Equipartition Theorem
Turns out when energy is proportional to a square, the average only depends on degrees of freedom — let's verify it step by step using the Boltzmann distribution!
In physics, it’s said that there are really many cases where “energy” is proportional to a square hehehe
Huh!? How can you declare that so definitively?!?!
Book, aren’t you just bullshitting me right now to make the explanation a bit easier?!?!
I’ll show you soon that the situation where energy is proportional to a square is actually quite universal…
For now, we just have to accept it first heh!!
So first let’s work through the material step by step
What this book wants to tell us with the equipartition theorem is this
“The average energy does not depend on anything.
It only depends on the ‘degree of freedom’.”
When, when??!?!?!
When the energy is proportional to the square of some variable!!!!
In that case it depends only on the degree of freedom
Let’s verify it!!
First, let’s accept this. “In physics, the case where energy is proportional to the square of some variable is fairly universal”
Let’s accept this first lol
So
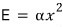
Let’s say energy is hung on the square of some variable like this
Here x is just some variable, it’s not a variable representing ‘position’
And this ‘system’, whatever kind of system it is, we don’t know,
each of those systems can exchange energy
and the overall system that encompasses each of them is thermally isolated… let’s assume that.
(Our overall universe can be said to be such a system, and what we’re talking about now is something like saying we’ll do statistical analysis in the concept of a canonical ensemble)
If we assume such a system, then as we learned in Chapter 4.
the probability that the system has energy
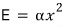
at temperature T will be proportional to the Boltzmann distribution.
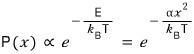
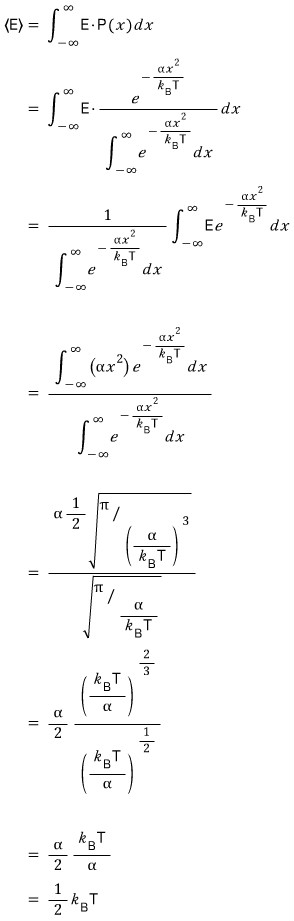
This is saying the probability is proportional to the Boltzmann factor, but we’re not curious about the probability, we’re going to find the expectation value now
So because we need the exact probability
we’ll change the above proportionality into an equation

Writing it like this, well, there’s no boundary condition either, so there’s no way to find out what the coefficient is.
….how am I supposed to figure out that ‘some coefficient’ over there……..
We don’t need to worry about this.
We can express the same probability differently!!!

If we reformulate it like this, we can now compute various expectation values
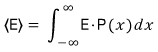
As for why the expectation value is calculated this way
I don’t want to describe it…..I’ll put a few links…..it’s something I described in detail a long time ago lol
http://gdpresent.blog.me/220446758426
Quantum Mechanics I Studied #1. On Quantum Mechanics, On Probability. (Chapter 1 Section 1…)
Alright~ Now the reason I enrolled in the Physics department! The moment to study quantum mechanics has come. At the time of writing, currently 2 weeks…
blog.naver.com
Then I’ll continue the calculation.
To continue the calculation there’s a necessary integral, and I’ll link that necessary integral
I proved it a long time ago lol (it’s such a simple principle it’s hard to even call it a proof.)
http://gdpresent.blog.me/220584291592

Thermal·Statistical Mechanics I Studied #9. Proof of integrals frequently used in thermal·statistical mechanics (link only)
I won’t derive this! Because it’s easy, I’ll pass over this, and by applying differentiation with respect to α on both sides of this resulting equation, thermal·statistical…
blog.naver.com
So now I really calculate!!!
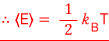
“Point to feel”: no matter what α is, it has no effect whatsoever on the average energy
Now what follows next is
We found out that the α hanging on the square has no effect whatsoever on the average energy.
But at the very beginning in the intro, I said that only “degree of freedom” is involved in the average energy, right!?!?
Then we should go find out about degree of freedom
To get a feel for this degree of freedom, let’s solve an example
And then, explanation, let’s go
Example 19.1
The energy of the system
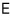
is given as the sum of
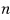
independent squared terms
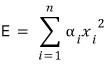
where
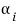
is a constant, and
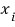
is a variable. Assume that each
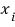
can take on arbitrary values with equal probability.
Find the average energy in this case
Then first let’s think about 2 of them
Since the variables hanging on the energy E are said to be independent of each other
let’s just think of them as particle 1, particle 2, and let’s say that particle 1 and particle 2 have no interaction with each other lol
The probability that some variable of particle 1 is
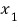
would be
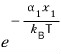
and
the probability that some variable of particle 2 is
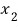
would be
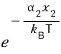
well
Then, the probability that 1 is
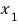
while simultaneously 2 is
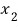
would be
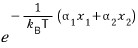
(since they’re independent,)
Now if we change this shape into a form suitable for computing the average, it could be represented like this.
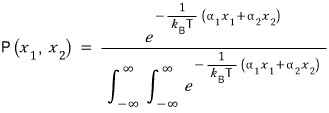
Oh yeah niiice!!!
Then now, since we’re going to find the expectation value of energy~~~~ let’s also think about the total energy~~ and the moment I say that, it’s over.!
Because we said they’re independent of each other
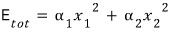
So then now, let’s find the expectation value (average) of the energy!!!

Each term, regardless of the value of α, becomes
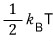
— we already sorted that out above, right??!?!?
So the result of the above integral is
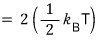
The problem said a sum of n squared terms,
the reason I did 2 is because I was trying to show that they cancel out like the red ones and the blue ones.
Then if it had been n squared terms hung on, there would be n terms
and each of the n terms would be organized into
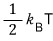
so the expectation value of the total energy with n squared terms is
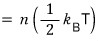
What we’ve organized like this was actually what our title was,
the equipartition theorem!!!!
Aigoo…..what the heck is this…T_T T_T
First let me transcribe the energy equipartition theorem.
Energy Equipartition Theorem:
If the energy of a classical system consists of the sum of n squared mode terms
and the system is in contact with a heat reservoir at temperature T, then the average energy of the system is
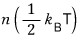
.
Seeing it like this, now I’m starting to get the feel.
In the example above, the total energy was
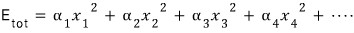
it was, but
When doing the calculation, I verbally interpreted it as, for n particles, the variable x of particle 1, the variable x of particle 2, and so on.
That’s just how I said it, and from now on is thinking time, so
let’s think of the individual
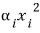
terms as just each one independent factor that determines the energy in a single system.

Something like this!!!!
So even if there are a crap-ton of various n factors, if those terms are hung on the total energy as a linear sum in the form of “squares”, the energies get equipartitioned!!
That’s what I was just saying!!!! Isn’t it amazing?!?!? lolololololol
Even if I called this the “Beauty of Nature” lolololololol I’m sorry lolololololol
Wondrous nature~~ ahhh~~~~~
So then, let me sort out one term and move on
n is now the ’number’ of terms that the energy of the system depends on as a square, right?!?!!
To say this a bit more difficultly, you could call it “the energy dependence of squared terms"
and in English they say “mode of system"
But more than that expression, the term degree of system (the system’s degree of freedom) seems to resonate more.
Originally, degree of freedom is a term often used in statistics
and in that statistics, what is the concept of degree of freedom
If you know that well, these things here also resonate better
But, I found a place that organized statistical degree of freedom well, so I’ll attach the link

Why divide by degree of freedom n-1 in sample variance?
Why do we divide by degree of freedom n-1 in sample variance? First, comparing the variance formulas of the population and the sample, you can see they differ by n-1. This n-1 is…
math7.tistory.com
Here, if I were to forcibly translate ‘degree of freedom’ into Korean… I mean, spell it out in plain language,
it could be summarized as “the number of independent factors that determine energy” (I’ve been repeating the same thing over and over here….lol)
You’ll see why I described it this way once we move further along
I’ll keep going.
Let’s see how the energy equipartition theorem is actually applied.
Let’s say there’s a gas molecule like this.
Let’s also say we’ll consider only translational motion, ignoring the vibration, rotation, etc. of the gas molecule (an ideal gas only has kinetic energy to begin with)
Then the relation for energy is
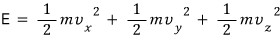
Huh?! The energy gets organized into 3 squared terms.
Then the degree of freedom would be 3, and following the energy equipartition theorem
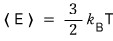
it would be
http://gdpresent.blog.me/220584329950

Thermal·Statistical Mechanics I Studied #10. Ideal gas expected speed, expected energy, speed of ideal gas…
Alright, so now I’ll stop talking about distribution. Now we’ll look at the average, so let’s look at ‘velocity’ again…
blog.naver.com
The proof is here, so I’ll skip it.
Are you now confident about the energy equipartition principle?!?!?!
We are now able to infer the expectation value of energy even when we face a slightly more difficult model. (If it’s hung only on squares, that is….lol)
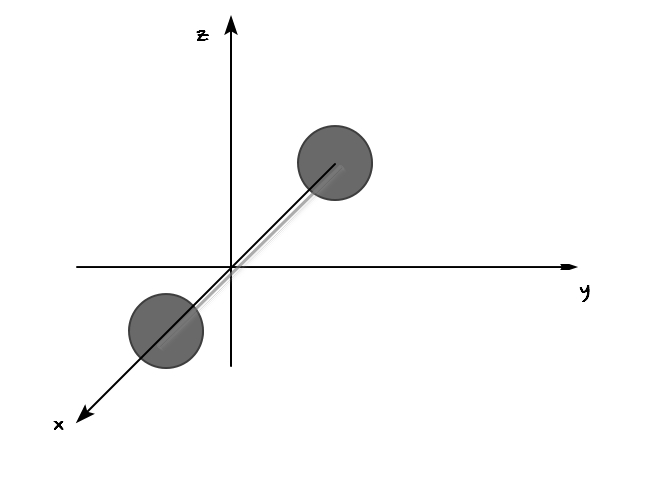
Let’s look at a diatomic that looks a bit complicated.
What would the degree of freedom be?!?!?!?
That is, how many terms are hung on energy as squares?!?!
First, the terms for translational motion
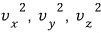
will still be there
And here now rotation is also possible.
But,
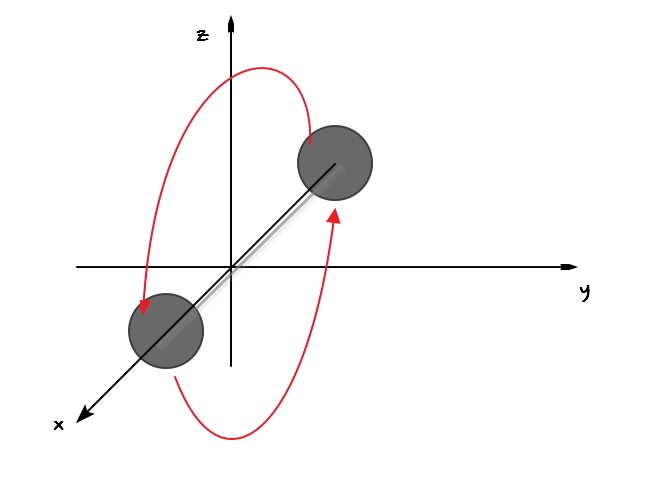
Compared to rotation about the y and z axes
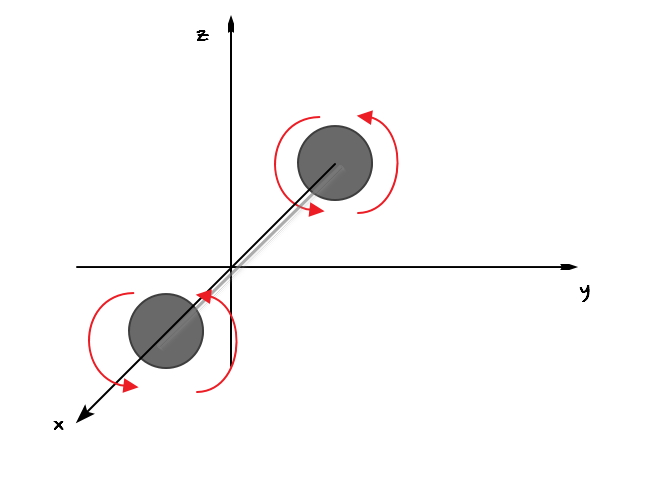
rotation about the x axis has significantly less angular momentum
so the energy term can be ignored, and the energy term is
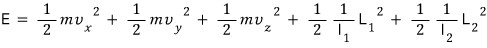
So there are 5 terms hung as squares, and the degree of freedom is 5.
The expectation value of energy resulting from the equipartition principle is
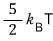
, and how do we verify this?!!?!?
It’s said that experiments back up the fact that this value is not wrong!!!!
If in that shape above, it were a spring instead of a rod?!?!?!?!!
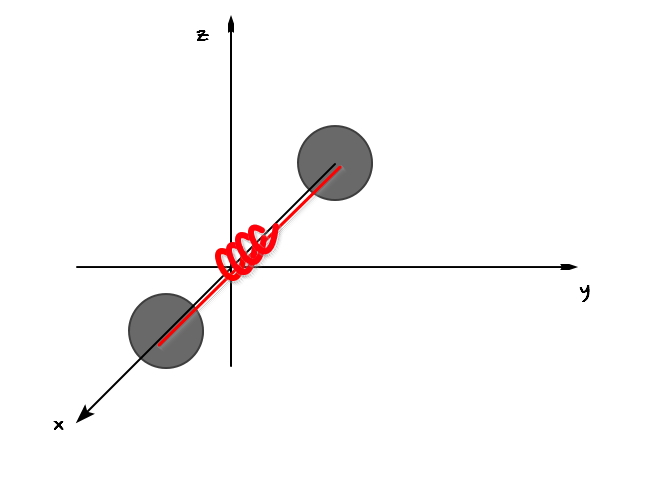
If I hadn’t known the energy equipartition theorem, I think I might have just thrown this problem away lol
But since I know a powerful law of nature, it’s worth tackling!!!
First, the terms it had when it was a rod will still be there.
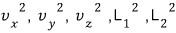
Hmm… and as the spring model is added, what energy term should be added?!?!!!
First, since it’s a spring, the potential for the spring
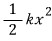
a term like this will be added.
And one more term
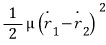
arises, and
why does this arise
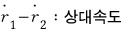
μ is the reduced mass.
If I explain the concept of reduced mass, probably the reason that term arises will become clear
First, when is the reduced mass concept needed?
It’s needed in the two-body motion problem
And it can’t just be any two-body.
What kind of two-body does it have to be? It has to be a two-body “that has interaction force between them”.
That’s why reduced mass was first introduced in the universal gravitation part of general mechanics.
Of course, when looking at the hydrogen atom from the electromagnetism side, there’s also the proton-electron interaction, so reduced mass could be needed
but when dealing with the hydrogen model in electromagnetism or quantum mechanics, we didn’t use the reduced mass concept
Why was that?!?!?
That’s because
when the reduced mass concept is needed is “when the mass gap between the two objects is not large”.
(The mass difference between a proton and an electron is about 1000-fold)
So when there’s a two-body with interaction force between them, and the mass difference isn’t too large, using the reduced mass concept….
was in order to reduce the two-body problem to a one-body problem.
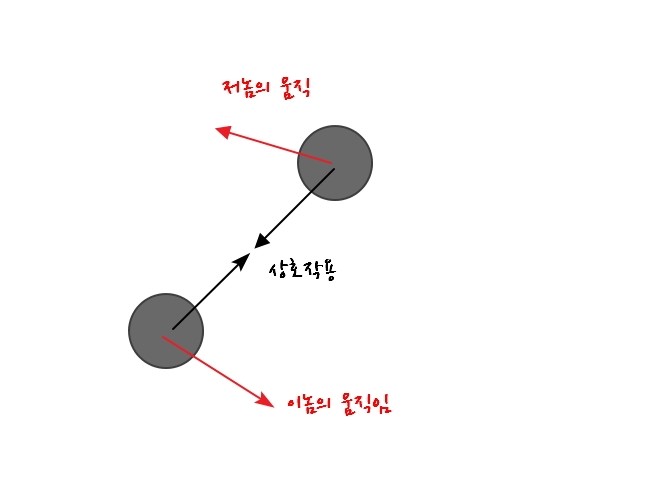
A problem in this kind of situation
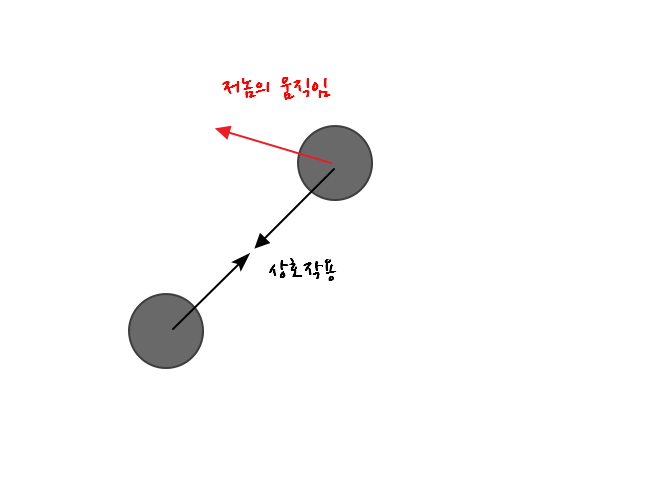
the concept that transforms the situation like this is the reduced mass.
Then why is the name Reduced Mass?!?!?!
If you manipulate some moving object to view it as stationary
the mass of the moving object has to be converted again
and at that time the mass becomes slightly reduced, that’s why the name is reduced mass.
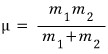
So when the two atoms above connected by a spring are lumped together as one
then viewed as if there’s one reduced mass
and then the kinetic energy that can’t be ignored due to the interaction force, the relative energy, will depend on the “relative velocity”.
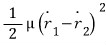
(While continuing translational motion, aside from that, you can understand it as something like relative kinetic energy (like 1/2 mv squared) I guess?!!!???)
That’s why this interaction term arises!!!!! hahahaha
Then finally
It’s content I’ve organized before, but using the degree of freedom and the energy equipartition principle
let me revisit heat capacity once more and move past this posting

And we already know that for an ideal gas
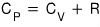
holds!!!!
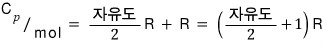
we can organize up to here hehehe hehehe
Really lastly
I’ll look at solids just once!! and then I’ll finish lolol
Up until now I considered gases,
now I’m going to briefly think about the energy equipartition theorem for solids
Is it okay if I draw a ‘solid’ here like this?!
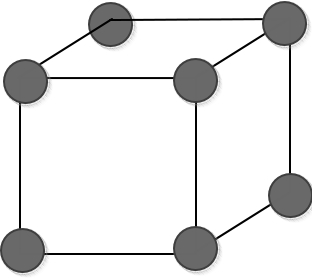
It seems like no one would object to drawing it this way.
Because, any solid being made up of the bonding of some atoms is a very obvious idea that even humanities students know
But, let’s say we gave the cube above a “thump~”.
Then the solid got distorted and
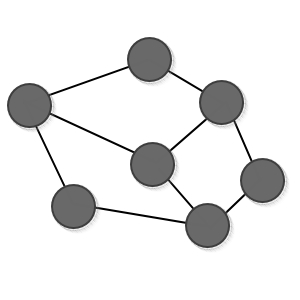
let’s say it became this state
Then of course, there are certainly solids that keep this form continuously
but the majority of solids are not like that and come back to their original place
(Try tapping your desk right now. The desk doesn’t stay in the distorted state, right???)
That is, because they want to keep staying in the most stable state, solids tend to return to that state.
So scientists, in order to inject the property that such a majority of solids have, do a lot of little tricks on the model it seems
How do they play a lot of tricks?
They draw the model like this and think about it.
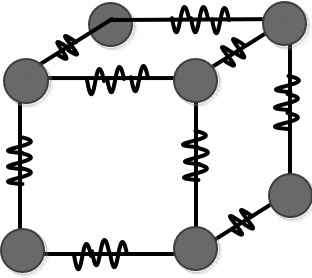
If we draw it like this
we can add each atom’s characteristic and tendency of wanting to return to its original position to the model!!!
Then now if we say, let’s look at the energy of some one atom in this solid!!!!
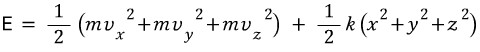
Well, we can organize the energy like this, right?!?!!
Then we can catch that the degree of freedom is 6, and by the energy equipartition theorem, the average energy of a solid at temperature T is
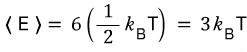
can we say this?!?!!!!
Discussion of this continues in Chapter 24.,
first, this resulting value is said to almost agree!!!!
We’ve justified the energy equipartition theorem once more like this~~~~ hehehe lol
Originally written in Korean on my Naver blog (2016-04). Translated to English for gdpark.blog.