Pressure Dependence of Chemical Potential
Wrapping up chemical potential once and for all by rewriting μ in terms of pressure using the ideal-gas relation, and linking it to the Gibbs function per particle.
As I previewed in the previous post, this time I’m going to finish off the chemical potential once and for all.
I’ll check what other state functions the chemical potential depends on.
So first, let me bring out the “chemical potential of an ideal gas.”
http://gdpresent.blog.me/220740093268
My study of thermo & statistical mechanics #43. The chemical potential of an ideal gas…
This post first sorts out the example “chemical potential of an ideal gas,” and then continues the next discussion…
blog.naver.com
I derived it here.
The chemical potential of an ideal gas was
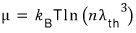
.
Actually that was the whole thing.
You can see it depends on n and T.
But n can also be expressed in terms of other variables.
In the ideal-gas case, n can be replaced using this relation
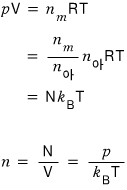
So
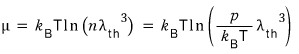
where
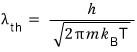
you haven’t forgotten the thermal wavelength formula, right
Now, since the form we’re going to use from here on isn’t this,
I’ll tidy it up just a bit more.
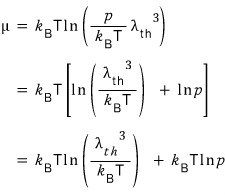
This is how I’ve checked the pressure p dependence of the chemical potential.
And having done that, let’s think like this.
“The chemical potential is the Gibbs function per particle!”
And the Gibbs function is a function of p and V?!?!?!
Oho, so that mu over there also has to be thought of as a function of p, V
niiice
So
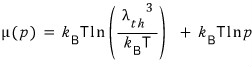
Once I write it like this, the front term is a constant!!! There’s not a single variable attached to it~
So I’m going to rewrite it in the sense of a constant
take a good look~
Let’s find the μ value when the pressure p=1.
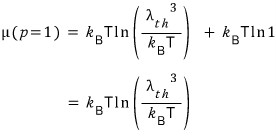
Aha, the front term was the value meaning the chemical potential when the pressure is 1.
I’m going to denote the chemical potential when the pressure is 1 as
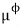
and I’ll write the chemical potential formula like this~
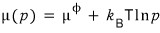
ref- to the -erence,
later on
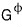
don’t be surprised if it shows up, it just means the Gibbs function when the pressure is 1 hehehe
Okay, so for what I organized above,
I’ll go through just one more step
I’ll subtract zero from the right side of the above equation~
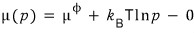
I’ll write zero differently.
Now I’ll write zero differently
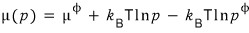
Please think of p-phi as “1 atm”~
So we can write it like this.
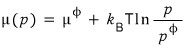
Of course, treating “k-B-T” as a gas constant, the above equation could also be written this way
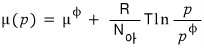
What I think one must be careful about when looking at the above equation
is not to forget that this isn’t the essence of the chemical potential.
Because right now we have the super-megaton assumption that it’s an ideal gas, I think that’s why the chemical potential comes out depending only on p.
If we didn’t have the super-megaton relation pV = nRT, I don’t think such a simple form would have come out.
Now, this equation is also a relation that can be pulled out from the Gibbs function.
Let me derive the above equation using G as well~
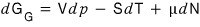
Let’s say T, N are constant (just as we looked at μ’s pressure dependence above, here think of it as looking at G’s pressure dependence~)
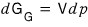
And if we express V differently with our super-megaton assumption
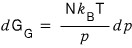
Smack an integral on both sides,
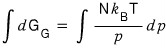
For the integral on the right side, let’s smack the integral from p-phi to p.
Then, as for what interval the left side should be integrated over,
we can smack the integral from G at pressure p-phi to G at pressure p.
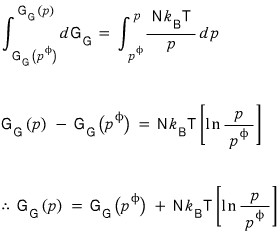
I told you the chemical potential is the same as the Gibbs function per particle, right???
If we divide both sides equally by N~
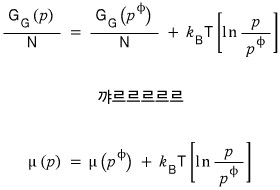
Confirmed, right!!? hehehe
From here on I’m going to talk a bit about chemistry,
ah,…….. if I write any more after this it feels like it’ll get too long, so I’ll cut it off here for a moment!!!!!
okokok let’s meet in the next post. lollll
Originally written in Korean on my Naver blog (2016-06). Translated to English for gdpark.blog.