Partition Function for Indistinguishable Particles
Turns out Z_sys = (Z_1)^n only works for distinguishable particles — we break down why indistinguishable ones mess up the math using a simple two-level system.
We’ve been dra~~~~gging the logic along from before
I didn’t emphasize it, but there was a certain premise.
Namely, that there’s only 1 particle
When there’s 1 ideal gas particle, if we define the partition function Z for a system,
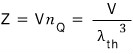
that’s what we had,
From now on, to denote 1 particle, I’ll use subscript 1.
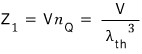
Alright, now let’s bump the count up one at a time
When we have gas a and gas b, the partition function is
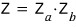
do you remember doing this~
Then now let’s say it’s n of them.
For n ideal gas particles, just like that,
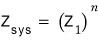
is it?!?!?!?!!!!
The reason I’m writing this post is because, nope hehehe
First, ideal gases, thanks to the ideal gas assumption, aren’t mutually dependent on each other
so
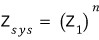
you could think we can say this.
But!!!(but) dats no-no.
First, the conclusion is
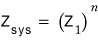
for this to hold, things need to be “distinguishable.“
Let’s look at Example 21.2
and get a feel for distingguishable vs indistinguishable.
We’ll think about it with the simplest two-level system!!!
When there’s 1 particle, the partition function is
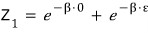
that.
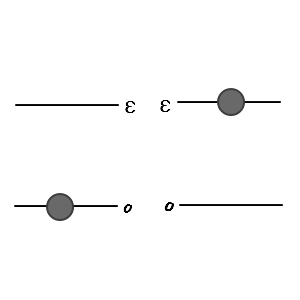
Let’s draw it out for 2 particles
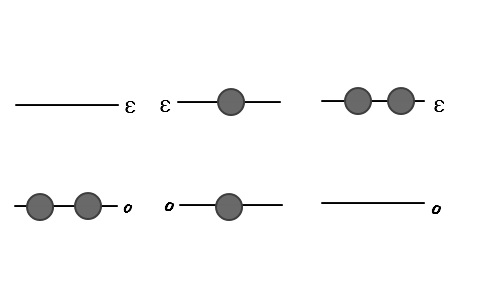
All cases will be limited to something like this.
So the partition function is
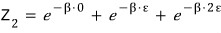
clearly this,
whoa,
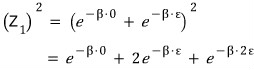
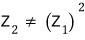
it’s this…..
This is because the “indistinguishable” assumption is baked in here.
Then for “distinguishable,”
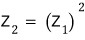
we just need to check whether this holds lolololol
When we say they’re distinguishable, if we draw out every possible case,
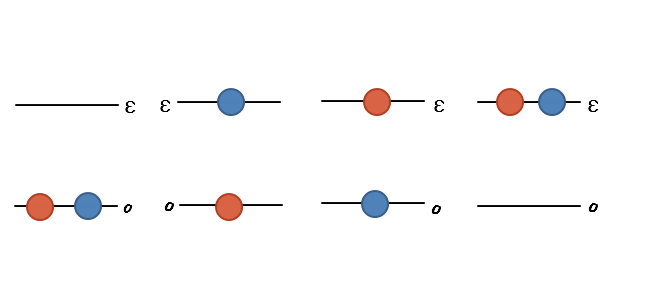
it becomes this, and if we compute the partition function by summing
all the Boltzmann factors over all these cases,
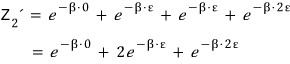
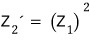
we can see this~.
But, is there anyone on earth who can distinguish gases?
(I don’t mean distinguishing different types of gases — I mean, within the same Ar,
this Ar, that Ar, and 2 seconds later being able to say this was that Ar from before and that was this Ar — can anyone do that?)
Anyone who can distinguish, I’m gonna call them
“Maxwell’s demon!” lololololololololololol
Alright, so humans can’t distinguish.
We can’t distinguish — so now what are we supposed to do
Humanity, come hell or high water, wants to
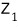
use this
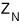
to figure out this
urgh
But if we just keep using
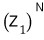
we’ve got duplicate counting,…..
Alright alright alright alright, so what people came up with was
first, just use
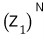
and freely overcount the number of cases
and then divide by N! to resolve the double-counting — that’s the methodology they proposed.
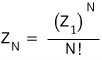
If we divide by N! like this, can we really resolve double-counting?!?!?!!!!
Yeseseseseeseses, we can resolve the double-counting.
But, we need a certain assumption
What assumption do we need — when things are double-counted exactly N! times each!
at that point, we bundle N! into a group, and count that one group as ‘one.’……
What this means — let’s try to understand it at the level of high school probability and statistics.
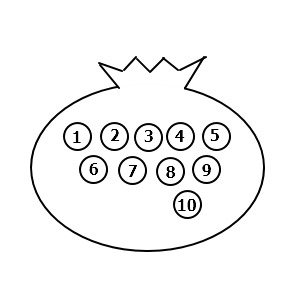
There’s a pouch with balls numbered 1~10 inside it, and let’s think about the number of cases when drawing balls.
How many should we draw?
Let’s try drawing like 3
Drawing 3 one at a time, what’s the number of cases where 1, 2, 3 come out
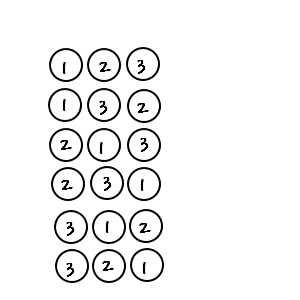
But here, if we say the order of drawing doesn’t matter, what happens
Back in high school days we did this
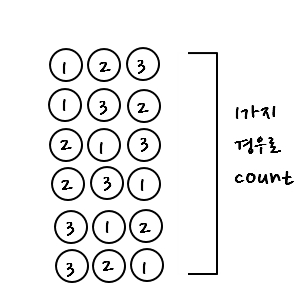
Counting those as one
we could mathematically express this by dividing the total number of cases by 3!!!!
(So we erase the numbers. Erasing the numbers means saying it’s the same as 3 being indistinguishable.)
The reason we can do this?
Among the 3, every single one was different.
And since we’re saying they’re indistinguishable, the 3 all become the same case
and dividing by 3! follows the same logical process as above.
It’s the same context for our partition function of a system with N gas particles,
so going back to what we wrote above
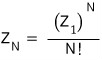
for this to hold, the assumption we need is
“there should be no states occupying two or more of the same energy level.“
That is, bringing this back to our situation
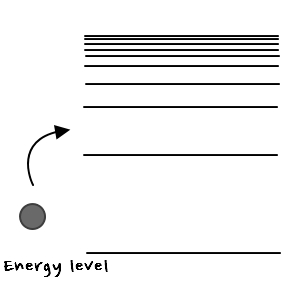
Think of what went into level-1 as a ball with the number 1 on it
think of what went into level-2 as a ball with the number 2 on it
think of what went into level-n as a ball with the number n on it
and then just whoosh-draw them, erase the names off,
and by thinking of them as indistinguishable we divide by N!.
Only then does the double-counting number become N!
and in the sense of counting the amount that was double-counted as 1,
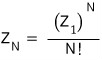
this holds up.
Restating the assumption statistical-mechanically,
we’re assuming “(for an ideal gas) the number of thermally accessible energy levels is much much larger than the number of gas molecules.” Got it!!!!
In other words, the number of particles
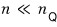
needs to satisfy this
(You could also say it’s the regime where particles’ matter waves don’t overlap and quantum effects don’t kick in!)
Then, based on the assumption above, the partition function for N ideal gas particles is
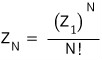
let’s say this, and continue the discussion.
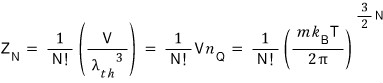
Alright, so we’ve written down the partition function!!!
Once we’ve got the partition function, what could we do
We could express a~ll the state functions in terms of the partition function.
I’m gonna use this to re-derive U, C, F, S, p once more!!!!! Let’s do it!
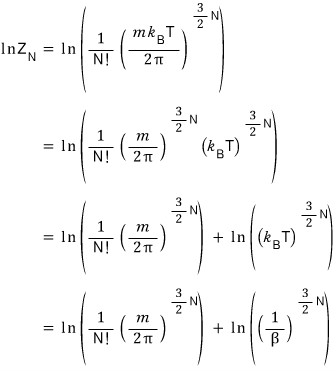
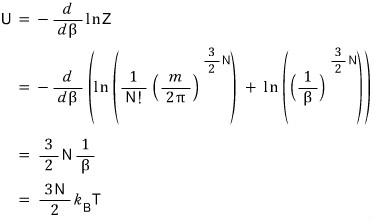
Whoa freakin sweet!!!
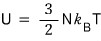
it comes out!!!!
http://gdpresent.blog.me/220584756334

My study notes on thermal/statistical mechanics #12. Chapter 6. Let’s solve some problems!!!!(Problem)
Ex 6.2 What’s the volume that 1 mol of ideal gas occupies at STP(standard temperature & pressure)? Ex 6.3 Pressure…
blog.naver.com
The formula that came out while solving problems has been derived from the partition function now!!!
Alright, let’s keep going forward
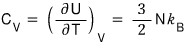
this comes out too~
Moremoremoremoremoremoremoremore
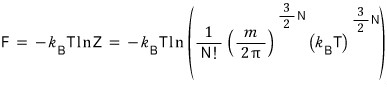
Why do we need this F that looks so messy?
With this, counting entropy becomes drastically easier.
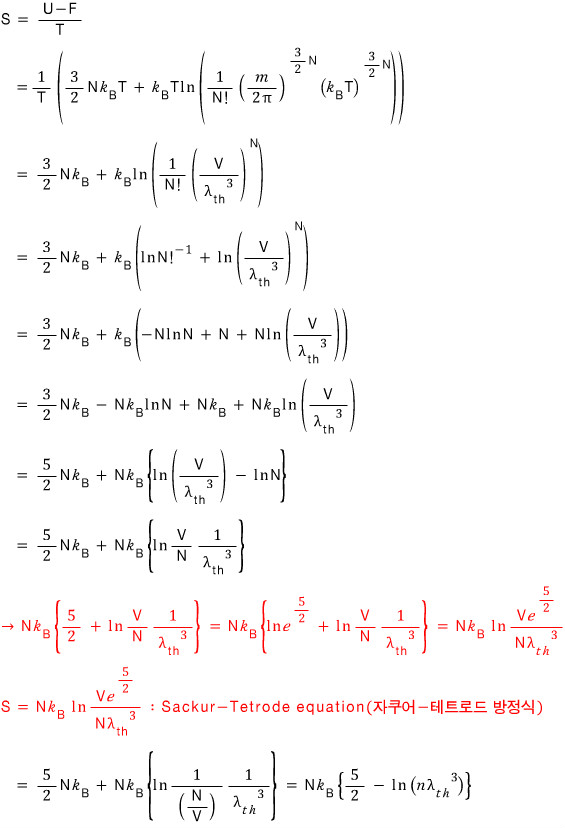
Entropy comes out like the following
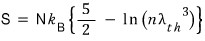
The Sackur-Tetrode equation (jakueo-teteurodeu bangjeongsik) in the middle there — what is that
It’s needed in the next section, and since we can derive it right now, I went ahead and did it in advance
Anyway, S is settled, and now let’s settle pressure p.
As for how I’ll derive p,
using the Maxwell relation
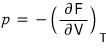
I’ll use this to derive it.
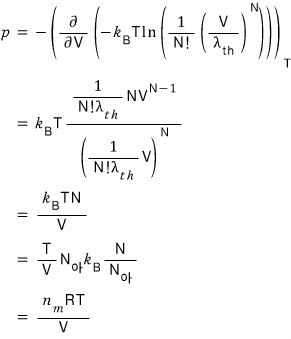
pV = nRT — the ideal gas equation of state gets derived!!!~~~~
Just a little bit left
Just hang in there a little more.
Enthalpy (H) go go go
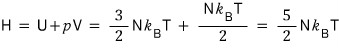
Gibbs (G) go go go
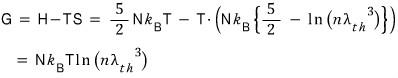
I wish Naver would just improve the equation editor already
dammit… this is seriously rough….
Originally written in Korean on my Naver blog (2016-04). Translated to English for gdpark.blog.