Le Chatelier's Principle
A casual deep-dive into Le Chatelier's Principle through the lens of Gibbs free energy, explaining why reactions reach dynamic equilibrium and how the system responds to disturbances.
Reaction
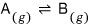
Let’s say we have something like this.
Well, if it was a state where only gas A existed,
then after some amount of time, there would be some amount of A still left, and some amount of B would have been produced.
That’s how it would look to our eyes.
The reason I said it like this and even underlined it is because it only looks that way to our eyes, and in reality it’s not so.
It only appears to be stopped, but on a microscopic scale the A→B and A←B reactions are continuously proceeding.
But the ’equilibrium’ that looks stopped to our eyes
that is, I was speaking of the concept of “dynamic equilibrium”.
And today I want to talk about that kind of thing a bit.
Hey dammit, just how much does it react and how much doesn’t react and remains?!?!?!
Well, since in the previous posting we dealt with the Gibbs function, we’ll focus on the Gibbs function.
(Or rather, looking at the Gibbs function is looking at the most general case.
When p,V are constant, the free energy has the meaning of the Gibbs function, and since in ordinary chemical experiments p,V are constant, that’s whyyy)
“Until when does a reaction continue and reach (dynamic) equilibrium?!?!”
The reaction will continue until the Gibbs function reaches its lowest state, and then it will reach equilibrium.
That is, at equilibrium, the value of the function G will not change from its minimum value, and dG will be 0.
Well, that’s the basic concept up to here, and let’s describe this basic concept using the tools we’ve learned before.
For now let’s say the reaction is 1:1
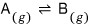
When one A disappears, one B is created, and conversely, when one B disappears, one A is created.
So,
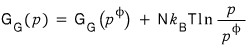
we had organized it like this,
when one A disappears, by how much does the Gibbs function change!??!?!?
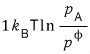
it will change by this much,….
And so when one B is created, the Gibbs function will thereby change by
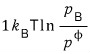
this much, see~
That is, the total change in the Gibbs function is
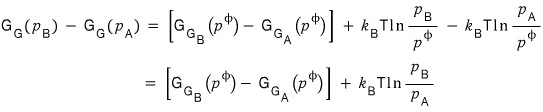
(Note. The forward reaction is the standard. That’s why I wrote the equation as A disappearing and B being created.~~)
However, at equilibrium the total change in the Gibbs function will be zero, so
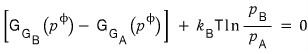
this would be satisfied.
Oh right.
Here, p_A, p_B are the ‘partial pressures’ of each gas.
Oho!! Look at that equation!!!! The ratio of partial pressures!!!!
This is why in chemistry the equilibrium constant K is defined as the ratio of each partial pressure.
To restate it cleanly,
“ratio of partial pressures at equilibrium = equilibrium constant K” is what it’s saying!!!
And the partial pressure ratio K at equilibrium would refer to this sort of thing:
“What has survived in greater quantities at equilibrium?!?!?!??~~~”
That is,
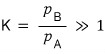
if this is the case, it means that B has survived in overwhelming quantities at the equilibrium point,
and that means that the A→B direction, the forward reaction, is the more dominant one.
Conversely,
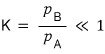
if this is the case, it means that A has survived in overwhelming quantities at the equilibrium point,
and that means that the A←B direction, the reverse reaction, is the more dominant one, right?!?!
That is, the equilibrium constant K is a number that indicates which of the forward and reverse directions the reaction is more dominant in~~
Alright then, let’s generalize one step further.
Just now it was
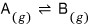
but what a step further generalization will look at is,
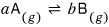
let’s think about a case where when a molecules of gas A disappear, b molecules of gas B are created~
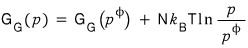
The change in the Gibbs function for each is

At equilibrium, the change in the Gibbs function will be 0, so
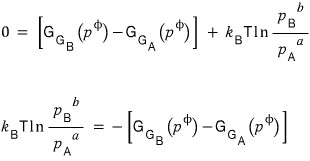
With this, we can also see why the ratio of numbers enters the partial pressure ratio of the equilibrium constant as a power.
Then
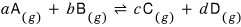
in this reaction, why the equilibrium constant K is
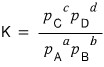
defined like this, you now understand!?!?!?!!
It seems we’re about to leave the realm of physics.
Let’s move on without going into a detailed discussion!!! hehe
Now I’m going to pull some BS to describe the same story using the gas constant.
The reason this BS is meaningful is something you’ll understand if you go all the way to the end lololol
First
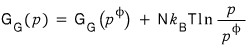
this equation
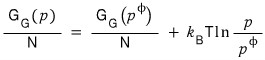
I’ll write it like this and proceed with the discussion.
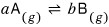
I’ll apply it to this reaction.
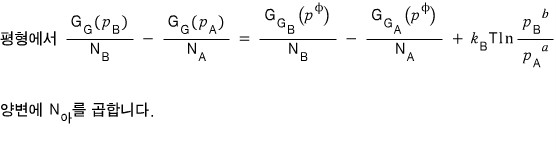
Writing it like this, we can say that the red guy and the blue guy are each as follows.
Red: difference in total Gibbs function per mole
Blue: difference in total Gibbs function per mole at 1 atm
So now let’s notate it like this

Let’s write it like this.
And thinking about equilibrium!!!
At equilibrium there will be no difference in the potential between the two sides (that’s why the reaction has stopped)
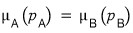
is how it’ll be, y’know.
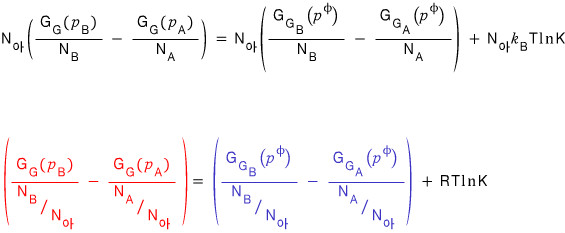
So
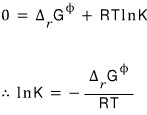
Now I’m going in for some more shenanigans with this equation using the “Gibbs-Helmholtz equation”.
The Gibbs-Helmholtz equation was
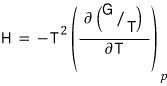
this.
Since we’re going to use this, I’ll hit both sides of the above equation with a derivative with respect to T.
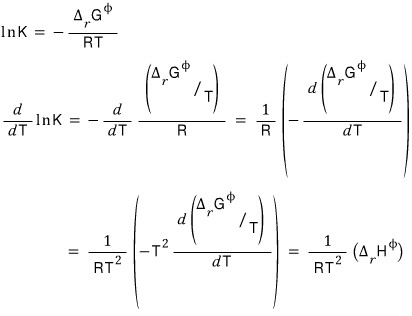
By playing tricks on our equation with the Gibbs-Helmholtz equation, we can pull out an equation in terms of “enthalpy”,
and the basic of the state function called ’enthalpy’ is ΔH = ΔQ, so
ΔH >0 indicates an endothermic reaction,
ΔH <0 indicates an exothermic reaction — that was the basic concept of enthalpy!!
Alright then, let’s organize.
Under the condition of equilibrium
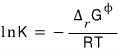
we were able to derive this equation,
and that equation
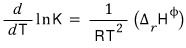
we could see that it can transform into this equation.
Therefore both equations are equations for “equilibrium”.
In other words, when not at equilibrium,
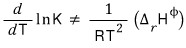
that’s what it would mean.
And so what this formula means is said to be “Le Chatelier’s principle”,
and I’m going to tell you the reason.
A system that was in balance —
so,
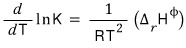
a system that was like this —
suppose we broke the equilibrium by raising the temperature.
So
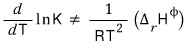
it became this, and this system will do some reacting.
If that reaction is an exothermic reaction, it means heat goes out, and heat will be generated in the surroundings.
Heat going out means ΔG< 0,
that is, ΔH < 0.
So

And this in turn means that the forward reaction in the A→B direction will appear to not proceed as well as before raising the temperature!
And the reverse reaction will appear to proceed better.
Conversely, by raising the temperature and breaking the equilibrium
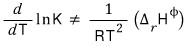
it becomes this, and now it reacts until they become equal,
and if that reaction is an endothermic reaction,
an endothermic reaction means heat is coming in, and this becomes ΔH>0.
So since the red side became positive, dlnK must be positive.
That in turn means the partial pressure of the product becomes larger,
which means that equilibrium is reached in a state where the number of particles of the product is greater than in the initial equilibrium state.
And this in turn means that the forward reaction in the A→B direction appears to proceed better upon raising the temperature,
and conversely the reverse reaction is observed as not proceeding as well……
Le Chatelier’s principle….. lololol
Originally written in Korean on my Naver blog (2016-06). Translated to English for gdpark.blog.