Chemical Potential as Gibbs Free Energy per Particle
We dig into what happens to Gibbs free energy and entropy once we let particles flow in and out, and see why chemical potential turns out to be G per particle!
So this time, let’s study the Gibbs function when we allow particle inflow and outflow
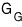
!!!!
It doesn’t seem too hard
First, the ‘First Law of Thermodynamics’, which states the law of conservation of energy when we don’t allow particle inflow and outflow, was written like this.
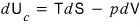
But actually, what we need right now is entropy
(Why we need entropy right now, you’ll find out by the end.)
So if we write it as an equation for entropy
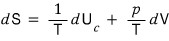
writing it this way, S was described as a function of U and V
that is, we described it as S = S(U,V).
But now that we’re allowing particle inflow and outflow, when we discuss ’energy conservation’, we have to also think about the energy that changes according to particles coming in and going out.
So
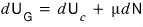
we wrote it like this.
If we also write this as an equation for dS
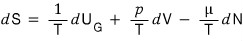
That is, in a system that allows particle inflow and outflow, the variables that determine dS
can be viewed as these 3: U_G, V, N
therefore S = S(U_G, V, N) can be called this kind of three-variable function.
Now what we need to think about here is that if those 3 variables hanging on S
all~~~ get multiplied by k equally, the overall function value S also gets multiplied by k.
In the book, since all 4 variables S, U, V, N have the characteristic of being extensive variables
the biggest characteristic of ’extensive variables’ is?!?!?!?! the biggest characteristic was that they follow linear addition when summed, right???
Since all the variables here are extensive variables characterized by linear addition
they say there’s a characteristic that if all the variables (or state functions) get multiplied by k, the function value S also gets multiplied by k!!!! hehehe
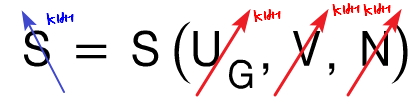
I’d like to briefly go over why this principle is valid.
(Just to confirm, what we’re going to check below is whether S really does get multiplied by k when U,V,N get multiplied by k!!!!)
First, Gibbs’ expression of Entropy was
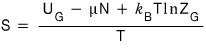
this.
Let’s use
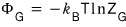
.
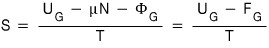
This equation says something.
What it says is,

So first, it’s been confirmed that if U,N,Φ get multiplied by k, S gets multiplied by k.
And it’s been confirmed that if U,F get multiplied by k, S gets multiplied by k.
Okay okay okay okay, and let’s think further

To summarize, S getting multiplied by k — if U,N,Φ get multiplied by k, S can get multiplied by k
and for U,N,Φ to get multiplied by k, if F and V also get multiplied by k, those 3 can also get multiplied by k
ahhhhh so, if we pull it up from the bottom aaaaaall the way and lump it together
we can say that if U,N,F,V get multiplied by k, S gets multiplied by k!!!!
There seems to be a tiny gap in the logic.
So I’ll say it like this — when U, V, N, F get multiplied by k~~.
So what can we milk out of this
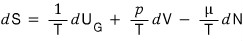
in this equation, let’s say U,V,N,F got multiplied by k.
Then S would also get multiplied by k, right?!?!!!
That is, we were in the state of U,V,N, and became the state of kU, kV, kN
Then each differential would be
dU = kU-U
dV = kV - V
dN = kN-N
Then since we said S also gets multiplied by k
dS = kS - S — I’m going to substitute this kind of thing!!!!
Then what happens
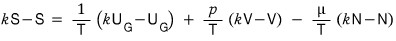
Since this k has generality that holds for all k, you can plug in any number,
let’s plug 2 into k
k=2 substitution, gogogo
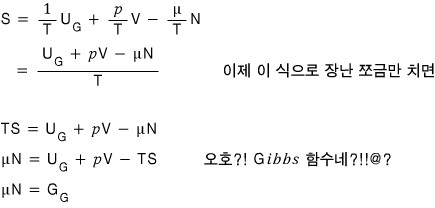
Getting half-swindled somewhat,
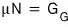
we derived the relation,
and I think figuring out this equation has great meaning.
Because
until now we only knew about the ‘amounts of change’ like dF, dG, but we knew nothing about the state functions themselves.
Alright and for reference,
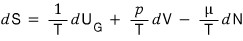
from this equation, if you just whoosh~~~ drop the d’s
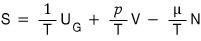
it ended up looking like we wrote that this kind of equation holds,
but this is something we need to think of as “happen to be”!!!
Not mathematically obvious, but literally happen to be….
If I had to squeeze out a reason, it’s because coincidentally all the variables were ’extensive variables’?!?!
Alright and also
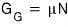
apparently can be interpreted differently
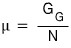
writing it like this, we can say this
“chemical potential: μ is the ‘Gibbs function per particle’.” like this!!!! hehehe
And one more more more more more more thing
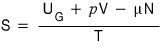
we just derived that we can write this,
and what we already knew was
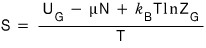
this?!?!?!
So
if we use
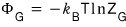
we can organize all the way down to
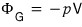
. lollollollol
Originally written in Korean on my Naver blog (2016-06). Translated to English for gdpark.blog.