Density of States and Statistical Mechanics of the Ideal Gas
We finally tackle the ideal gas and why we can actually compute Z for it — turns out independent subsystems make partition functions way more manageable lol.
If I take a roo~ugh look at what we’ve done so far,
We find Z, then calculate U and F,
And through simple operations like differentiating that by T, or by V, or dividing by T and so on, we derived and analyzed various state functions,
There’s a dark backstory here.
The reason this was possible was????
As I mentioned before, the reason is exactly that we were able to find Z.
That is, if we can just find Z, for a system that has that partition function, we can basically call it game set.
But…..is reality that easy??
Just like our lives lolololololololol, if things worked out as we thought they would, well, reality would be OK to analyze.
Beot!!! (but) reality is not that easy.
lolololololololololololololololol we learn about life even in physics
Now, starting from this posting, we’re going to talk about the ideal gas,
And the conclusion is: for the ideal gas, we can compute Z.
But, at the start, I want to draw a big picture of why this is possible for the ideal gas before going in.
Let’s say we have ideal gas a and ideal gas b.
Then the energy of the total system with these 2 ideal gases can be written like this.
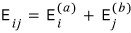
The right side means the energy when ideal gas a is in the i-th state,
And the second term means the energy when ideal gas b is in the j-th state.
Then we can write the partition function Z like this
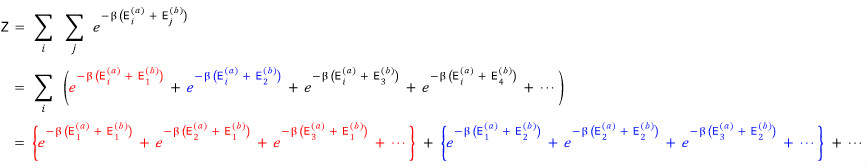
It’s small so it’s hard to see, let me just write the very last line.
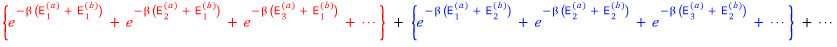
Argh, it’s almost the same lolololololololol
Anyway, now if you take a looo~~~k at each of those terms, you should feel the combinations.
The overall exp exponent part that can be made
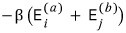
The combinations here
are the same as the number of cases you can make by picking one at a time from baskets like these.
So we can write it this way.
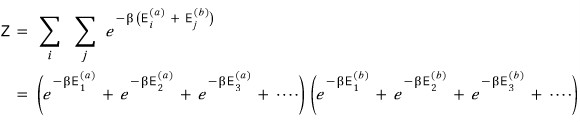
Whether you list this all out using the distributive law you learned in middle school,
Or look at it through the lens of the binomial theorem concept you learned in high school,
You can be sure that all the terms listed above come out the same.
That is,
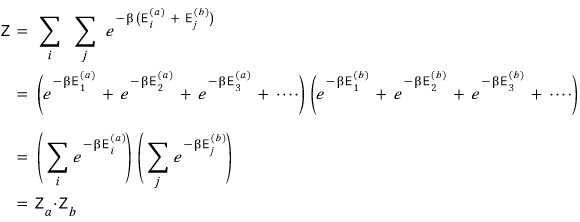
This is how we can find the partition function.
Did you catch on???? Why it’s possible in the ideal gas??!?!?!
Then before I write this posting,
I’ve picked out a few things to watch out for, both for me as I write and for you as you read.
i) It is said that for an ideal gas, each gas can have energy independently, without interacting with each other.
Ideal gas ideal gas ideal gas
ii) As you can see from above, it is said that ideal gases are distinguishable….
If you’re careless, you can mistakenly treat indistinguishable as distinguishable and cause errors….
So actually, for particles that do interact (representatively fermions or bosons), the distribution gets redefined.
And for indistinguishable identical particles (representatively fermions or bosons), the distribution also gets redefined.
Now then, since I’ve introduced the big picture like that, let’s jump right in.
This chapter deals with the ideal gas.
Do you all remember, from hearing it even back in high school until your ears filled with pus, the assumptions of the “ideal gas”?
Gas molecules have mass but no volume.
Gas molecules do not exert forces on each other.
All collisions caused by gas molecules are perfectly elastic collisions.
Gas never liquefies or sublimates at any temperature or pressure.
The average molecular kinetic energy of gas molecules is proportional only to the absolute temperature, and is not affected by the size, shape, or type of the molecule.
(https://ko.wikipedia.org/wiki/%EA%B8%B0%EC%B2%B4_%EB%B6%84%EC%9E%90_%EC%9A%B4%EB%8F%99%EB%A1%A0)
Well, there were assumptions like these.
Then let’s do the setting.
Gases that contain those assumptions
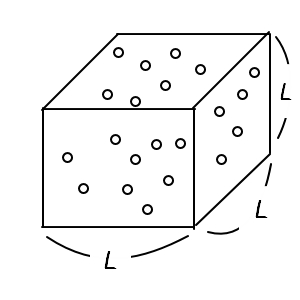
let’s say they’re in a cube of side length L like this.
Well now…. ain’t that a picture we’ve seen many a time?!?!?!
In the previous chapter, depending on what the temperature T is, we did this and that based on the Boltzmann-Maxwell speed and velocity distribution laws,
but now here, since we’ve learned the partition function, let’s first use the partition function.
So let’s first write down the definition of the partition function,
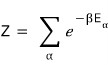
Now, but what is energy here?!?!?!?!
Do we have to do it like this?!?!!!
Alright, then here comes the core of this posting.
To find the partition function Z?!??!?!
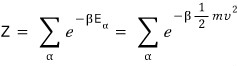
Since the key is to look at all v!,
If we write this as an integral (roughly)
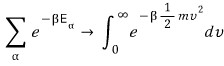
Is doing it like this fine?!?!?!?!
Nononononononono
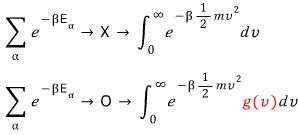
The core of this posting is that g(v) has to be attached like this.
Why does such a g(v) come into being and why do we have to account for it?!?!?!!!
That’s the core.
Let me spoil it a bit first: it’s “quantum mechanics — standing wave conditions” and “the number of cases that have the same energy”.
So in handling the ideal gas model, we won’t use the classical mechanical model; we’ll describe the behavior of the ideal gas quantum-mechanically.
To describe the behavior of the ideal gas quantum-mechanically, let’s first summon the ‘wave function’.
We’re looking at a gas inside a 3D cube right now, right?!??!!
Did everyone’s lightbulb flash?!?!?!?!
Yes, that’s right. It can be described as the wave function in an Infinite square well.
(The ideal gas has no chance of ever piercing through that cube, so we use the infinite square well model.)
That wave function is
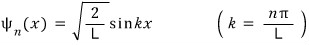
The derivation of this is at http://gdpresent.blog.me/220447856128 .

Quantum mechanics I studied #6. (Infinite square Well) infinitely deep square potential well
Now as announced before, we’re going to handle V(x) case by case, and the first potential case is the infinite s…
blog.naver.com
Huh???? But since we have to look at the behavior of ideal gas molecules inside a cube in dimensions,
is needed.
But! separation of variables should be possible right
It’s a static state where nothing is changing. And the ideal gas assumption of no interaction between each gas is included,
So it should be fine to say the x, y, z components are independent.
Therefore the ideal gas wave function inside the cube is
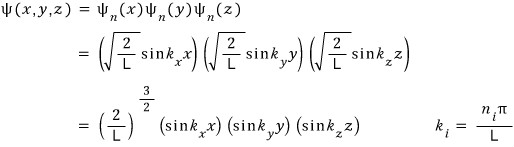
What always! followed when dealing with this was the expectation value of the energy,
(Because the square of the wave function is the probability density function)
So calculating the energy expectation value in 1D,
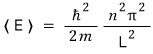
it was this.
Then I’ll just extend straight to 3D.
In 3D the energy expectation value will be like this. (Actually we do it all in quantum mechanics~~?)
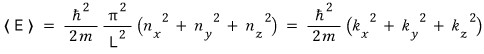
Ah….. why in the world did we come to talking about wave functions?!?!?!
Yeah yeah yeah…. above, I said I’d reveal the identity of g(v), and started the story with the wave function.
And I said g(v) means the standing wave condition and the number of states that have the same energy, right????
So we have to go back to the energy story.
Go go go go
We were looking at the partition function
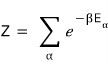
and decided let’s look at energy from a quantum mechanical perspective~~~
and that’s how we got here.
And then looking at energy,
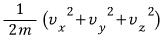
rather than this,
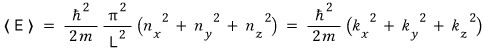
let’s say it’s this~~~
that’s where we’ve gotten to.
Now, but, I’m not sure if I can explain well from here lolololol
This is absolutely, totally dependent on k right now.
The moment k_x, k_y, k_z are fixed (that is, the moment the quantum numbers n_x, n_y, n_z are fixed)
So now we just look at E as a function of k.
(The reason we suddenly swiiitch~~~ from n to the wave vector k as the variable is because viewing the wave vector k as a variable of energy happens not just in this situation but in many situations.
So, as a story that’ll come up in a bit, since the concept called k-space is used not just here,
but also widely in other areas of physics, rather than writing in terms of n which is limited to the infinite potential well, from here on we take k — which can be used as a variable in many quantum mechanics problems — as our variable.)
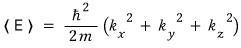
If we massage this equation just a little more, we can express it more simply.
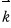
— we define this.
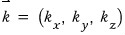
After defining it like this,
express
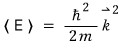
like this.
And,
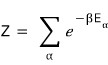
since in here E is now also absolutely dependent on the vector k,
we just go ahead and express it as
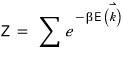
Alright, now what we’re going to do.
We’re going to introduce k-space.
http://gdpresent.blog.me/220584272242
Thermodynamics/statistical mechanics I studied #8. Maxwell-Boltzmann speed distribution (Maxwell-Boltzmann Distributi…
Originally, this content too, I was going to write it all in the previous posting as one single post, but if I did that the wall-of-text would be ridiculous so …
blog.naver.com
In this posting I introduced v-space,
And since it’s basically the same story, if you can follow the logic of the posting above well, it’ll be easy to follow the k-space logic.
(When I wrote that posting, I too had only dealt with Real-space before and was encountering v-space for the first time, so I described it very carefully. So the above posting will be helpful.)
Now….. energy is dependent on the vector k.
That is, it’s determined by k_x, k_y, k_z. (Actually this means n_x, n_y, n_z determine the energy, but still)
Then when we look at Energy states, looking at k-space is more convenient.
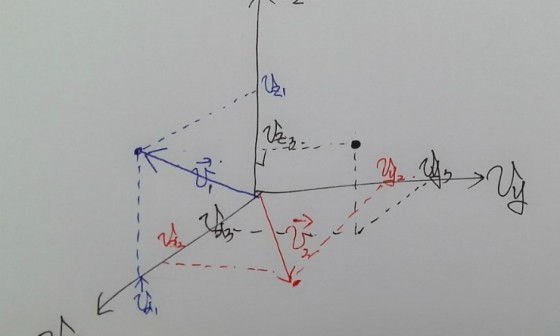
Say we identified the vector k in order to observe some state and handle the Energy.
For example
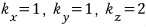
suppose.
If I plot this coordinate as a point in k-space,
I can plot a point like this.
(I won’t make a leap in logic. If the logic here doesn’t make sense, please definitely refer to #8.!! It’s very detailed http://gdpresent.blog.me/220584272242)
But.but.but.@
Is that point the only state that can have that Energy?!?!?!?!
Desu, nonononono, right?
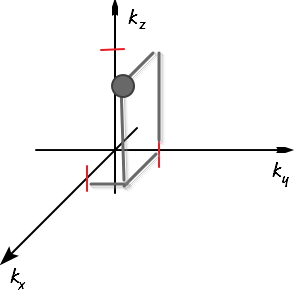
All (k_x, k_y, k_z) combinations representing all points on the surface of a sphere of radius 2 in k-space will all have the same energy, won’t they??!?!?!?
Let me plot all such points continuously.
Buzzzzz~~~
This is not it either.
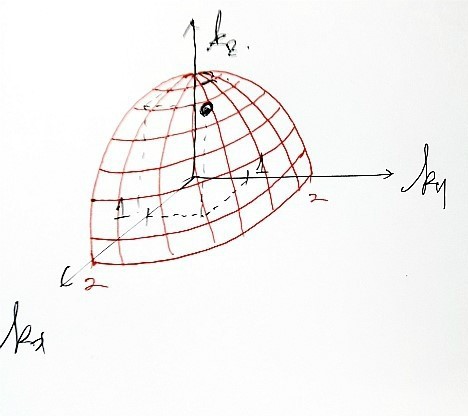
Because when you handle k-space, it’s not continuous like Real-space or velocity-space, so saying ‘on all of the surface’ is wrong.
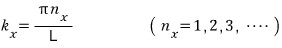
Because it’s this,
k_x is not continuous.
And since k_y, k_z are also not continuous, the possible ‘points’ in k-space are spaced out discretely.
(Wait!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!! Where does this condition that k is not continuous originate from? Yes!!! That’s right. It originates from the standing wave condition!!! I don’t think we should forget this hehehe)
If we sketch those possible k-space points roughly,
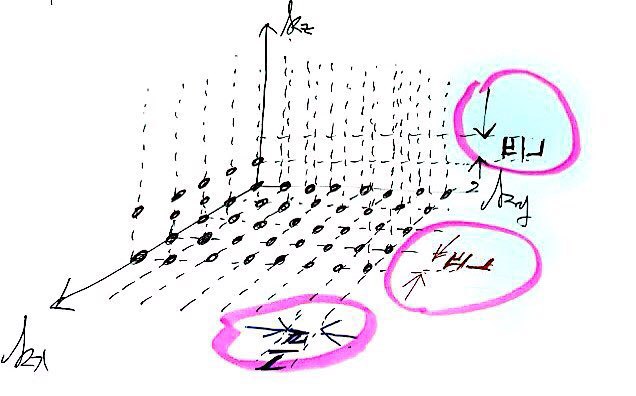
Doesn’t everyone kinda see what I was trying to draw?!?!?!
And those spacings are all equal, π/L!!!!
Then coming back to ’this picture’,
how many quantum-mechanically allowed points are on the red surface above?!?!?!
How does the book explain it? It counted the number by the concept of division.
Just in case, let me write down the concept of division I heard back in high school.
<‘Splitting’ is also fine, but the concept I’m going to talk about now, though the same story, has the origin-of-thought slightly different, and it’ll be good to have it too>
“Division” → “How many bundles fit in?!?!?”

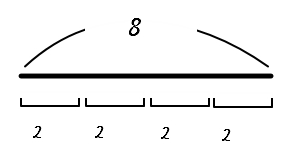
Let’s go go go go to 3D.

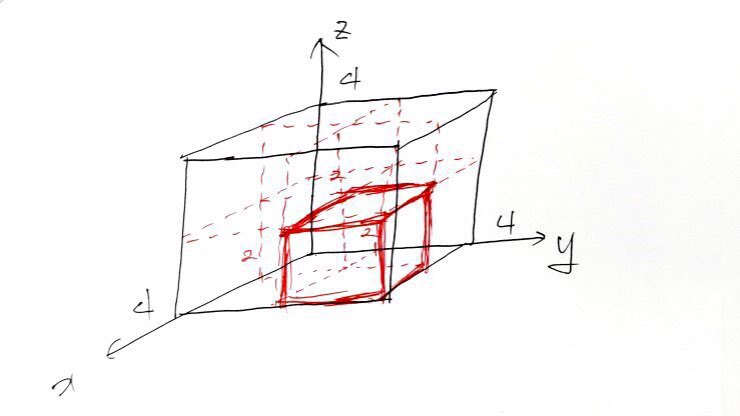
Alright so what we’re going to do,
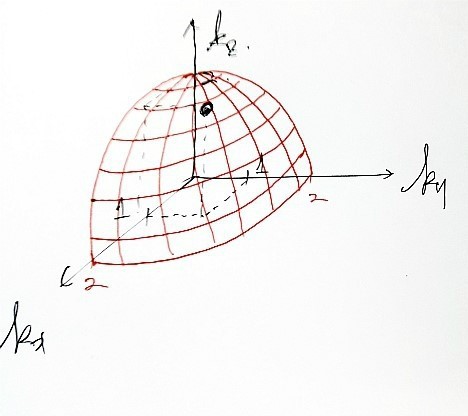
into this red surface area here!!!!!!
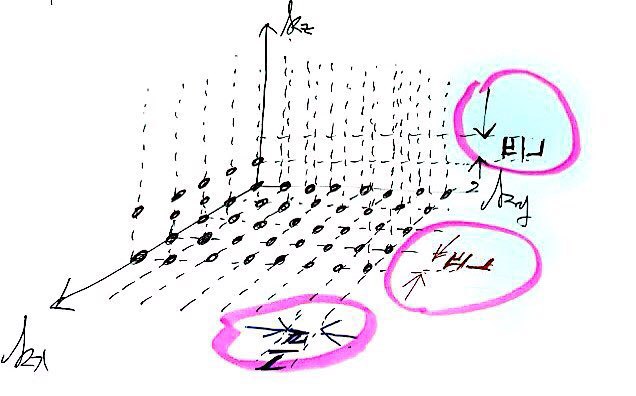
how many bundles of quantum-mechanically allowed states fit?!?!?!?!!
That’s the question we’re going to answer.
Now, then, we’re going to think that those dots representing quantum-mechanically allowed states are inside
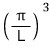
one per each.
If you draw it out this way, it’s fine to think of those dots as having one each per
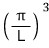
.
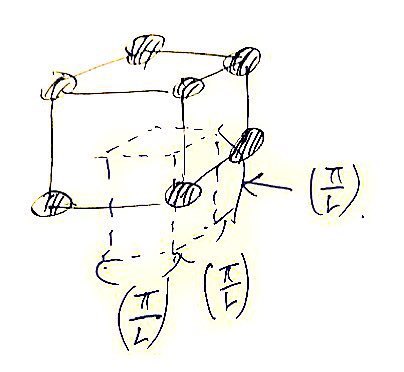
Here a dot is referring to an Energy state,
not an atom!!! The single black dot in the k-space above!! That’s what it is!!!
So in the red surface area,
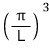
how many fit?!?!?!!!!
Let me derive this equation.
Ah… but, the red surface area is a ‘surface’, which is 2D.
We’re going to multiply by dk and think of it as a volume with a little thickness.
Like this!

Then how do we find the red surface area?!?!
Since in k-space the ‘red surface area’ means the place where the magnitude of vector k is the same,
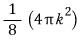
This is the red surface area.
(The 1/8 is because of the condition k>0. The condition k>0 comes from n.)
Alright then now the question!!!
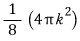
into this,
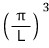
how many bundles fit?!?!?!?!

I expressed L^3 as the volume V!!!hehehehe
That is, when we look at a state with some Energy,
the number of states with that energy is the number of dots that the quantum numbers make in k-space, and that number of dots is
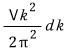
and
we’re going to express this as g(k).
This probably means the density of state,
and if we unpack this again, it means: in the range [k, k+dk], the number of possible states that have some energy!!!!!!
Did you get it?!?!?!!
And let’s go one step further.
What that actually is, if you think simply, is something that pops out when doing a change of variables in integration.
Roughly

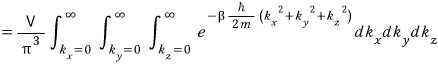
If you write it all out like this, since k_x, k_y, k_z are all real numbers
it’s fine to just write them as x, y, z,
and if we just change the names like this,
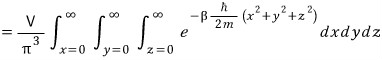
Now how do we solve this integral??? Did everyone feel it?
If we ditch the Cartesian coordinate system and introduce polar coordinates, the integral becomes super easy.
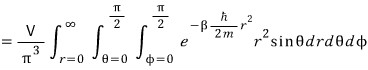
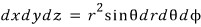
please keep this in mind,
and the r, theta, phi integration ranges come out like that above because we’re only looking at the part where k_x, k_y, k_z > 0.
Now let’s go go with the above integral.

Originally written in Korean on my Naver blog (2016-04). Translated to English for gdpark.blog.