Gibbs' Expression for the Entropy
A casual (and slightly reluctant) walkthrough of Gibbs' entropy formula, showing how entropy gets re-expressed through probability over macrostates and microstates.
Now it’s the last one on entropy.
I was going to skip this concept, but I’m writing it down just to record that I did try it at least once….
Ha.. entropy, seriously entropy….lollollol
Let’s go
Based on the concepts we learned before, they say we’re now going to express entropy not thermodynamically,
not by number of cases, but by probability.
Apparently we can do that.
Say~~ there’s a system that happens to be measured as some several macrostates,
and for each of those macrostates, let’s assume there really are multiple microstates that have the same macrostate. (I’m talking about micro Entropy~)
Then,
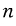
macrostates are possible,
and for the specific i-th macrostate,
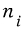
microstates represent that macrostate.
That is, if we say the number of all~~~ the ‘microstates’ that the system can have is N,
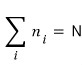
then this holds.
Then the probability of the i-th macrostate appearing can be expressed automatically.
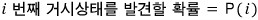
if we say this,
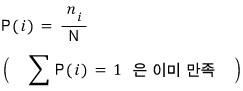
And so
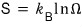
using this definition, writing the entropy of the system we laid out as
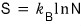
is the entropy considering all possible microstates.
The entropy considering the macrostate is
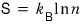
and figuring out this one is the goal!!!!!!
What we can find out experimentally isn’t the microscopic stuff… it’s the macroscopic stuff….
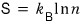
So is this value the truth~~~?????!!
Nope.
In the previous example we got a glimpse of the possibility of additional entropy,
and that possibility (the value with micro entropy added) S + Smicro
we said this would be the total entropy.
Computing Smicro makes no sense!!!!! no sense!!!!!
But, we did assume that there really are multiple microstates that have the same macrostate…..
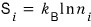
Ha…. since we can’t know this guy,
let’s at least compute the expectation value! is the idea.
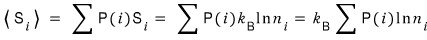
That is,


Since earlier we already said
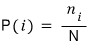
,
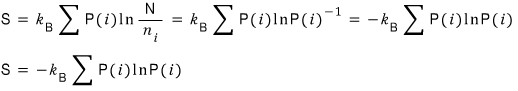
like this, entropy can also be expressed as the probability of having a macrostate.
This expression for entropy is called “Gibbs’ expression for the entropy.”
Prob 14. 8
The probability that a system is in the i-th microstate is
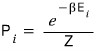
.
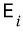
is the energy of the i-th microstate, and β and Z are constants.
Show that the entropy
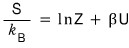
is given as above. Here U is
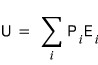
the internal energy.
Well, Gibbs’ expression for entropy
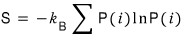
let’s plug in the Pi above.
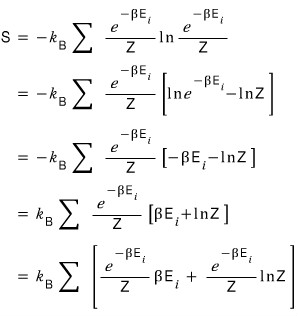
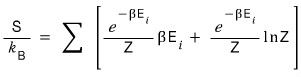
Got this far.
Let’s run toward the answer.
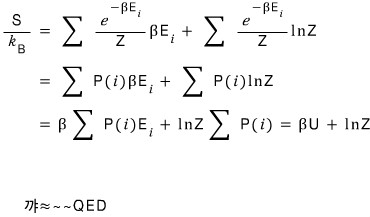
Originally written in Korean on my Naver blog (2016-01). Translated to English for gdpark.blog.