Isobaric Expansivity, Adiabatic Expansivity, Isothermal Compressibility, and Adiabatic Compressibility
Wrapping up the chapter with a casual intro to susceptibility — covering isochoric/isobaric heat capacities and all four flavors of expansivity and compressibility.
This post. It’s the wrap-up of this chapter.
We came over here after doing the Maxwell relations, but, well, it’s not like there’s a direct connection….
Just think of it as sort of a wrap-up.
As for the Maxwell relations, later on, when we solve problems, I’ll show how to put them to use.
This time. We’re going to define a new concept called ‘susceptibility’.
So first, we need to look at heat capacity.
Actually, we could have looked at it earlier, but since we’re going to use it here, I’ll organize it here.
And using that, let’s go ahead and define this thing called susceptibility~


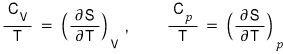
Having organized it like this~~
Alright, so now at last we’re going into susceptibility.
Susceptibility in English is suseptibility, and the meaning, if you look it up in a dictionary, is ‘sensitivity’.
Huh???~ Sensitivity to what???
No no, I should put it this way.
What is sensitive to what?
The definition of susceptibility is precisely:
‘Sensitivity of volume V’. It’s about how sensitively volume V reacts to some change~ that’s what it is.
Well then, as for what change volume V reacts sensitively to!? — well,
It’s precisely the sensitivity of volume V to changes in temperature T and pressure p! — that’s what we’re looking at.
Again, again, “depending on the substance, the sensitivity of the volume change according to temperature change differs!!!”
“depending on the substance, the sensitivity of the volume change according to pressure change differs!!!”
That’s the concept, and expressed as a formula

Well, it could be written like this.
For now we’ve only grasped the rough concept.
So now I’ll throw out the real 4 definitions of susceptibility.
● isobaric expandivity
● adiabatic expansivity
● isothermal compressibility
● adiabatic compressibility
Up above a moment ago
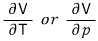
I put it in this crude kind of way, but.
But the definitions being introduced now are not the ‘amount’ of change of V with respect to temperature or pressure change, but the “rate of change”.

We’ve been continuously playing with the 4 variables T,V,S,p,
When differentiating V with respect to T, with the condition that S or p is constant,
When differentiating V with respect to p, with the condition that S or T is constant,
And so in this way they’ve been divided into a total of 4.
And to add on, as for “adiabatic”, it means δQ = 0
That is, TdS = 0, that is, dS = 0 → S is constant.
I’ve added on even the part about why adiabatic means grabbing S and differentiating.
Ex 16. 5
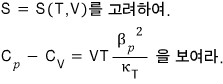
I’ll write the real definitions, re-organized with ‘rates’, of the things above.
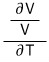
If you do it like this, it becomes the rate of change!
Then I’ll write it containing even the meaning of ‘isobaric, adiabatic, isothermal’.
Since we were told to consider that S has been defined as a function of T and V,
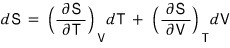
we can write it like this.
If we differentiate both sides of the above expression with respect to T holding p fixed,
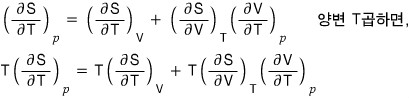
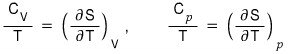
I organized this earlier, and if we apply it,

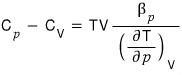

Therefore
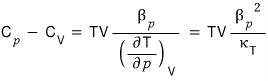
QED
Originally written in Korean on my Naver blog (2016-01). Translated to English for gdpark.blog.