Chemical Potential of Photons
Turns out when a system doesn't conserve particle number — like photons — the chemical potential just goes to zero at equilibrium, and that's actually a huge deal!!
This time I want to talk a bit about photons.
So, what we can figure out while thinking about photons is
After learning the Grand canonical ensemble,
we’ve now studied the case where the number of particles in a system can change.
And while doing that, we called it a system that allows the inflow and outflow of particles, and we tossed up a picture like this
But now it’s time to think about this.
“Does the number of particles HAVE to change by particles flowing in and out??!?!?“
Rightttt..heh.heh.hehheh the number of particles can just change inside there on its own!!!~~
A representative example is that photons can do that sort of thing, so it’s worth discussing here.
And to give a slight teaser of the posting, the next thing we’re covering is photons hehehehe heheheheh
Alrightalrightalrightalrightalrightalrightalright let’s first think about the Helmholtz function
The Helmholtz function means the free energy of a “system when volume and temperature (TV) are constant"
and the natural world moves in the direction that minimizes the Helmholtz function~~~
This is something we already know.
But, in our current system, the Helmholtz function will be made to vary so as to minimize the Helmholtz function by the number N changing.
That is,
at equilibrium
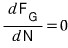
will be satisfied!!?!?!
Agghhh!!!! Datsunononononono!!!
That’s a bit lacking
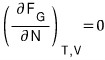
Phew, now I feel a bit at ease heh heh heh
Uwugh?!?!?!?!!!!! Wait?!?!?!?!? I’ve seen this somewhere before?!?!!!
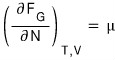
Remember?!?!?!
Well then!?!?!?!!!
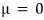
Alrightalrightalrightalright let’s think again.
We assumed that the number of particles can change,
and having set things up that way, we’ve arrived at the conclusion that “at equilibrium” the chemical potential becomes 0
In other words, if a system doesn’t obey number conservation!!!
the number adjusts itself so that the Chemical potential at equilibrium becomes 0!!!
Since the most representative example that shows this best is the Photon,
I’ll now toss up an equation you’ve seen many places.
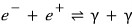
e- : electron
e+ : positron
γ : photon
Two beams of light combining produce an electron and a positron.
(The positron is the antiparticle of the electron, with the same mass, charge magnitude, and all that, except the charge is opposite…..it’s the electron that plays the role of the electron in the Digimon world.)
This kind of reaction is the reaction of photons,
and in this kind of reaction, as in the picture drawn wayyyyy up above, the number of particles will not be conserved.
But but but in the case of the photon, at least the charge will be conserved
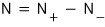
let’s say it’s conserved as this!~~~
Now, still in a situation where T, V are constant, we view the Helmholtz function as being determined by the numbers N+, N-.
That is, we’re going to view the Helmholtz function as a two-variable function of N+ and N-.
Therefore this partial differential equation holds.
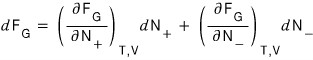
And since we already know that at equilibrium the Helmholtz function is a minimum, let’s look for dF = 0
That is, at equilibrium this equation will hold.
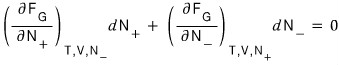
Then now I’ll manipulate it like this
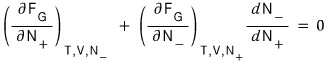
The reason I wrote it like this is because the electron and positron react 1:1!!!!!!
Then
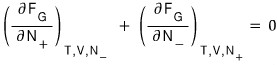
Now, since each of these refers to the chemical potential of the electron and the positron respectively,
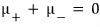
We’ve obtained the equilibrium equation for the case of the photon!!!!
Then what we need to know further is,
just what state function is the chemical potential a function of?!?!?!
One thing that’s clear is that we could get the hint that it’s not a constant, so
What!!! Just what state function are you hooked on!!!!
That I’ll continue in the next posting.
Originally written in Korean on my Naver blog (2016-06). Translated to English for gdpark.blog.