Quantum Concentration and Thermal de Broglie Wavelength
We grind through the ideal gas partition function integral step by step, then dig into what the result actually tells us about quantum concentration and the thermal de Broglie wavelength.
This time I wanna move past density of states and push ahead
The concept we’ll learn this time is simple, but finding its meaning doesn’t seem that simple.
So, first let me write down the discussion we’ve had so far once more.
If you calculate the partition function Z for an ideal gas,
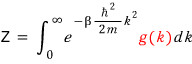
The discussion up to now was figuring out the identity of the red g(k),
and for the ideal gas model g(k) was
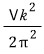
so let’s substitute it in and calculate.

http://gdpresent.blog.me/220584291592

Thermal/Statistical Mechanics I Studied #9. Proofs of integrals frequently used in thermal/statistical mechanics (link only)
I’m not going to derive this! Because it’s easy, so I’ll skip it, and take a derivative with respect to α on both sides of this result equation to…
blog.naver.com
If you refer to this, you can see that we can integrate it,
and I’ll just use it.

So when we compute the partition function,
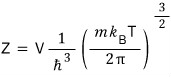
it turns out like this.
The partition function is the sum of all the Boltzmann factors for each state,
and the meaning of the Boltzmann factor is
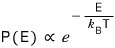
it was the factor for the probability that a particle with energy E is in some microstate.
Wow, but this is interesting,
that such a partition function depends on the volume V, and came out in a proportional relationship.
And
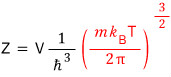
defining the red part as

they say we express Z simply as follows.
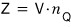
First, let’s check the units.
Z is a sum of exponentials.
So it will have no units.
But, the unit of V on the right side is
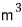
so for the whole thing to be dimensionless, the unit of the quantum concentration must be
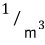
Oh, so n_Q is 1/㎥
It could be interpreted as some kind of ‘density’.
Now now now now now now now now now now what was the meaning of Z
http://gdpresent.blog.me/220689943911
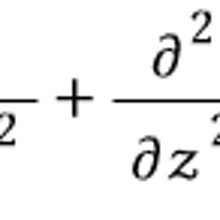
Thermal/Statistical Mechanics I Studied #33. The Partition Function
First, to hammer in the definition of the partition function,the partition function among many many…
blog.naver.com
I said here that Z can be interpreted as ’the size of the probability distribution'.
Then it means that the size of the probability distribution is proportional to V,
which is a bit interesting
But it makes sense.
The bigger the volume, the more situations you can have — this could happen, that could happen — all kinds of situations become many!!!!
So in some sense Z being proportional to V also makes sense.
But, that thing on the side with units of 1/㎥
what could we call it….
Right!
Particle density?!?!?! Would it be okay to call it that????
Since it’s not a solid, it’s a bit awkward to call it density, so I’d like to call it concentration
And the higher the concentration, the bigger Z — this also could be said that the higher the concentration, the more cases there are for this situation, that situation, etc., so the size of the probability distribution grows.
So
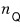
is also called particle concentration, they say. But then why does our book introduce n_Q as quantum concentration?!?!?!!!!??
Let’s see.

Now if we take the reciprocal of this, the unit will be m
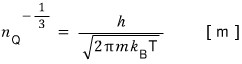
What kind of distance does this represent??????
Since it’s some ‘distance’ pulled out from a concentration of particles,
this is probably the distance between particles.
But here’s the thing. The energy of an ideal gas is one-dimensionally E = (1/2)kT.
No it’s not. Actually E = πkT.
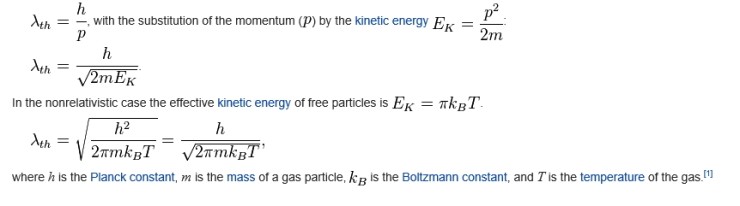
(https://en.wikipedia.org/wiki/Thermal_de_Broglie_wavelength)
So
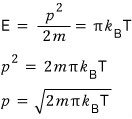
That is, let me write it like this.
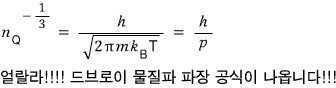
That is,
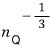
is related to the wavelength of a matter wave, and this

is what they call it.
That is, expressing it in terms of thermal wavelengh,
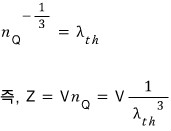
We can express the partition function like this too~
Just a lil more
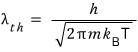
they say this can also be seen as an indicator determining whether we go classical or non-classical.
If the distance between particles is smaller than this, the matter waves of the substances start to overlap
and then quantum effects occur……
So using this as a reference point, they say you can make the choice.
Originally written in Korean on my Naver blog (2016-04). Translated to English for gdpark.blog.