Entropy: Thermodynamic and Statistical Mechanical Perspectives
Nobody really knows what entropy is (not even the pros lol), but let's stumble through Clausius' definition and the second law of thermodynamics together anyway.
So we’re finally getting to learn about this thing called entropy that I’ve only heard about in passing.
Let me give you a heads-up in advance. I don’t really get it lol lol lol lol lol lol lol lol lol lol lol lol lol lol lol lol lol
I still have no idea what entropy actually is lol lol lol lol lol lol lol
But lol lol lol lol lol lol the thing is lol lol lol lol lol lol nobody knows it lol lol lol lol lol lol lol lol lol lol
There’s an anecdote about a world-renowned master of statistical physics who was giving a lecture.
“Are there any physicists here who know what entropy is?”
(Silence…)
“Right??? It’s not just me who doesn’t know, right?”
lol lol lol lol lol lol lol lol lol lol lol lol lol lol lol lol lol lol lol lol lol lol lol lol lol lol lol lol lol lol lol lol lol lol lol lol
Even a physicist famous in statistical physics doesn’t really know, so let’s not be too sad about it lol lol lol lol lol lol
Alright, let’s go in then.
So we already proved Clausius’ Inequality earlier.
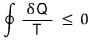
We proved it like this,
and if it goes through a reversible process,
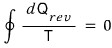
we got as far as saying we can write it like this.
For Q(reversible) I didn’t bother marking the inexact differential.
Since reversible processes have an exact differential, we can write dQ,
and I just put rev as a subscript. (Not that it matters much…)
Alright, now we’re getting into the definition.
Apparently Mr. Rudolf Clausius made the definition.
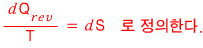
Doesn’t it feel kinda iffy? Right?!?!
Yes… this is the definition of entropy.
If I were to emphasize it more, it’s “the definition of thermodynamic entropy.”
(The reason I’m bothering to emphasize it is so you don’t get flustered when the definition of statistical-mechanical entropy comes up later? ^^)
(Rather than the definition of entropy, it’s more like a definition of the change in entropy!?!)
Also, there’s one ‘important concept’ I want to point out here.
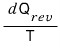
doesn’t depend on the path, right?
dS doesn’t depend on the path. That is, we can call S a state function.
Alright, so now using the definition of entropy,
let’s try to state the second law of thermodynamics that we know from common sense.
(It usually gets introduced as the law of increasing entropy, doesn’t it?)
If you go around once via a reversible process,
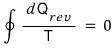
it’s clearly zero.
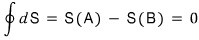
Ah but, let’s try going via an irreversible path on the way there, and a reversible path on the way back.
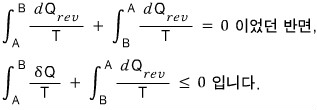
Why? It can be explained by Clausius’ Inequality…
If you follow even a tee~ny bit of irreversibility,
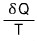
we already proved earlier that the closed-loop integral of this is less than 0, so I don’t think we need a long explanation.
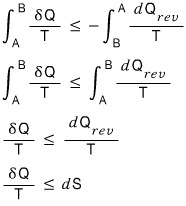
Don’t get confused here. It’s not that dS depended on the path,
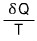
it’s this that depends on the path.
dS is
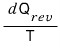
so it has some kind of absolute value,
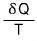
is saying that this is smaller than that absolute dS.
This was meant to once again emphasize that the entropy S is a state function.
Now let me add one assumption.
※Assumption alert※
Let’s assume “a thermally isolated system.”
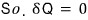
we can say this, can’t we?
That is,
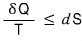
for a thermally isolated system
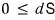
we can write it like this.
cf. Among thermally isolated systems, what’s the biggest one?
Well if we say the universe, that’d be the biggest, right? (No, this is actually an assumption. The assumption that heat doesn’t go out of the universe, doesn’t come in, is built in…T_T T_T)
This is the second law of thermodynamics that we know from common sense, that we learn in high school physics 2 or in General Physics in our first year of university……
Anyway, this is another expression of the second law of thermodynamics.
Wait, why can we think this is the same as the Clausius or Kelvin expression of the second law of thermodynamics that we covered earlier?!
If you think about it carefully, you can say that Clausius and Kelvin spoke about the direction of heat as a law, right?!?!?!?!
That is, dS ≥ 0 (in a thermally isolated system)
is also defining the flow of heat as a law.
It’s a bit of a stretch to say they’re saying the same thing, but in terms of defining the direction of heat, they are the same.
(In the universe (in a thermally isolated system), in any change the change in entropy is 0 or (+) increases…. the law that it becomes maximum….)
Now with this
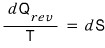
definition,
let’s think about the first law of thermodynamics once again.
If we write down the first law of thermodynamics, which states the conservation of energy in thermodynamics,
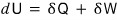
right!!!!
Now an assumption comes in! If we assume it’s a reversible process,
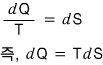
we can write this,
and also, if we assume reversible change,
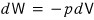
we can write this.
That is,
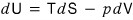
we can say this.
Now now now now now now now
This might really get confusing, but I just said that the above equation is correct under the assumption of a reversible process.
But actually, the equation above, dU = TdS - pdV,
is correct even without the assumption of a reversible process.
Let me put it this way.
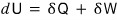
When we wrote this, the change in Q is an inexact differential and isn’t unique,
and the change in W is also an inexact differential and isn’t unique,
so the change in U might not be unique either!!!!!!
Then shouldn’t we write the change in U as δU instead of dU!!!!!
You might think this, but as I mentioned once before, the change in U absolutely has to be unique.
For our universe, if the change in U weren’t unique,
it would mean that energy could be created from nothing, or that existing energy could suddenly disappear,
and that could violate the law of conservation of energy, which is something like the foundation of our universe, so it wouldn’t make sense if U weren’t a state function.
Yes, so U is also a state function,
and what the first law of thermodynamics tells us is that it can be expressed by the state functions T, S, p, V.
That is, U is a state function, and at any moment U has to be unique.
(Now what I really want to say)

is not only correct in reversible processes, it’s also an equation that holds for irreversible processes.
Of course, the value of TdS in a reversible process and the value of TdS in an irreversible process will be different.
It will have a larger value in an irreversible process.
The blue pdV will also have a smaller value in an irreversible process than in a reversible process.
So dU will always have the same value!
Then now, whether reversible or irreversible process,
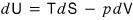
I’m throwing this equation out there.
Take a good look at this equation.
It’s expressing the change of the function U in terms of the change of S and the change of V.
That is, the variables of the function U are expressed by S and V.
That is, we’re thinking of the function U as a two-variable function U(S,V).
In math class we learned that the change dU for a two-variable function U can be expressed like this.
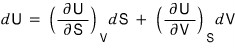
Makes sense, right?
Then let’s compare it with this.
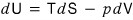
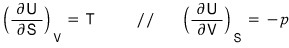
We found out that the state functions T and p can be expressed in terms of other state functions!
Using the reciprocal Theorem on the above two relations,
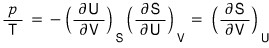
we can organize it up to here.
(I’ll prove the reciprocal theorem in the following post.)
Lastly, let me organize the statistical mechanical definition of entropy.
In Chapter 4, by using the zeroth law of thermodynamics that temperatures become equal in thermal equilibrium,
“In two connected systems, the most likely way to divide the energy between the two systems”
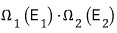
will reach equilibrium at the distribution that maximizes this.
We then differentiated this with respect to energy
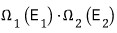
to find the maximum value.
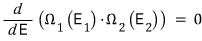
When we did this,
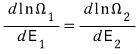
this kind of relation came out, and what this relation means is “they are at the same temperature,” and we used this to define temperature.
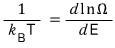
we learned that it’s defined like this.
(For details, link. http://gdpresent.blog.me/220583972835)
What I studied in Thermal/Statistical Mechanics #4. Temperature
Statistical definition of temperature. Finally now the book is going to look at what ’temperature’ is!!! Thinking about the definition of temperature, and finally my…
blog.naver.com
N-ow. Let me add to this here.
Earlier, with the first law of thermodynamics, we
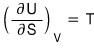
learned this.
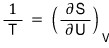
we can write it like this.
Let me put the definition of temperature that we skimmed over in Chapter 4 into the same form.
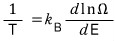
Therefore,

Like this, we were also able to confirm the statistical mechanical definition of entropy.
Ω was our ‘microstate,’ right?
Thinking about it that way, I kind of get why people call S, entropy, the degree of disorder.
(For your reference, from here on I’ll talk about thermodynamic entropy as ‘useless energy,’ and I’ll talk about statistical mechanical entropy as “degree of disorder.”)
And the equation above is exactly what Boltzmann defined and is apparently the equation engraved on Boltzmann’s tombstone ^^ hehe
(What we wrote as Ω, they wrote as W~)

Originally written in Korean on my Naver blog (2016-01). Translated to English for gdpark.blog.