Entropy Practice Problems
Working through entropy practice problems—cooling tea and bathtub heat sources—to confirm the second law of thermodynamics (dS ≥ 0) holds up in real scenarios!
Prob 14. 1
A mug teacup was cooled from 90℃ to 18℃.
If there is 0.2kg of tea in the cup and the heat capacity at constant pressure is 4200 J/K·kg, show that the entropy of the tea decreases by 185.7 J/K.
Explain the sign of this result.
I’ll interpret this problem as the contact between a small system and a large heat reservoir.
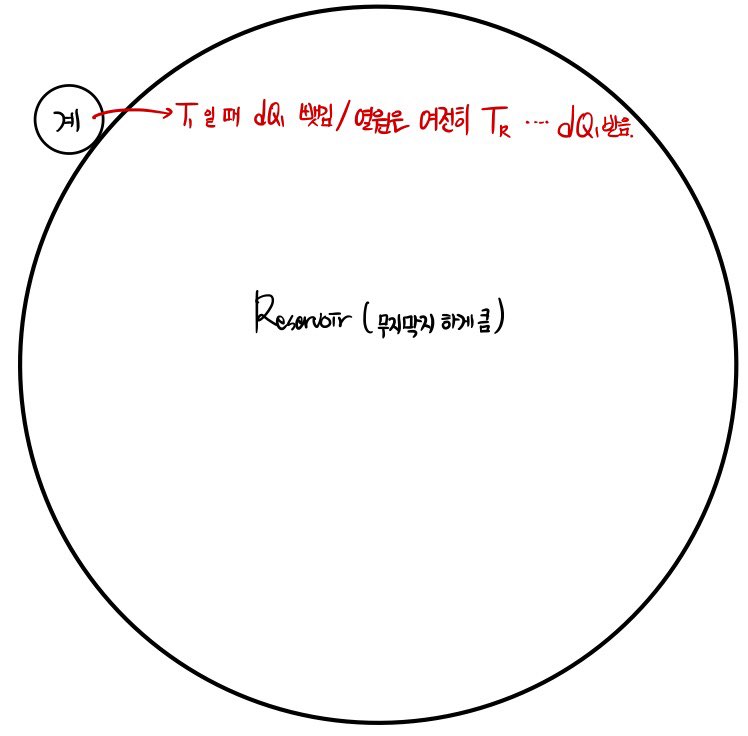
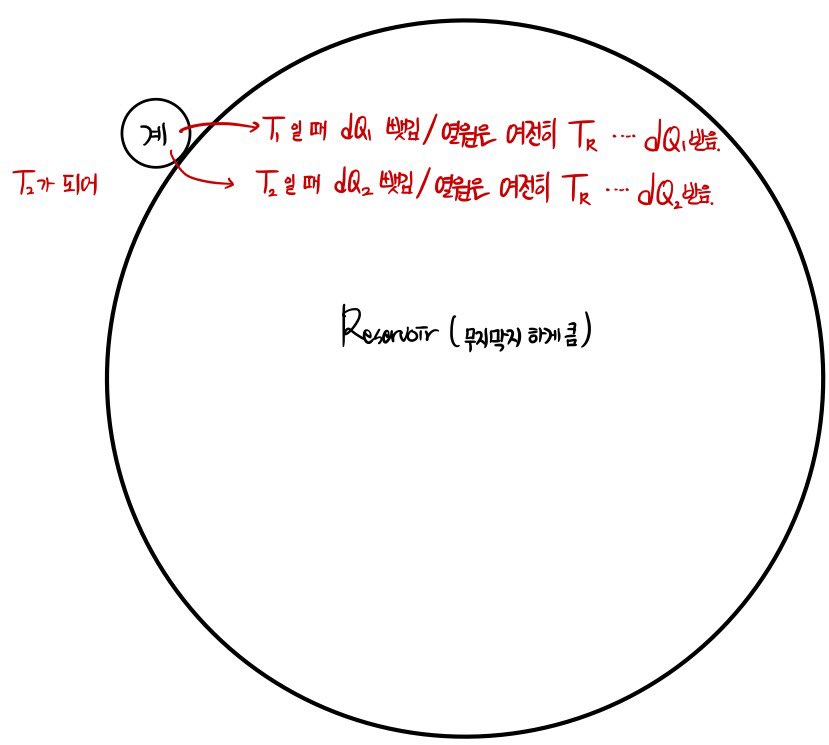
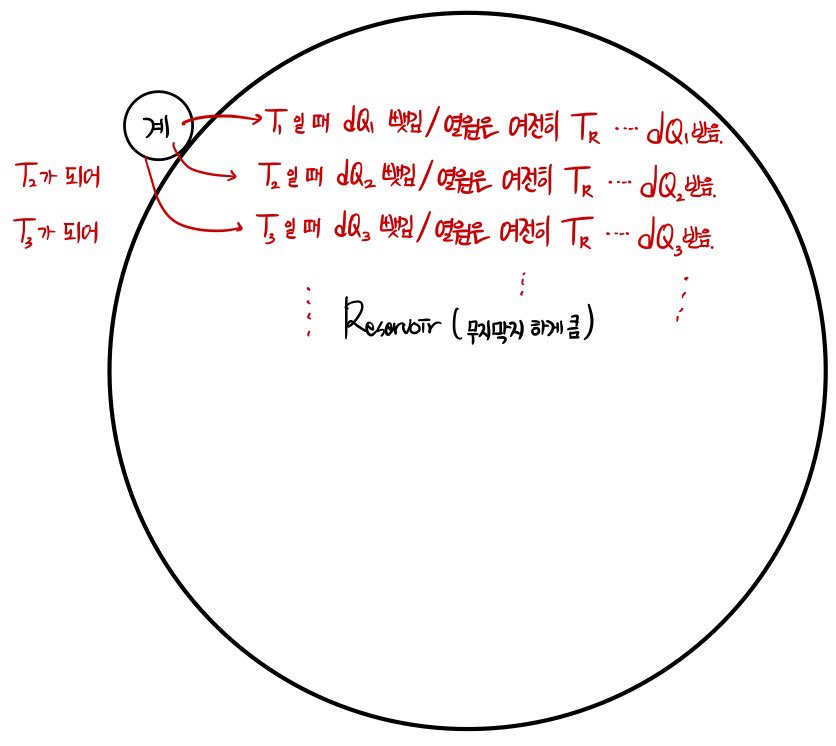
Since calculating the entropy of the system and calculating the entropy of the Surrounding are slightly different, I tried to express them with pictures.
So if I do the system entropy calculation first, the system has its temperature T changing from moment to moment.
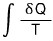
In here, T keeeps changing.
Now, the situation in the above problem is one where the pressure is constant, so the change in heat δQ can be calculated as C(p)dT.
That is,
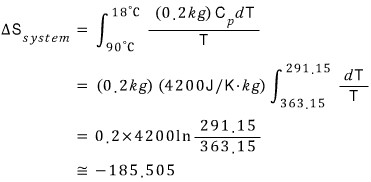
If I also calculate the entropy of the Reservior,
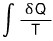
When I try to do this, the temperature T of the Reservior is constant at 18℃. So it can come out of the integral~

That is, the change in total entropy is 207.73 - 185.505 = 22.225
Yesss!!! It doesn’t violate the second law of thermodynamics dS ≥ 0.
And if I think about why the entropy of the system decreases,
The thing about entropy increasing,,…. that was when we assumed it was a thermally isolated system.
So for systems that aren’t thermally isolated, it doesn’t always have to be positive…!!!
okok
Prob 14. 4
a) When a tank of water initially at 20℃ comes into contact with a heat source at 80℃, find the change in entropy
b) Find the change in entropy of the heat source when process a) occurs.
The bathroom and its contents have a heat capacity of 10^4 J/K
Should I understand it as a bathtub?
Anyway, the entropy change of the bathtub is
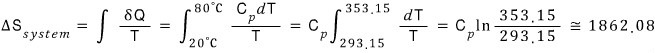
The bathroom temperature will be 80℃ the whoooole time, so
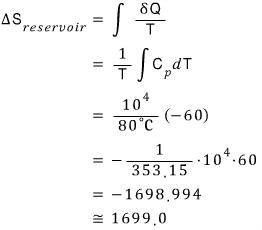
That is, the change in total entropy is 1862.08 - 1699.0 = 163
<I did solve part c, but I’m not confident….T_Tlololol>
Prob 14. 5
A lead block with a heat capacity of 1 kilo J/K is cooled from 200K to 100K
in two ways.
a) Drop it into a large fluid at 100K.
b) Cool it down to 150K in a fluid, then cool it down to 100K in another fluid at 100K.
For both cases, calculate the change of the system composed of the block and the solution tank, when cooled from 200K to 100K.
Prove that in the limit of infinitely many intermediate solution tanks, the change in total entropy is zero.
a) The entropy change of the lead block is
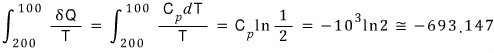
The entropy of that fluid is
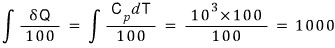
That is, the change in total entropy is 1000-693.147 = 306.853 J/K
Now I’ll do the cooling in two stages.
The entropy change of the lead in round 1
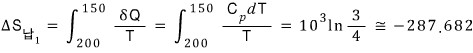
The fluid entropy change in round 1
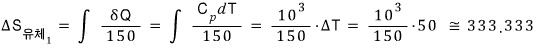
The entropy change of the lead in round 2
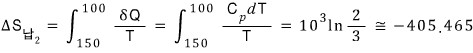
The fluid entropy change in round 2
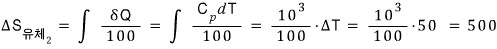
Therefore the change in total entropy is
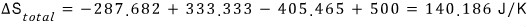
c) Now I’ll try doing it infinitely many times.
Looking at the entropy change of the lead, the integrals have this property of merging into one??!?!!
Then if I set the difference in fluid temperatures as ε,
plop into 200-ε,
plop into 200-2ε,
plop into 200-3ε,
.
.
.
.
plop into 200-nε
Then the change in total entropy is

That is, the change in total entropy is

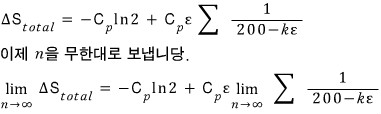

Prob 14. 7
n moles of gas are confined at temperature T and volume V.
a) When it becomes αV through reversible isothermal expansion,
b) When the partition is removed and it undergoes free expansion into a vacuum, derive the expression for entropy.
i) Isothermal expansion necessarily requires heat to be supplied.

ii)

Originally written in Korean on my Naver blog (2016-01). Translated to English for gdpark.blog.