Chapter 16 Practice Problems
Working through Chapter 16 problems on the four thermodynamic potentials, Maxwell relations, and general identities using the reciprocity theorem.
Prob 16. 1
a) Using the first law dU = TdS - pdV, write the definitions of the 4 thermodynamic potentials.
That is, write the definitions of the 4 thermodynamic potentials U, H, A, G, and find dU, dH, dA, dG in terms of T, S, p, V and their differentials.

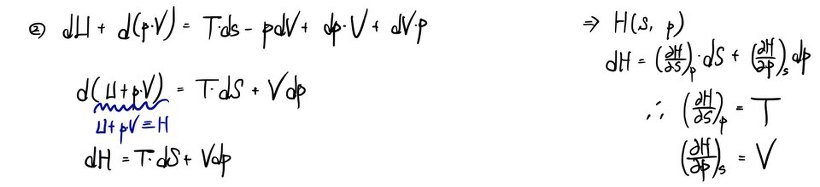
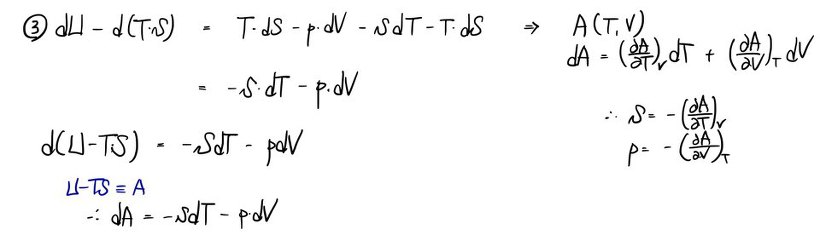
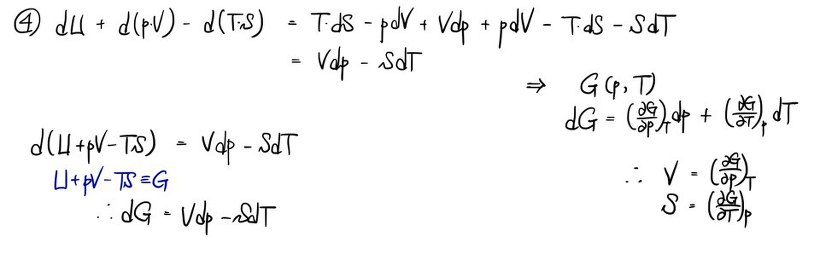
b) Derive all the Maxwell relations.




Since I derived them in detail in the Maxwell relations posting, I’ll just cover it with photos~
Prob 16.2
a) Derive the following general relation.
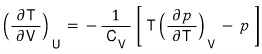

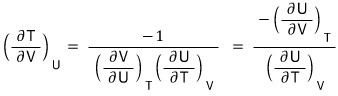


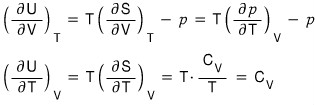
That is,
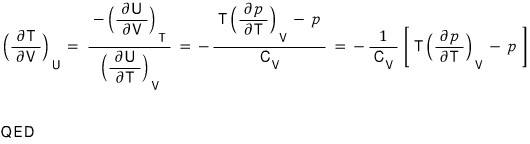
ii)
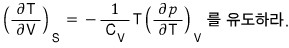
The LHS,
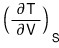
let’s mess around with it a bit using the reciprocity theorem first
(because the RHS is a PTV rotation, and the third Maxwell relation is the PTV rotation and STV rotation, so I want to rotate the LHS variables and then use the Maxwell relation!!!!)

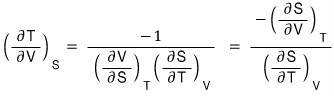

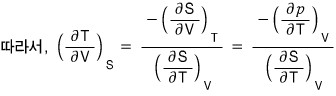

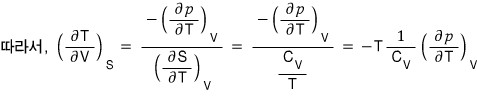
QED~
iii)
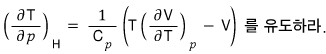
After doing a T, P, H rotation, using dH = TdS + Vdp,
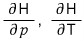
let’s use these.
So first let’s apply the reciprocity theorem to the LHS and rotate the variables a bit! (I’m not sure if it’s a good expression) rotate rotate! hehehe
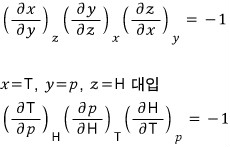
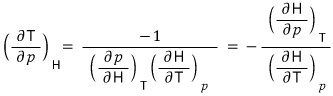
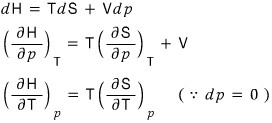
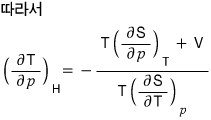


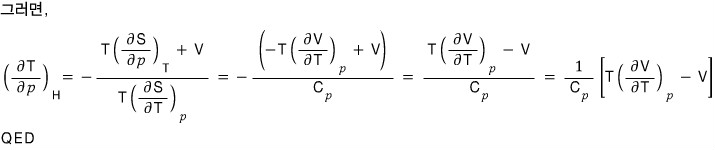
b) Using these relations, for the ideal gas prove
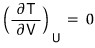
, ,
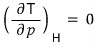
.
(The 3rd problem,,,, what does it mean to follow along an entropy line… T_T)
i) Holding U constant means… for an ideal gas, T is constant.
Therefore.
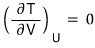
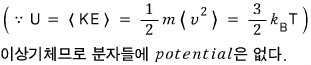
ii)
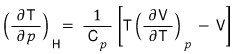
Since it’s an ideal gas, let’s use pV = nRT.
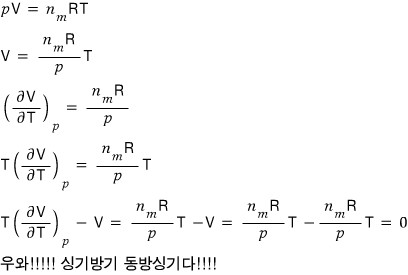
Prob 16. 5
Using a thermodynamic argument, show that for any gas at temperature T, the pressure is given by
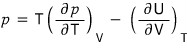

Huh?? It’s done???
Prob 16. 3
Using the first law of thermodynamics, prove
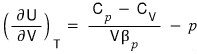
.
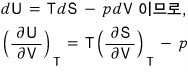
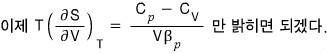

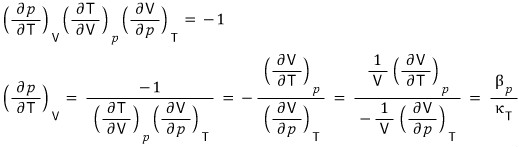

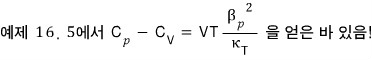
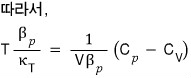
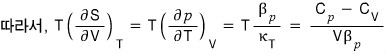
The proof is done, but,,, it feels unsatisfying….
Prob 16. 4
The natural variables for U are S, V.
This means if we know S and V, we can know U(S,V).
Show that this fact has a simple expression for T and p as well.
? I don’t get the intent of the problem…
Is it asking to find T and p as functions of other variables?
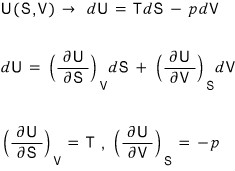
Or,

Is it this…?
b) Instead of knowing V, T, suppose we know the function U(T,V) (that is, we have U expressed in variables that are not U’s natural variables).
Show that from this we can derive an expression for p.
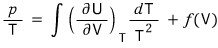
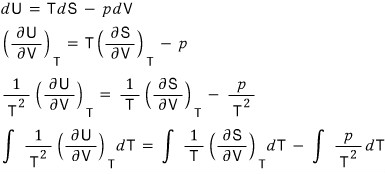


Since it’s the indefinite integral of something differentiated while holding V, we get a Function of V, not a constant~
Prob 16. 6
Another expression for the molar entropy of an ideal gas is
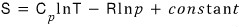
show this

Originally written in Korean on my Naver blog (2016-01). Translated to English for gdpark.blog.