The Partition Function
A casual deep-dive into what the partition function Z actually means — from its German roots to why dividing by Z magically turns Boltzmann weights into real probabilities.
First, let me just slam down the definition of the partition function:
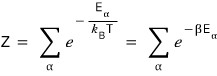
The reason we specifically use Z, out of all the many letters of the alphabet, to denote the partition function —
I’ll transcribe it.
“The partition function is denoted by the symbol Z, because this concept was originally named in German.
Zustandssumme means ‘sum over all states,’ and this exactly captures what Z is. The English name ‘partition function’ indicates the way Z shows ‘how energy is partitioned among the states of the system’: Partition function.“
Actually, earlier we were treating this concept just like a counting-of-cases thing, understanding it as “the sum of probabilities of each of all possible states existing,” and we were subtly using the Z concept already.
So let’s dig a little deeper into the meaning of Z.
It was “the partition function is the sum of the probabilities of being in some state,”
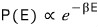
and since we want to turn this proportionality of the Boltzmann distribution into an equation, we put in a coefficient
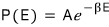
and write it like this
And if the possible energies are
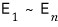
then
at temperature T the probability that the energy is E_k is
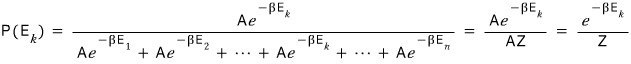
and while playing around like this you can see that the definition of Z just pops out.
What does Z mean…
You can grasp it easily in this way:
the probability of being in a state with energy E is, as a factor,
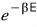
proportional to this — but
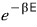
itself is not a probability.
We want to call that thing a probability, I’m telling you?!?!!
And right then, snap~~~ if we divide by Z
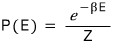
then we can call it a probability, you see?!?!!
We’ve seen stuff like this in lots of places
When you flip a coin,
say heads came up 30 times — you can’t call that 30 itself a probability, right????
So we divided by something like 30/75, or 30/300, i.e. the “size of the probability distribution,” which is the number of trials.
That is, Z can be understood as this kind of “size of the probability distribution.“
Now now now now, but,
the partition function Z plays a very important role in statistical mechanics.
As we go along you’ll see right away why Z is so important
(spoiler: if you know Z, you can derive U, F, G, H, p, V, S, etc. etc. That is, I’ll go so far as to boldly claim that all the information in thermal and statistical mechanics is melted into Z.)
To give a sense of the scale: if classical mechanics has F=ma, electromagnetism has Maxwell’s equations, and quantum mechanics has the Schrödinger equation,
then statistical mechanics has the partition function…. it’s important at about that level.
If you want to analyze anything in statistical mechanics,
the first button to fasten will be computing the partition function
> A preview of the limits of the partition function
└ collapse
OK so in solving problems in statistical mechanics the first button will be “computing the partition function,”
so: yeah, so (if possible) how do we compute it?!?!?!?! — let’s go get the answer to that.
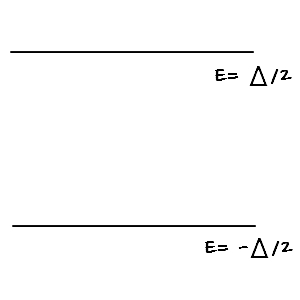
Tough situations can wait; first let’s compute the partition function in the easiest easiest easiest case!!!! (we’ve done this one before too!!!!)
The definition of the partition function is
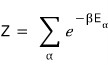
so
plugging in the possible energies for the two-level system above:
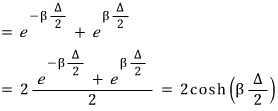
Eh… this was a bit much. Way too easy, no….???
Then let’s compute Z for the second simple model, the simple harmonic oscillator!!!!!
Quantum-mechanically, the allowed energies for the harmonic oscillator were
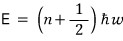
right
So applying it to the definition of the partition function,

This one’s easy too hehehe
Now let’s consider rotational energy.
By rotation here I mean spin ^^hehehehehehe
That name that makes your chest tighten just hearing it…. spin….hehehe
First, classical-mechanically the rotational energy is
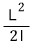
.
If you understand it as
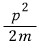
it’s not hard, right???
But quantum-mechanically the angular momentum L had to be defined as an operator,
so we split the angular momentum into components
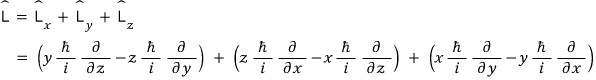
and had to express it like this,
but this also couldn’t serve as a good weapon, so the physicists’ drastic measure was to
turn their attention not to L as the variable of interest, but to
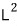
.
That’s because L^2 has a commutator of 0 with each and every one of L_x, L_y, L_z,
and commutator being 0 means the commutation law holds,
which means you can specify them ‘simultaneously,’
and in matrix-mechanics terms, what enables “simultaneous diagonalization” was precisely L^2, which is why they took this drastic measure.
And if you massage L^2 nicely
using the Ladder operator:
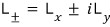
you could express it like this.
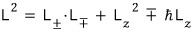
You can write it like this,
and writing it this way you can easily find the eigenvalues
the eigenvalue of
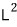
is
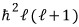
and
the eigenvalue of
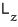
is
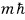
you can find them like this…
Ah… I was trying to keep it short and came way too far.
For the details, please refer to http://gdpresent.blog.me/220573887676 .
Quantum Mechanics as I Studied It #21. Angular Momentum
Okay!! Everyone, 2nd semester starts now! You’re screwed!!!hehehehe it’s gonna be a very fun time hehehe I’m sure…
blog.naver.com
Anyway, the energy of the rotational state is
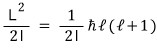
that’s what it is.
Okay then we can compute Z, right?!?!!!
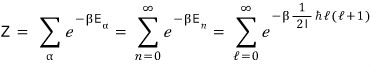
Is it done if we compute it like this?!?!?!?!?!!!
nononononononononononononono
There’s one more thing to think about
there isn’t just one state with this energy, you know
the number of states that can have angular momentum l…. that is, the number of degenerate states is (2l+1) for each l.
Why?!?!?!!!!!
The ones that can have angular momentum l are
can be -l, -l+1, . . . , l-1 , l, these are the possibilities,
so as many as that number of cases can have angular momentum l.
(e.g. l=2 is possible with m=-2, m=-1, m=0, m=1, m=2 all of them)
So when summing energies in the partition function, you have to do a weighted sum with this weighting for the number of states.
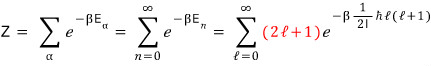
Kyaa~~~~ so if you write it like this, we can count up the partition function Z~~~~~~~~~
I’ll do the detailed calculation in the post where I solve practice problems.~~~
Aaaaaah and, that red part up there —
please keep in mind that it’s the number of states that can have E_n
because it’s going to be treated very importantly later on~~~ lol
Originally written in Korean on my Naver blog (2016-04). Translated to English for gdpark.blog.