Partition Functions for a Two-Level System and the Harmonic Oscillator
We check that the partition function formulas for U, C, and S actually make physical sense using a simple two-level system — and yep, at 0 K everything ends up in the ground state. Ohong~!
With no thought at all,
we derived U, F, G, S, p, C, and so on.
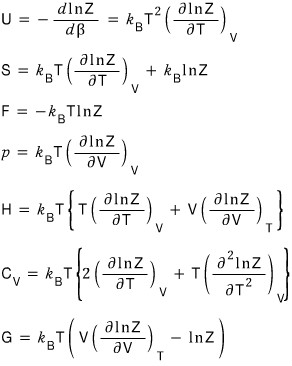
I’m going to give some justification that those equations are meaningful!!!
In other words, I’m going to look at the Physical meaning.
Doing all of them is too much, so I’ll just check U, C, S.
So let’s check U, C, S for the easiest and simplest two-level system.
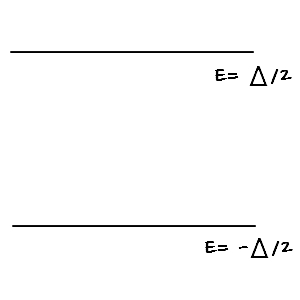
First, let’s find the partition function.
Z was organized like this~
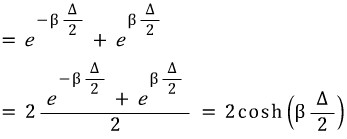
Then, let’s derive U through the partition function Z~

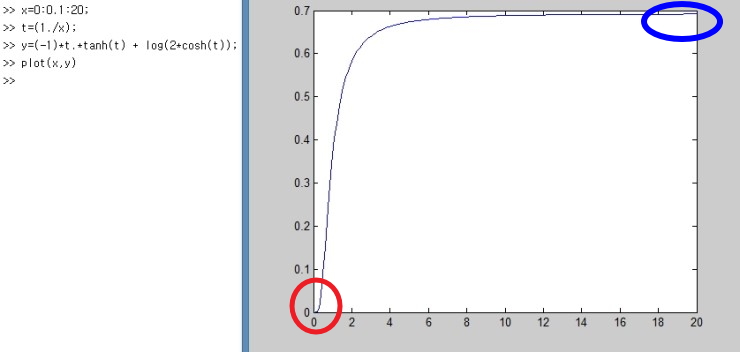
Let’s just plot it simply with Matlab lololol
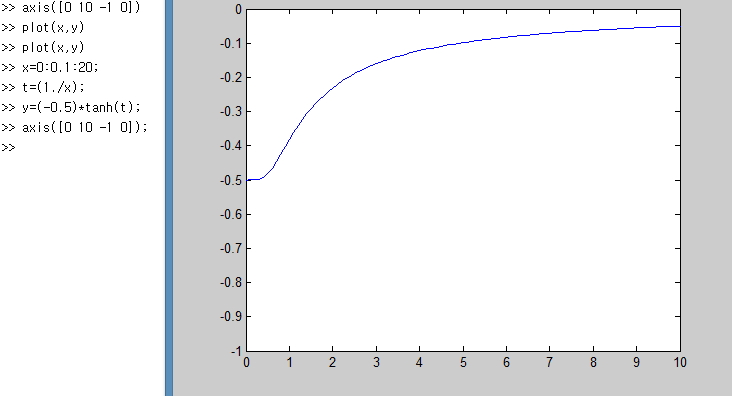
I plotted it like this.
Let me point out the parts you should focus on here.
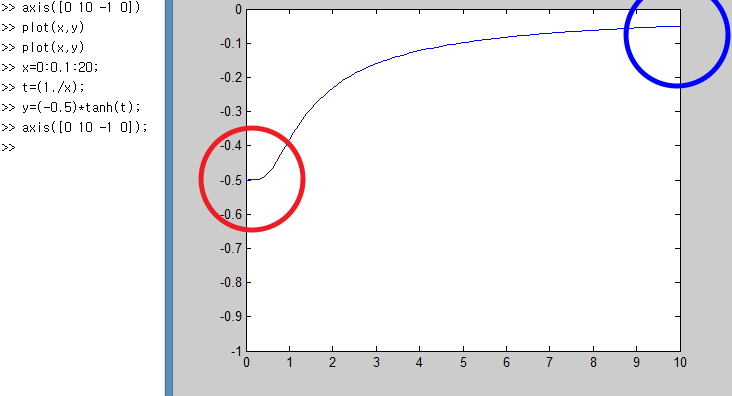
x is the variable that means temperature T, and at zero Kelvin
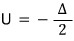
is what it says.
What does that mean….
It means that everything (every particle) in the two-level system
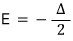
is in this state,
and that means at 0 K everything is in the ground state!!!!
Ohong~!!~~!! this makes sense physically too hehehe
And again and again and again, when T → ∞, (U/Δ) converges to 0,
which means when the temperature is crazy high, of the particles
half are in the state E = -Δ/2, and the other half exist in the state E = Δ/2,
which means the probabilities of being in each state become equal,
and we…
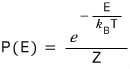
had this, right?!?!!!
When T goes to infinity, the probabilities become equal..hehehehe
This also meyk sense!!!!hehehehe yeahp
Then now let’s also look at the entropy.
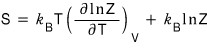
since this was what we had, let me substitute Z and solve it.
Z was
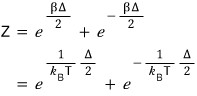
was what we had.
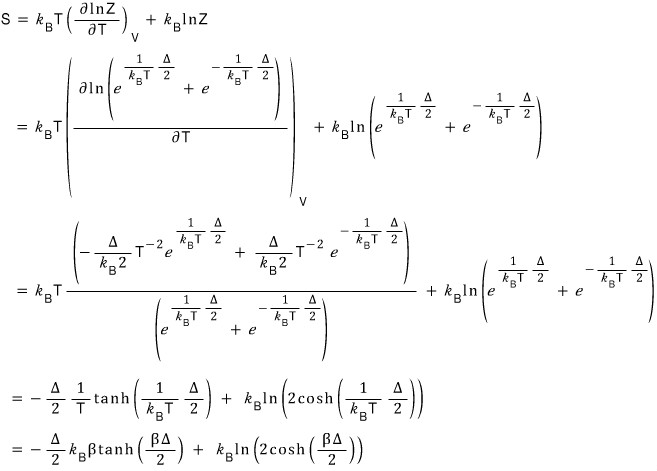
So I just plotted this simply, roughly with Matlab too hehehe
Of course there are parts to focus on here too hehe
It’s this part right here
The red part shows that at 0 K, S = 0,
and S = 0 means that the number of possible microstates at absolute zero is 1!!!
So that means it allows only one case where everything is in the gro~~~~und state, right?!?!!!!
And the blue part converges……
Why does it look like it converges….
If we focus on it like this and think about it
entropy
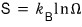
since it’s a two-level system,
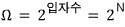
is what we have.
Hmm… it makes sense that it converges hehehe
Then let’s move on.
This time let’s also take a look at the heat capacity at constant volume in the two-level system~
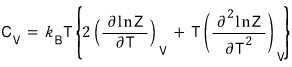
was what we had….
Ah…. I’ll just,
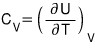
use this.
Above
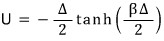
we showed this, didn’t we?????
Now if we differentiate this with respect to T,
<Note (tanh(x))’ = sech^2(x) ……….isn’t this the kind of thing everyone forgets the moment they graduate high school? lolololololololol>
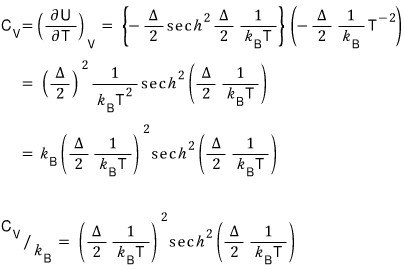
I plotted this with Matlab too~
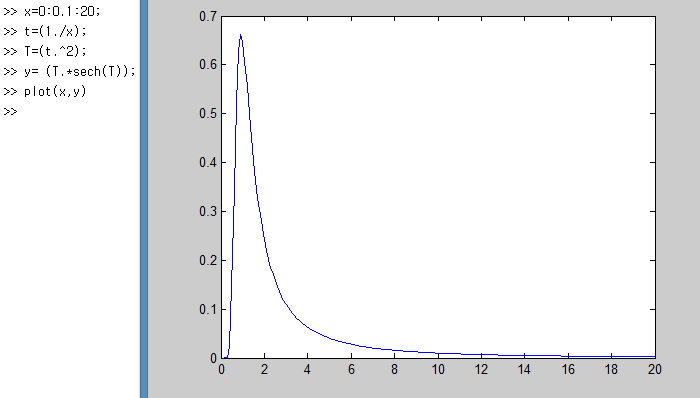
There are 3 parts to focus on here.
Right here!!!
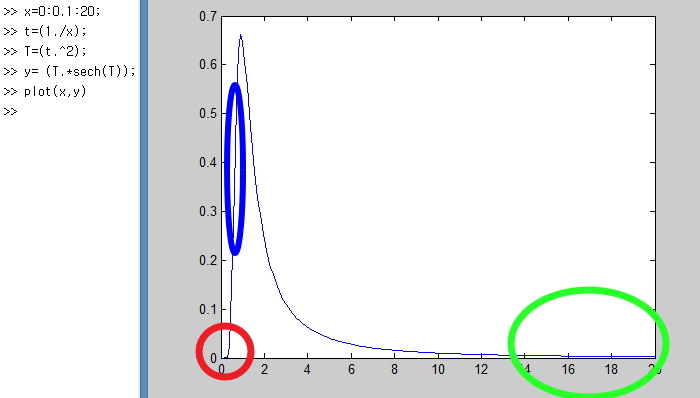
If you look closely at the red part first,
there’s an interval where C_v doesn’t rise even when you increase the temperature T quite a bit.
The reason?!?!?!?!?!!!
A minimum amount of energy needs to be applied
before a particle can do a quantum jump or whatever, right?!
It means that minimum energy is needed and small changes in temperature can’t change the system’s energy state.
This
also explains the part in the U graph from before where the red part is somewhat horizontal and then goes pang~ shooting up!!!!!
And the blue part of the C_v graph!!!!
There’s a part where C_v shoots up bang bang bang like crazy.
This part is called the Schottky anomaly interval, apparently.
It’s a part where, despite small changes in temperature, the change in energy of the system is crazy huge!!!!
It means the number of things doing quantum jumps is overwhelmingly larger than the number doing quantum down!!!!
Oooh
And the last green part!!!
The green part means when T is too high, the ratios of things in the high energy state and the low energy state become constant,
and the ratio of jumps to downs becomes about even, so for small energy changes there’s almost no change in the system’s energy.
Ain’t that hard hehehe
Also let’s look at the simple harmonic oscillator that we organized before.
For the simple harmonic oscillator, the partition function could be easily found as an infinite geometric series.

For the detailed derivation,
http://gdpresent.blog.me/220689943911
Thermal·Statistical Mechanics I Studied #33. The Partition Function
First, to throw in the definition of the partition function,the partition function among the many…
blog.naver.com
it’s all in there,
and we’ll interpret U, S, C the way we did above.

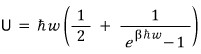
There’s one note.
If we’re looking at when T is insanely large,
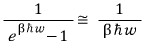
we can approximate it like this,
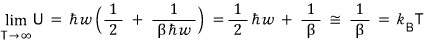
if we throw away (1/2)hbar omega too,
(1-dimensional) energy equipartition
we can also confirm — please take note of that.
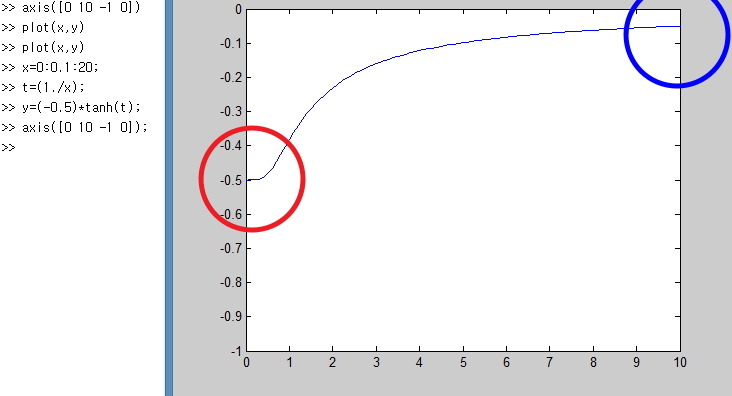
Alright, so if we plot the above graph,
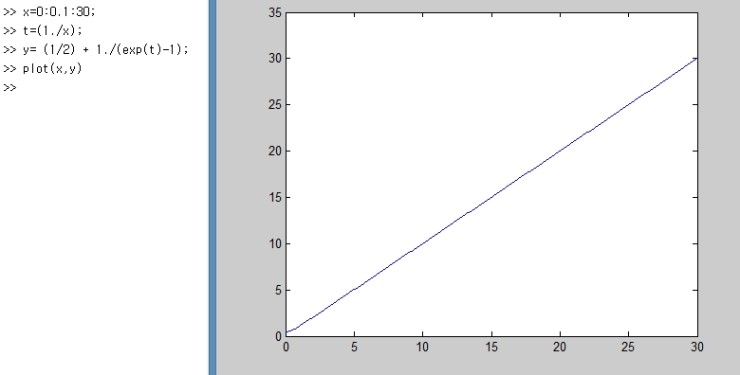
it gets plotted like this,
and the part we should keep an eye on here is
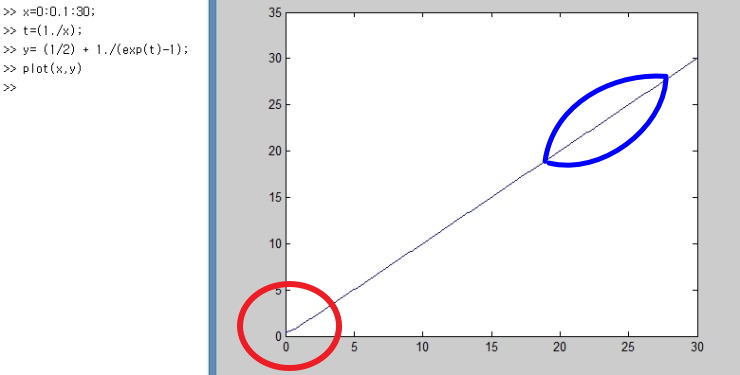
this part.
In the red part, it’s the point that U ≠ 0.
That is, even in the ground state there’s energy,
and the cause that quantum mechanics identified for this was something called quantum fluctuation in the ground state.
And the blue part — we did everything above, but as the temperature gets higher and higher,
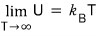
it gets closer to this,
and, theoretically, since there’s no limit on the energy in a harmonic oscillator,
that is, since everything up to n=∞ is possible, it means that as the temperature gets higher and higher, U also keeeeeeps getting higher!!!!
Alright, this time let’s look at C.
Let me look at the heat capacity C first!!!!!
I’ll fix V on the U above and take a partial derivative with respect to T.
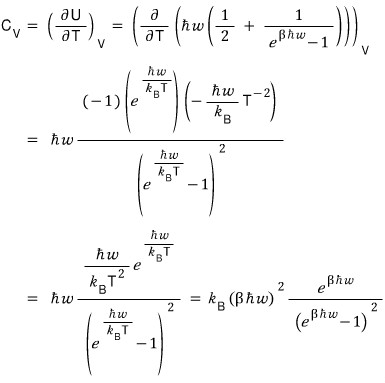
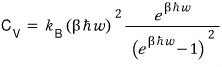
Let me try plotting this too!!!!
Ta-da~
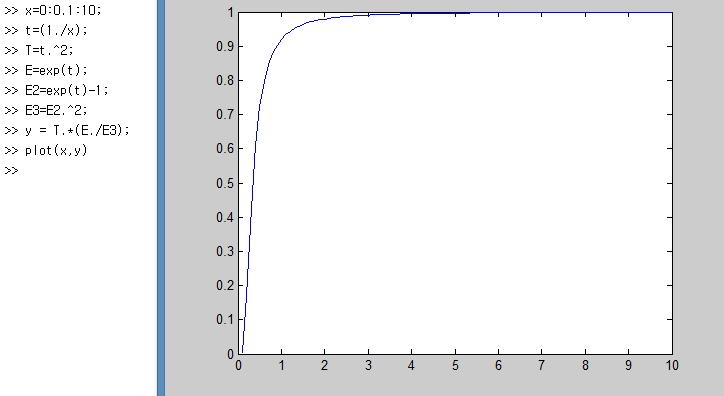
The place we should focus on here is
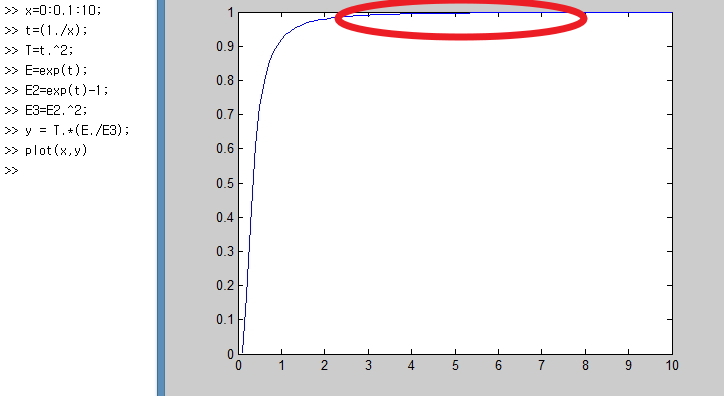
right here!!!!!
It means that when the temperature reaches a certain level, the variation of dU with T is almost constant.
Earlier above, U was
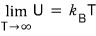
right????
Then what value is the variation of dU constant at?????
Yep, it’d be saying it’s constant at the Boltzmann constant.
We can also confirm that it’s constant at the Boltzmann constant via the C_v equation.
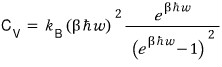
From this, we can derive it using the Taylor series expansion.

If you learned function limits in high school
and grasped the concept well at that time, you’ll know why we Taylor-expand the denominator but instead of Taylor-expanding the numerator, we send it directly to 1.
Since we’re not in math class right now,
now let’s move on to look at entropy S.
The equation for entropy S
is easier to derive via the Helmholtz function F.

The reason for using this is
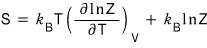
rather than computing the entropy out like this,
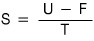
it’s easier to count it like this!!! So let’s plug in and compute it.

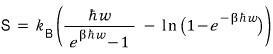
I plotted this too!!!!hehehe
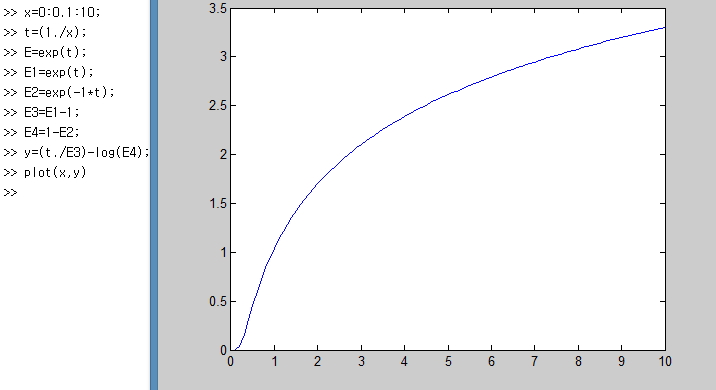
Let’s just look at the part to focus on for this too
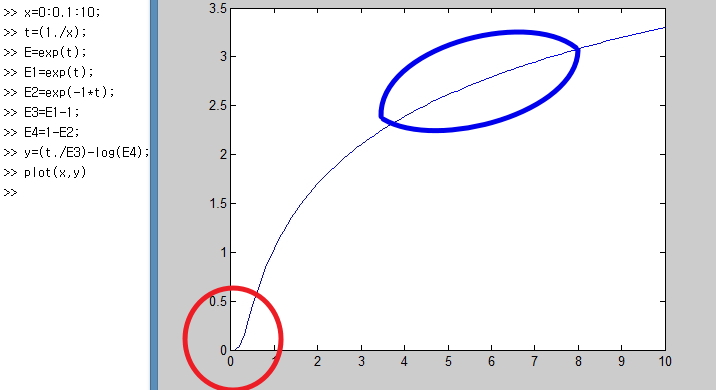
The red part!!
It’s showing with S = 0 that the number of possible microstates at 0 Kelvin is 1,
and that S also diverges here.
Why does it diverge?!?!?!
I mentioned it when we were looking at U, but,
it’s because right now the harmonic oscillator allows an infinite number of energy levels!!!!!!!!!
You know that picture (the one we saw a lot in quantum mechanics lololol)
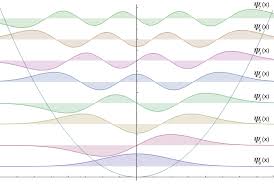
Since this kind of energy level is possible up to n=∞, the higher T goes to infinity, the energies will keeeeeeep going up too…hehehehehehe
So here’s a question.
Not n=2 like the two-level system,
but bigger than that,
and not extremely big like the harmonic oscillator’s n=∞ either,
what would happen if the allowed energy levels were, say, around N=20?!?!??
If N=20, they say it lands somewhere in the middle of what we plotted above.
(Plugging in N=20 and computing the equations isn’t difficult, but since I don’t think the numerical part is important, I’ll just patch it up with the graph from the book.
(Picture below: concepts in thermal Physics, stephen blundell, Lee Jae-woo, p293)
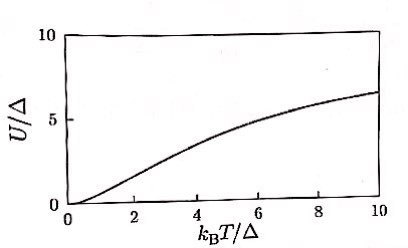
Doesn’t converge extremely like when n=2, doesn’t diverge extremely like n=infinity… this kind of lolololol
(Of course at T=infinity it’d converge, right? hehehe)
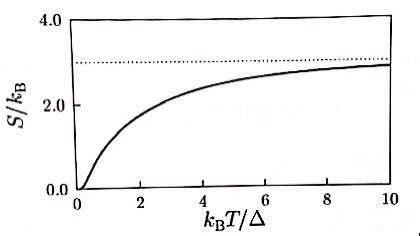
Entropy too — it doesn’t diverge like n=infinity, but converges at a higher place than n=2 — that’s the kind of picture!!!!
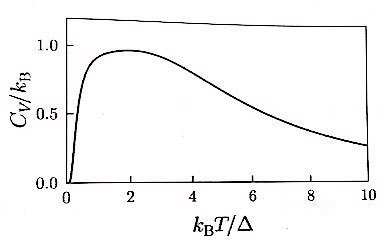
Heat capacity, heat capacity~
It means that for C_v to go to 0, a higher temperature than n=2 is needed.
Why? Because the time when the probability of being in each state becomes equal is at a higher temperature!!!!~~~~!!!!
Wow……………….
Sorry for writing such trivial content this long…
It’s really easy content but I feel like I made it feel unnecessarily difficult…hehehehe
But still it’s fun hehe
Originally written in Korean on my Naver blog (2016-04). Translated to English for gdpark.blog.