Free Expansion (Joule Expansion) and Entropy of Mixing
A casual walkthrough of Joule (free) expansion and entropy of mixing, showing why both processes increase entropy even when internal energy stays zero.
- Joule Expansion
What is Joule expansion
You can just think of it as free expansion.
Let’s take a look at free expansion!!!
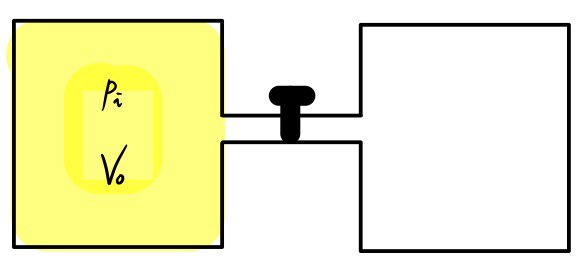
Let’s think about gases spreading out into double the volume
And let’s say the two containers are thermally insulated
So we pull out the pin and the gases go pwoosh~~~~ and spread out
.
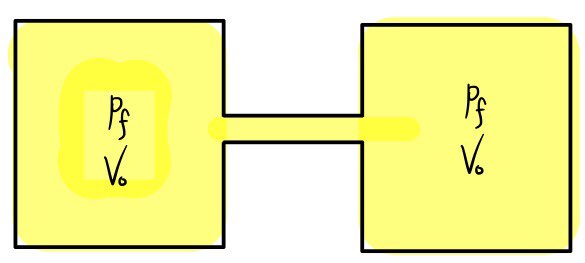
It really doesn’t seem like the gases would be gaining or losing energy
That is,
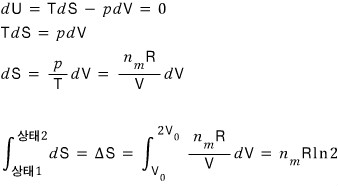
The change in entropy is not equal to zero….
That is, free expansion is an irreversible process.
What the heck….T_T
The fact that entropy increases while dU = 0 means the pressure decreases!!!!!
We’ll work through some practice problems on Joule expansion in the next post.
And going further on, related stuff to think about will come up.T_T_T_T The wording’s hard, right T_T_T ah….T
- Mixing Entropy
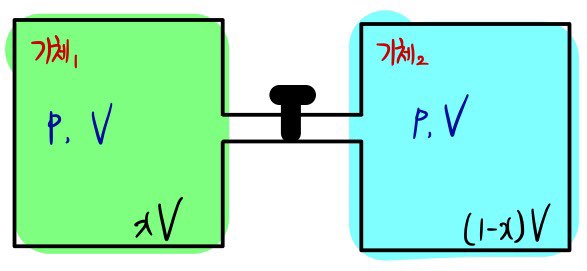
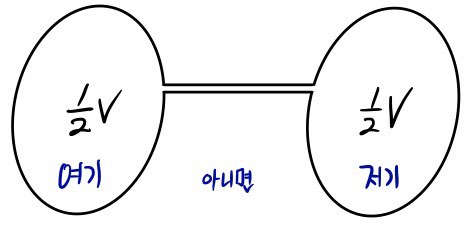
Let’s say two containers are connected, one with volume xV and the other with (1-x)V.
And let’s say the two containers were in the same p and T state
That p and T are the same is also taught in chemistry class
It means there’s gas in proportion to the volume ratio.
(If you write down the ideal gas equation of state, you can see that n is in a proportional relationship with V.)
(Actually, this is also taught as Avogadro’s law)
That is, since the volume ratio is x : 1-x
The mole ratio of the gases will also be x : 1-x
Let’s say there’s
Nx mol
and N(1-x) mol respectively.
OK so now, let’s pull out the pin!!!
There won’t be any great revolutionary who claims the gases won’t mix, right?
Because gas 1, which had been in xV, also wants to be in the total V,
and gas 2, which had been in (1-x)V, also wants to be in the total volume V…
Let’s think of each gas as undergoing reversible isothermal expansion.
That is, dU = 0
Therefore TdS = pdV
Let’s look at the entropy change with this equation.

Oho, so how the volume is initially split between the two affects the entropy change.
So I used Matlab to plot y = xlnx + (1-x)ln(1-x) once.
The conclusion is that it’s largest when x = 1/2.
Let’s interpret it.
When x=0, x=1, it means the volume of the other side is “none,”
so it makes sense that the total entropy change is zero.
And if we plug in x = 1/2
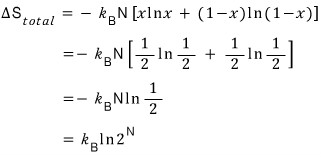
Oh!!! This also makes sense.
Because, by the statistical definition of entropy,
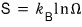
Ω is the number of microstates, and the fact that at x = 1/2 the number of microstates increased by 2^N means
the number of ways to put a gas in is

it must be because it increases in this janky way.
(Earlier in Joule expansion, when we did free expansion to double the volume, this kind of number came out, right? I think we can think about it in the same context.)
OK so now, it’s time for your brain to hurt.
Let’s rack our brains.
“What if the gases that had been in the two containers were the same gas?!?!?!??!?!!!!!”
x = 1/2. The p, T conditions were all the same.
After removing the pin, the total entropy change would, as above,
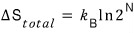
come out like this…
But, common-sense-wise, if they were the same gas the number of microstates wouldn’t be affected at all, and
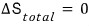
it should be like this?!?!?!?!!
What went wrong….
The answer is
what was wrong was that we developed the equations treating S1, S2 — that is, gas 1 and gas 2 — as distinctly different.
The assumption that was implicitly laid in our equations was the assumption that “the gases are distinguishable.”
So if they’re not distinguishable, it doesn’t work, they say.
Sigh… how can gases be distinguishable.
This concept will be dealt with more profoundly in chapter 29, supposedly.
For now, just for fun, let’s listen to the story of “Maxwell’s demon” (a thought experiment).
Originally written in Korean on my Naver blog (2016-01). Translated to English for gdpark.blog.