Relationship Between the Partition Function and State Functions
We derive thermodynamic state functions like U, F, and S straight from the partition function Z — turns out one sneaky differential trick does all the heavy lifting!
In the previous posting I said something like this
That the partition function Z can be seen to contain all the information about U, F, G, S, T, p, V and so on
So this time it’s going to be a session where we derive those state functions from the partition function Z.
Alright, let’s start
For the internal energy U, by the first law of thermodynamics, which signifies the law of conservation of energy,
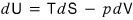
it was defined like this.
But that’s the change in U, let’s look at U itself, U
Since U is ’the energy of the interior'
it wouldn’t be too much of a stretch to just call it the “expectation value of the energy of the system,” right?!?!
Then if we first describe the expectation value of the energy, we can call that result U
The expectation value of the energy is
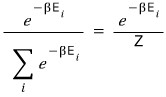
the probability of having this
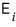
multiplied by
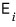
and summed up all together, so
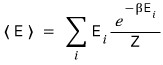
let’s write this as U.
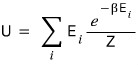
But if we write this as
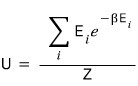
and look at it, we can express the numerator above using Z
Because

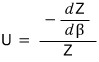
This is what we’ve got up to here, and if we work our brains a little more we can express it as
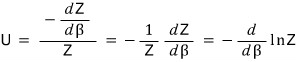
we can organize it this far
But β is actually
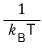
right??????
It’s something related to temperature,….. do we really have to express it as ‘differentiation with respect to β’….T_T
I want to express it in terms of temperature T…T_T T_T T_T
I mean I want to reveal that this is differentiation with respect to temperature T hehe
alright alright alright
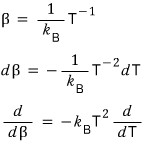
then
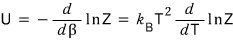
it becomes like this
But actually, this isn’t quite the precise expression,
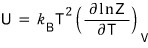
is the precise expression.
Now let’s move on to enthalpy, Helmholtz, and the Gibbs function
But before doing those, first I want to express entropy S in terms of Z
Because if we know S when deriving the state functions above it’s convenient~~~ hehehe
The statistical mechanical definition of entropy was
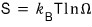
And here
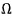
meant ’the number of microstates’
I mean how can a mere human take the number of microstates as a variable
So Gibbs re-expressed the expression for entropy probabilistically

Here
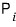
meant “the probability of finding the system in the i-th macrostate.”
Now by expressing P_i in terms of Z, we have room to express entropy S in terms of Z!!!
The probability of finding the system in the i-th macrostate (if the energy of the i-th macrostate is E_i) is
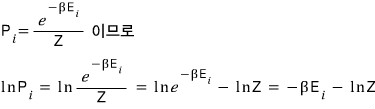
Now if we plug this straight into the Gibbs entropy expression
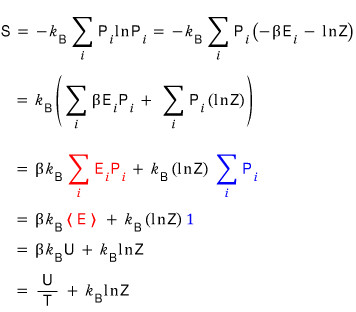
Now if we plug into U here the U that we expressed above in terms of Z
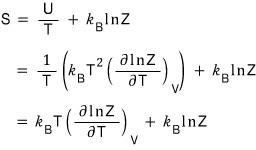
Yo~~~ entropy is done too~~~~
Now let’s move on to the Helmholtz function F
(In Thermal Statistics 1 we wrote the Helmholtz function as A, but from here on, following the book, I’ll write it as F T_T T_T T_T T_T hehehe)
In Chapter 16 we define the Helmholtz function, and
if we define F as F(T,V), the definition of F was F = U - TS, and
the definition of its change was dF = -SdT - pdV
Ah, now that we’ve defined both U and S in terms of the partition function earlier,
F should be easy to derive just by substituting.
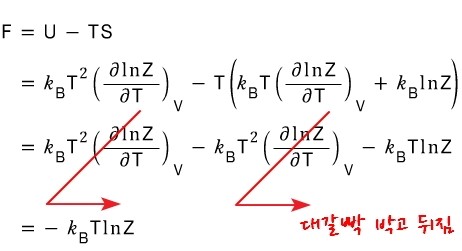
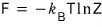
There’s a bit of content we can extract from this, so let me pick out some of the juice
The meaning of the Helmholtz function was that, in a situation where T and V are fixed, it represents the free energy of that system
The amount of energy that can be converted into ‘meaningful’ energy available in that system?
That’s the kind of thing free energy meant,
so look at where that Energy — which you can understand as being something like potential energy among mechanical energies — is hanging
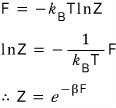
I’m planning to show off this fact when I deal with the Grand Canonical, so
it would be good to keep this fact in mind.
- Since F is a two-variable function of T and V, the mathematical notation of dF is
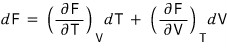
But thermodynamically, since dF = -SdT - pdV
that is,
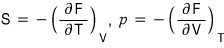
If we apply
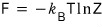
to this relation (it was a Maxwell relation),

Let’s sort out the pressure p too hehehe
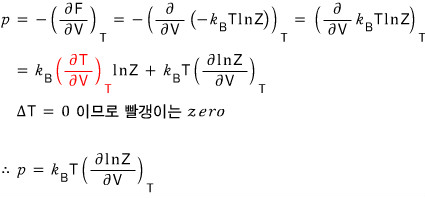
Let’s handle enthalpy too!!!!
Since the definition of enthalpy was H = U + pV
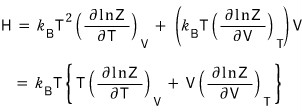
Let’s handle the isochoric heat capacity too hehehe
Since the meaning of the isochoric heat capacity is “if you keep the volume fixed and raise the temperature by thiiiiis~~~~ much, by how much does the energy change~~~~?^^”
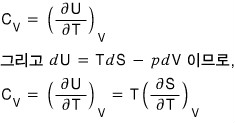
Now if we plug in here
the equation for the entropy S that we extracted above,
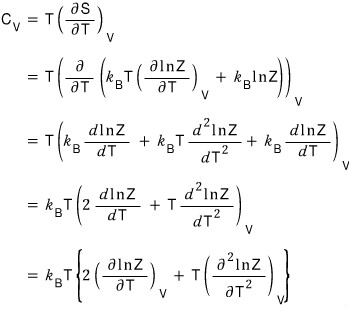
Now it’s getting a bit tiresome,,,, but still as the last one let’s handle the Gibbs’ function too.
We write G = U + pV - TS like this, and G was G(p,T), a function with T and p as variables!!!! so~~~
We can just substitute in everything we got before and be done with it…hehe
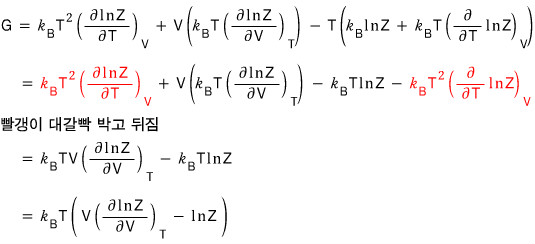
Everything we’ve sorted out so far……
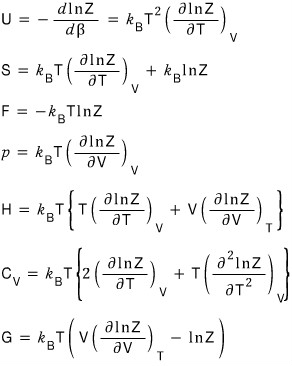
Originally written in Korean on my Naver blog (2016-04). Translated to English for gdpark.blog.