Enthalpy, Helmholtz Free Energy, and Gibbs Free Energy
Starting from dU = TdS - pdV, we add clever terms to both sides to cook up enthalpy, Helmholtz, and Gibbs — basically just a change of variables to make life easier.
Alright, now, finally, let’s take that “conservation of energy” equation dU = TdS - pdV, and by adding the same thing to both sides,
by adding ΔU, we’ll look at other kinds(?) of energy.
Saying “other kinds” is a bit off… um…?
I think it’s more like a change of variables.
As you’ve all probably caught on, I’m about to define the enthalpy, Helmholtz, and Gibbs functions.
After defining all~~ of these functions!!
Why on earth did we define these functions.
What on earth is the meaning of each function?
Let’s dig in. (A slight spoiler: the content is about how, in such-and-such cases,
one kind of energy equation is more convenient, easier to handle, and meaningful.)
Let’s just get started.
① Enthalpy function
The units of p times V are ’energy’. Right!!!
So take the equation of the first law of thermodynamics, dU = TdS - pdV,
and let’s add Δ(pV) equally to both sides.
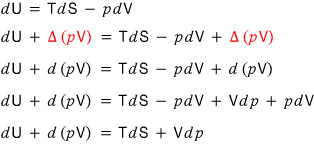
Now let’s write the left-hand side as d(U + pV). That’s okay, right?!


② Helmholtz function
Before, we added d(pV) equally to both sides of dU.
This time, we’ll add -d(TS)!!!!
Are the units of TS energy????
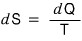
The units of S from this are J/K.
The statistical mechanics definition of entropy is
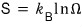
and the units of the Boltzmann constant are J/K.
(The reason the constant has units of that shape can be answered by thinking about how the statistical-mechanics entropy was derived.)
Anyway, since there’s no room to doubt that the units of TS are energy,
let’s look at dU - d(TS).
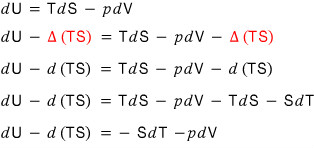
Now let’s write the left-hand side dU - d(TS)
as d(U-TS), and go into the definition!!!


③ Gibbs function
This time, let’s do the trick we did to dU for the enthalpy AND the trick we did to dU for the Helmholtz simultaneously!!!
That is, let’s apply +d(pV)-d(TS) to dU!!!!!
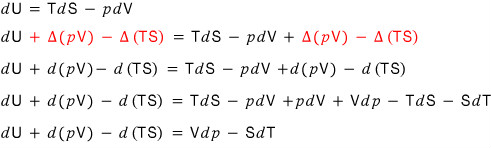
Now let’s take the left-hand side dU +(pV) - d(TS)
and write it as d(U + pV - TS), and go into the definition!!!


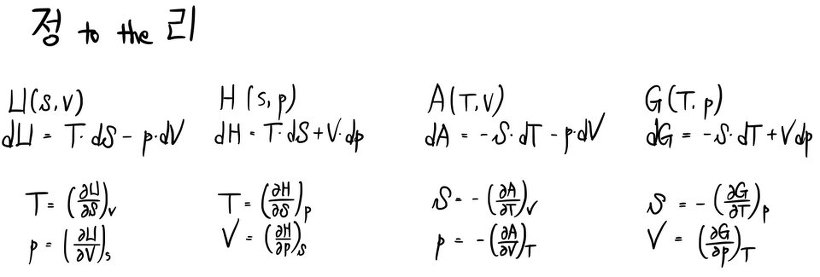
We’ve done something, and we even did the summa to the ry,
but what on earth did we do. What do those things actually mean?
Why do we have to look at the same energy as 4 kinds of this sort of crap — U, H, A, G….
Why’re we learning this, huh? Let’s think about it once.
First, let me transcribe the definition of ‘free Energy’ as it’s introduced.
‘free Energy’: a physical quantity that depends on the available and useful amount of work the system can give.
Big underline under “available”, and a little pig-tail squiggle~, and let’s move on.
And one more thing — let me introduce the concept of availability.
It’s said to be a concept used in extreme~~ly general situations.
The extremely general situation usually means: in chemistry experiments, many experiments are said to be carried out under constant temperature & constant pressure.
Okay, so let’s say we’re doing an experiment at temperature
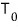
constant, and pressure
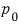
constant.
In this setup, let’s write out the first law of thermodynamics.
dU = δQ + δW
Now, if δQ amount of heat flows from the environment into the system, and the system does work,
the amount of heat that flowed in from the environment, δQ,
this much heat came in, and let’s say the system did work such that its volume changed by dV.
Then, we can say that we did at least
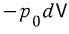
worth of work ‘clearly’ (visible to our eyes, and measurable).
So, as far as we know, out of the δQ worth of energy,
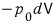
this much has already been used, so let’s subtract that much off.
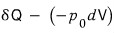
Since this is subtracting the energy worth of the ‘work done’ from the incoming heat δQ,
I’ll write the remaining energy as
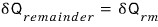
.


(If you skim this, it’ll probably be hard to understand…T_T)
We played around with the remainder.
That is, the equation above can be interpreted roughly as ““the amount of work that can still be done””.
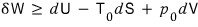
And let’s understand this inequality like this.

That is!!!!
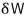
The minimum value it can take is
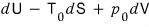
this much!
At the very least, this much is available!!!!
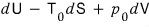
This is what’s defined as availability. I’ll denote it with F.
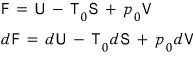
That is,
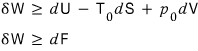
On top of this, since chemistry experiments are usually mechanically constrained,
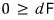
Let’s look at various situations with this inequality.
This inequality is said to represent the ‘principle of minimization of availability’.
But let’s not forget our assumption.
This equation was derived assuming an experiment with temperature
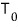
constant, and pressure
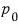
constant!
(So, although I don’t know for sure, I’m guessing that chemistry folks are the ones who defined availability.)

“That is, the equilibrium state is where S has its maximum value”
This is the conclusion we can draw.

“No way, this!!! It’s the Helmholtz function!”
//
“Wait!!! The Helmholtz function means, for a system at “constant temperature, constant volume”, the availability,
that is, the ‘free energy’!!!
Wow~~ And on top of that, at Equilibrium the Helmholtz function will be a minimum!!!!”
Now I see why it’s called the Helmholtz free Energy.

“Ah!!! For a system at constant pressure and constant temperature, it’s the Gibbs function that’s the availability…
that is, it means the Free Energy!!!”
Now I get the meanings of the Helmholtz function and the Gibbs function.
Then what about enthalpy!!!!!!
Enthalpy, they say, shows its power in a system at ‘constant pressure’.


Summary again!!!!!!
Internal energy:
At constant temperature and pressure, the sum of the kinetic energies and potential energies of the particles that make up the system.
Helmholtz free energy:
At constant temperature and volume, the useful work that can be obtained from a closed system.
Gibbs free energy:
At constant temperature and pressure, the non-mechanical work obtainable from the system, and spontaneity.
Enthalpy:
At constant pressure, the heat energy going into and out of the system.
From here on, it’s Maxwell Relations!!!!!!!!!!!!!!!!!!!!!!!!!!!
If I keep writing it here in this posting, rocks will come flying, right?!?!?!?!
I’ll continue in the next posting!
Originally written in Korean on my Naver blog (2016-01). Translated to English for gdpark.blog.