference.
Going back and forth between cooling and heating!
And then there’s also the engine used in cars, the ‘internal combustion engine,’
so the car engine isn’t all there is to engines in the world, it’s just one of many many engines…? lolololol
You know how a car engine works, right?!
Anyway, there are engines like these! Engines that don’t cycle through the Carnot cycle!
Anyway, let’s find out what on earth a Carnot engine really is.
We learned about the Carnot cycle in high school physics II as well,
but they never teach you what the engine actually looks like, always out of nowhere
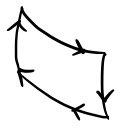
they just throw this kind of diagram at you and start talking, right???????
So I copied down just the essence of Carnot’s life…..(p.185).
Lazare Carnot (the father) was preoccupied throughout his life with the problem of mechanical motion,
and in particular had great interest in the study of waterwheel motion.
A waterwheel has falling water produce useful work.
The water falls from a high-potential-energy reservoir to a low-potential-energy reservoir,
and as it falls, the water turns the waterwheel and drives useful machinery like a flour mill.
Sadi Carnot (the son) was struck by the similarity between a waterwheel and a steam engine, in which heat flows from a high-temperature heat source into a low-temperature heat source.
Carnot’s genius was that, instead of focusing on the details of the heat engine, he decided to think of the engine in an abstract form that focused only on the pure flow of heat between two heat sources.
He idealized the operation of an engine composed only of a simple gas cycle (this is the Carnot cyclic process we know).
He realized that to be as efficient as possible, the engine must pass slowly through a series of equilibrium states — that is, it must be reversible.
At any stage, one must be able to reverse the operation and turn the cycle in the other direction.
He was then able to use this fact to prove that all reversible heat engines operating between two temperatures have the same efficiency.
Ahaaa!!!! So from the start Carnot devised that cycle,
he didn’t devise the shape or design of the engine!!!
Now, let’s take a look at the Carnot cycle.
The cycle that Carnot devised
consists of two adiabats and two isotherms.
Schematized on a V-p graph,
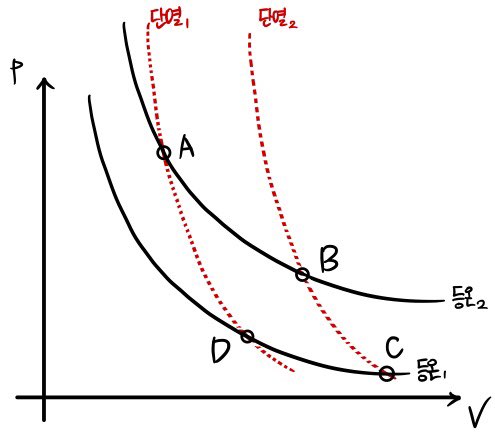
When you cycle around in the order A, B, C, D, in the (reversible) adiabatic process, it’s literally ‘adiabatic’ so there’s no heat exchange (ΔQ=0), and in the isothermal process there is heat exchange! (ΔQ≠0)
heat
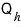
(the heat that flows in when maintained at temperature
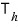
) is the heat that flows in during the isothermal expansion A→B process.
The reason it’s called isothermal ’expansion’ is, looking at the x-axis, the x-axis is V, and the A→B process is one where V gets bigger, so it’s called isothermal expansion!
And, heat
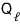
(the heat that flows out when the temperature is maintained at
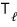
) is the heat that flows out during the isothermal compression C→D process.
Likewise, since it’s a process where V gets smaller, it’s called isothermal compression.
It’s what we did at (http://gdpresent.blog.me/220585533439)!!!?!?!?

What I studied in thermo·statistical mechanics #18. Isothermal expansion, isothermal compression, adiabatic expansion, adiabatic compression
We’ll once again verify the isothermal and adiabatic processes we learned in high school physics II using the tools we’ve learned. Doing…
blog.naver.com
Now you can understand the reason too.

The balloon’s volume changes.
To do isothermal expansion, that is, for the temperature to be maintained, some heat Q must come in for T to be continuously maintained,
and conversely, when it changes from a big balloon to a small balloon, for the temperature to stay constant,
I don’t know exactly but for T to be maintained, a specific amount of heat Q has to leave in order to keep the temperature!!!!!
Then, you can also understand why the adiabatic process on the adiabat moves between two lines of different temperatures, right?
(Is the wording hard? You’ll understand in a bit. I’m going to organize it further.)
So let me work through an example related to the Carnot cycle.
Not only does it help with understanding, but we have to solve it for the later discussion T_T T_T T_T

Let me begin.

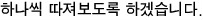
This is what we did earlier~~
(http://gdpresent.blog.me/220585533439)

What I studied in thermo·statistical mechanics #18. Isothermal expansion, isothermal compression, adiabatic expansion, adiabatic compression
We’ll once again verify the isothermal and adiabatic processes we learned in high school physics II using the tools we’ve learned. Doing…
blog.naver.com
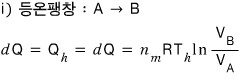





The conclusion is out. This conclusion is something we’ll keep using!!!!
In a Carnot engine, the ratio of the heat flowing in and out equals the ratio of the temperatures of the two heat reservoirs!!!!!!!!!!!
(That is, under the assumptions of a reversible process and ideal gas)
At the very beginning, we defined an engine as a system that operates as a cyclic process converting heat into work.
And, out of all the many cyclic processes, we looked at the cyclic process that Sadi Carnot devised,
and an engine that follows that cyclic process would naturally be an engine that follows the Carnot cyclic process,
and a system that converts heat into work while following that process is what’s called a Carnot engine, right?!!?
Solving example 13. 1, I caught on.
No actually I caught on back in chapter 12.
In an engine that converts incoming heat into work…
when exactly does the heat
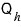
, the material that gets turned into work, come in?!?!
It comes in precisely during the isothermal expansion process.
And then converting the heat into work is done in the adiabatic expansion process, right?!?!?!
With pV^γ.
After doing work like this, in order to do work through the isothermal expansion process in the next cycle,
we have to receive heat, the material, again. To do that, the isothermal compression process is indispensable!!!!!!!
Wouldn’t we do isothermal compression before doing work again or whatever????
That is, a certain amount of heat
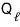
must necessarily go out.
Ah… so out of the received
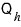
, we leave aside the heat
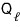
that has to go out for the next process,
and the remaining
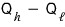
amount of heat is what gets converted into work — that’s the engine that follows the Carnot cyclic process, so that’s the Carnot engine!!!
Now, about the engine’s efficiency.
Efficiency means — how much of the input is converted into work relative to the input~~~~
Can’t we call that efficiency???
That is, the efficiency of the engine is
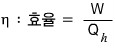
we can express it this way
The efficiency of the Carnot engine that follows the Carnot cyclic process is
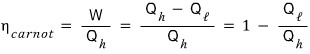
And, borrowing the result of example 13. 1,
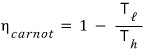
we can also express it this way!!!!
Now, let me spill the secret of the Carnot engine.
The secret of the Carnot engine!!!!
Carnot’s theorem:
Among engines operating between two given temperatures, there is no.ne more efficient than the Carnot engine.
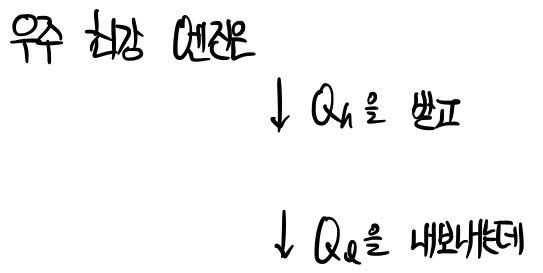
We were learning about the world’s strongest — no, the universe’s strongest!!!
a super ideal engine that doesn’t exist in our world.
That is, efficiency higher than
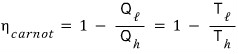
is of course nonexistent, and even this doesn’t exist.
The engines in the world are all~~~~ said to have lower efficiency than that.
And by quite a lot, it’s said… what’s the reason?
If we think about it simply, first of all, our assumptions were wrong, right.
The fact that in reality it’s not an ideal gas, and that the series of processes aren’t reversible changes,
things like that, right???? lololololol is it too obvious?
Anyway let me do the proof.
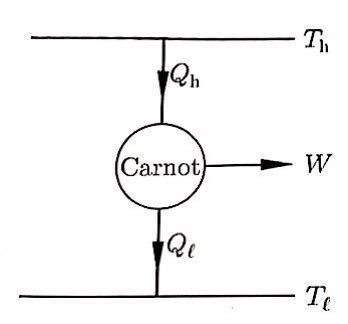
The Carnot engine works like this.
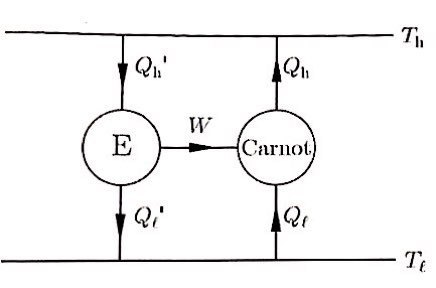
But ※assumption alert※ let’s suppose there’s an engine more efficient than the Carnot engine.
And let’s connect this more-efficient-than-Carnot engine with the Carnot engine like this.
The reason such a connection is possible is because the Carnot engine is reversible!
Since we said that engine E has higher efficiency than the Carnot engine,

Beep beep beep beep!!!!Beep bip bip bip bip bip bip!!!!!!
This violates the second law of thermodynamics.
How can it flow from a lower temperature place to a higher one….T.T
Yes. It has become something ridiculous.
That is, the assumption was wrong.
There is no engine more efficient than the Carnot engine!!!!
(How could you beat an engine made under the super-ultra-premium assumption of being reversible T_T T_T T_T)
Since the engines in the world aren’t (ridiculously) reversible engines, they have lower efficiency than the Carnot engine
And if all the engines in the world were reversible engines, the efficiency would all be the same as the Carnot engine!!!
This way Kelvin’s statement of the second law of thermodynamics also makes sense.
That it’s impossible for every last bit of heat to turn into work….
There has to be some bit of
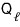
that needs to drop out!!!!! yoho
Now we’ll prove the Clausius inequality
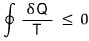
.
First, there’s a Carnot engine cycling through the Carnot cycle.
Then let’s look at exactly 1 period of the Carnot cycle that the Carnot engine cycles through,
that is, just one lap.
During one lap, this one has heat Q flowing in during isothermal expansion, and heat Q flowing out during isothermal compression.
Let me divide the inflowing/outflowing heat Q by the respective temperature at each time and then add them all up.

Okay, I get that that sum is 0, but what about the part where being greater than 0 is allowed!!!???
I don’t like the textbook’s proof of the Clausius inequality, so I’ll use a different method.
However, the conclusion of the Clausius inequality
is actually something you may have already been subtly intuiting while studying Carnot’s theorem. (That thing everyone’s intuiting right now! Yes, that’s the one.)
Anyway let me prove the Clausius inequality.
We can do it using Carnot’s theorem.
Carnot’s theorem: the efficiency of the Carnot engine is the king-king-best.

Ugh…..what is this…. just sort of obvious….
I also thought of it this way.
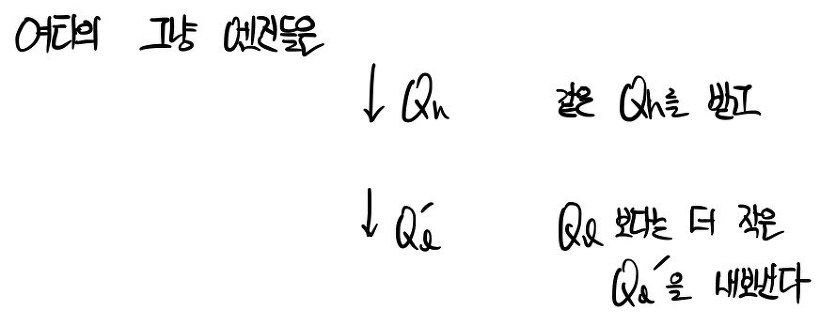
Why?!?!!?!? Because it’s irreversible,
for example, including things like friction heat.. we lose that too….. something like that????lolololol
P.S.
Did you know?
I’ve converted all blog posts to pdf
and am selling the pdf material :-)
https://blog.naver.com/gdpresent/222243102313

Blog post pdf (ver.2.0) for sale (What I studied in physics, finance)
Purchase info is below ~Hello! If there’s something unsatisfying in the blog posts, or if it’s too much…
blog.naver.com
Originally written in Korean on my Naver blog (2016-01). Translated to English for gdpark.blog.