Chapter 13 Practice Problems
Working through Chapter 13 problems on refrigerator efficiency, Carnot engines, and why perpetual motion machines just don't exist in this universe.
Ex 13. 4 Efficiency of a refrigerator?!
A typical engine or system takes in heat from a hot reservoir, scarfs it down,
and it’s standard practice to dump the rest into a (relatively) cold reservoir.
But a refrigerator is the opposite
It takes heat from a cold reservoir. (artificially….)
Here’s a refrigerator like so.
The refrigerator “does work.”
What kind of work does it do? It does the work of pulling out the internal heat.
By doing W amount of work,
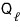
it extracts this much heat, and spits out the even bigger
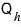
amount of heat, let’s say
If we sketch that out roughly
Alright, now let’s think about the efficiency.
The efficiency here is

this kind of ratio should be what we call the efficiency, right?!
That is!!

Prob 13. 2
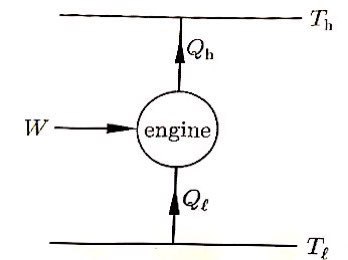
What is the maximum possible efficiency of an engine operating between two reservoirs?
One reservoir is at 100°C, the other reservoir is at 0°C.
The strongest heat engine in the universe is the Carnot engine, and the thermal efficiency of the Carnot engine is
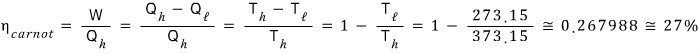
Prob 13. 3
i) Perpetual motion machine of the first kind:
It violates the first law of thermodynamics, the law of conservation of energy.
ii) Perpetual motion machine of the second kind:
The Carnot engine, a heat engine that follows the Carnot cycle, has the strongest thermal efficiency in the universe.
Since even the strongest engine in the universe has less than 100% efficiency, a perpetual motion machine of the second kind,
that is, a heat engine with 100% efficiency, does not exist, at least in the universe.
Prob 13. 4
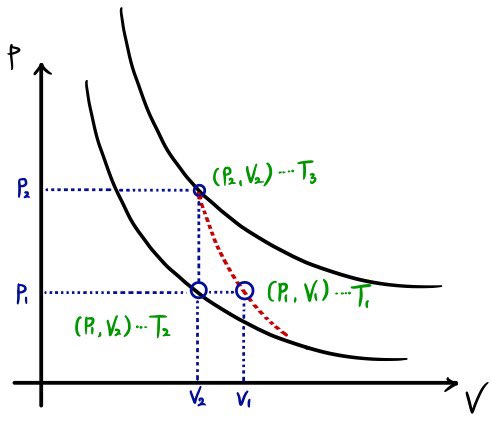
A possible ideal gas cycle operates as follows.
i) From the initial state (p1, V1), the gas is cooled at constant pressure to (p1, V2).
ii) The gas is heated at constant volume to (p2, V2).
iii) The gas expands adiabatically and returns to (p1, V1).
Assuming the heat capacity is constant (with respect to temperature), show that the efficiency is as follows.
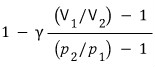
First, since this is a problem of finding efficiency,

we need to head in the direction of getting this.
That means we need to calculate the heat obtained and the heat released!!!


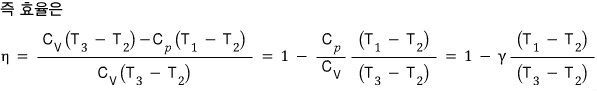

If we plug this in!!!!!!!!!
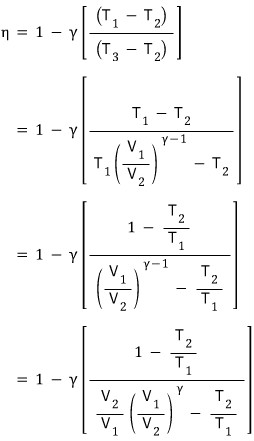
Here let me list out the ideal gas equation of state

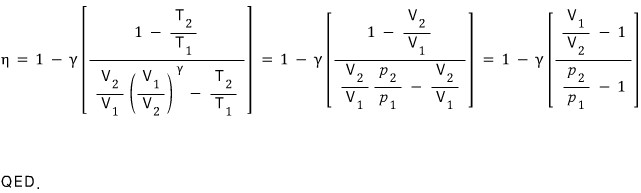
Prob 13. 5
Show that the efficiency of the standard Otto cycle is
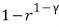
(The Otto cycle is a 4-stroke cycle used in internal combustion engines for cars, trucks, etc.) r is
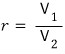
the compression ratio.
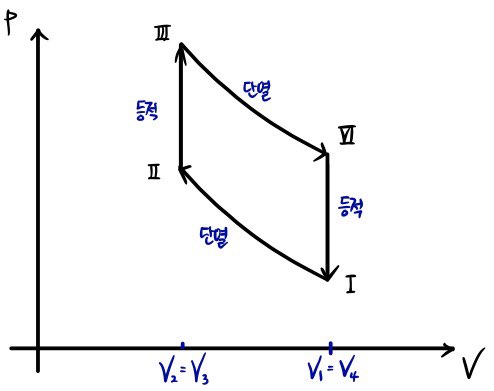
This problem is also about calculating efficiency,
Ⅰ→ Ⅱ & Ⅲ → Ⅳ are adiabatic processes so there’s no heat exchange,
Ⅱ → Ⅲ
Ⅳ → Ⅰ
in the isochoric process parts there will be heat exchange.
Naver’s equation editor doesn’t support Roman numerals so…..T_T
I’ll go with Arabic numerals T_T





Alright now let’s plug them in and unify things a bit.
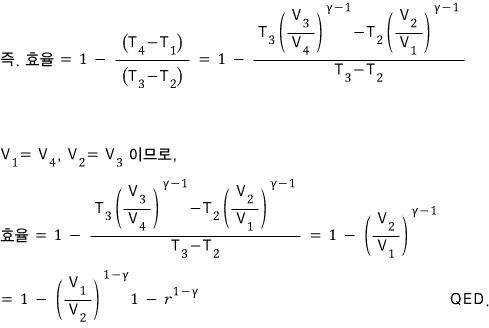
Prob 13. 6
An ideal air conditioner absorbs heat Q2 from the house at temperature T2 and releases heat Q1 to the outside at temperature T1, operating while consuming electrical energy E.
The heat loss leaking from the house follows Newton’s law
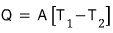
Here, A is a constant.
When steady state is reached, express T2 in terms of T1, E, A for continuous operation…
The air conditioner is controlled by a thermostat.
The air conditioner is designed to operate at 30% of its maximum electrical input, with the thermostat set to 20°C and the outside temperature set to 30°C.
What is the highest outside temperature at which the house can remain at 20°C.
This problem is kinda the same principle as the refrigerator we did earlier hehehehe
We can think of it as a refrigerator that uses energy E to extract heat Q2 at the house’s temperature T2 and throws Q1 to the outside at temperature T1.
Ah… since this is an air conditioner, I drew another picture.
Whisen let’s go
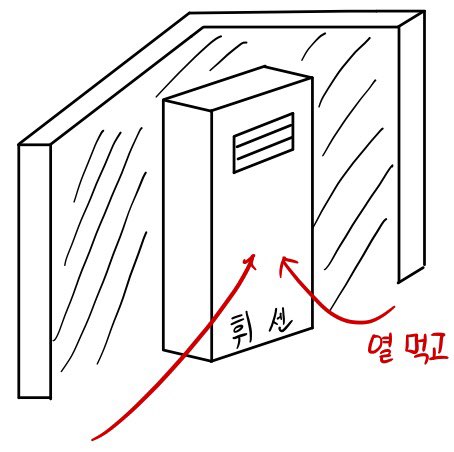
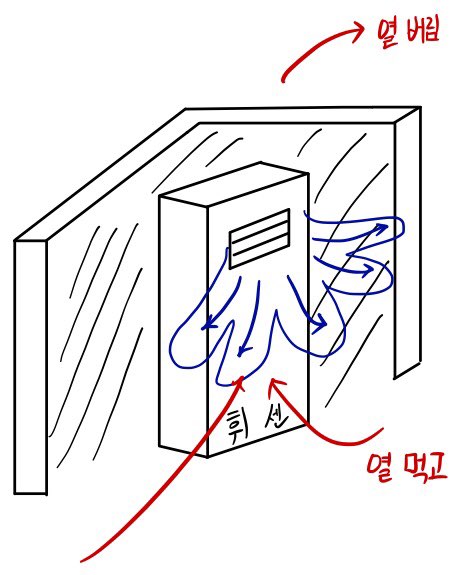
The air conditioner runs on energy E,
that is, it uses energy E to extract Q2 and throw it outside,
since we’re told it’s an ideal air conditioner (engine),
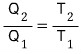
it should satisfy this!!!
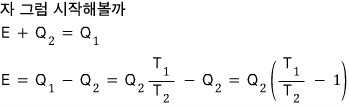
Here we need to consider the loss.
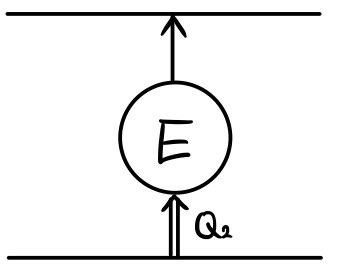
In the middle of pulling the heat up
Yikes!!! heat loss occurs.
So in the end, after extracting Q2,
it ends up spitting that much back out.
That is,
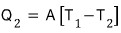
balance!!? Equilibrium will be achieved
Ah ah thermal equilibrium will be achieved!!!!! That is!
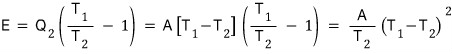
If we first organize this equation,…
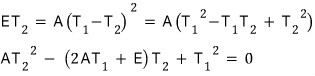
We can find T2 using the quadratic formula, and express it in terms of T1, E, A!!!
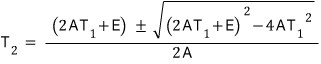
Since the air conditioner is designed at 0.3 input for 20°C(T1), 30°C(T1),
we can obtain the relation between E and A with this!!!

For the maximum outside temperature at which the house can be maintained at 20°C, we just need to find T1 when the air conditioner’s efficiency is 100%.
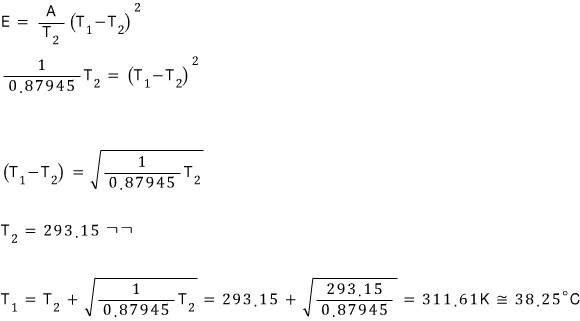
Prob 13. 7
Two identical objects with constant heat capacity Cp serve as the two heat reservoirs of a heat engine, with temperatures T1, T2.
If the objects are under constant pressure, show that the obtainable work is as follows.
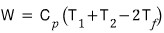
If an efficient engine is used,
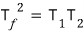
.
Under constant pressure, the heat obtained and the heat lost are

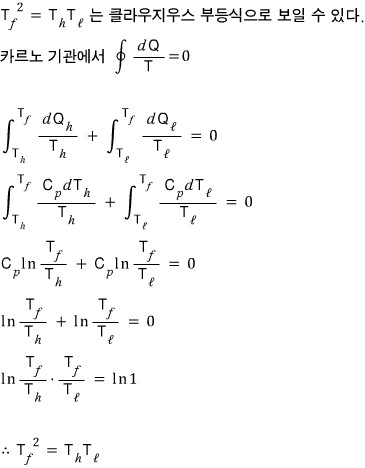
Originally written in Korean on my Naver blog (2016-01). Translated to English for gdpark.blog.