The Stefan-Boltzmann Law
Turns out everything — even you — emits electromagnetic waves, and the Stefan-Boltzmann law basically explains why Super Saiyans glow different colors lol.
Any object whose temperature is not zero emits electromagnetic waves.
A person’s temperature is about 36.5℃, right?
That’s not 0K. So that means a person emits electromagnetic waves too
Huh?!?!?!??!?!?lollollollollolwhat kinda Super Saiyan is this??lollollollollol A person emitting light????
That’s right. People emit electromagnetic waves too. It’s just that humans can’t perceive it — machines can.
What kind of machine perceives it??? The “infrared camera” perceives it.
That is, people emit electromagnetic waves in the infrared range that humans cannot perceive.
Not only people, but many objects also emit light. (Since objects are, one way or another, made of atoms, I’m not writing a novel here.)
When you look at the world through an infrared camera, it’s not like only people are visible, right?
You can see everything, everything
That means everything you see emits light in the infrared range
Then isn’t there something that emits light in the visible range that our eyes can see?!?!?!
There is!!! You can see it if you go to POSCO!!!!
Irons heated freakin’ hot emit light.
An extremely hot iron emits red light,
and an f-ing hot iron emits yellow light with higher energy lol
Then ※Warning: For Fun※

The fact that a Super Saiyan emits yellow light
means they possess that much higher energy lol lol lol lol lol lol
The cartoonist who drew people emitting yellow or blue light for no reason probably graduated from the physics department lol lol lol lol lol
Then the Super Saiyan lol lol lol lol lol lol lol lol lol lol lol lol how massive an energy do they contain lol lol lol lol lol lol lol lol lol lol
If you get hit by an energy wave you’re a goner….. yeah, that’s right… lol lol lol lol lol lol lol
We can even rank them
Kaio-ken emitting red light < Super Saiyan emitting yellow light < Super Saiyan emitting blue light
We can rank them like this.
Because light
has energy in the order red → orange → yellow → green → blue → indigo → violet.
So
infrared (赤外) | red → orange → yellow → green → blue → indigo → violet | ultraviolet (紫外)
If you get hit with UV, you even get skin cancer as much as you got hit with high-energy light,
and infrared is used a lot for treatment purposes, right?????
What has higher energy than UV is X-ray.
That one just pierces straight through the body lol lol lol lol lol lol lol lol lol lol
What’s bigger than X-ray is γ-ray
Gamma rays are representatively the light emitted when a nuclear bomb detonates,
and if you get hit by this, apparently all your skin tissue just dissociates lol lol lol lol I have no experience since I’ve never been hit,
but getting hit means you just die right away^^hehehehehe
Anyway lol lol lol lol lol lol lol lol Dragon Ball~~~ oh ho~~~~~lol lol lol lol lol lol lol lol lol
This chapter is the chapter that discusses this “thermal radiation”!
But there’s a bit of a discrepancy,
in the late 19C ~ early 20C as this thermal radiation was being researched, Planck’s constant was created,
and as the meaning of that Planck’s constant was revealed through the photoelectric effect, the boundary between particle and wave broke down, and we obtained a new perspective that electromagnetic waves can be viewed from the standpoint of photons
(all of these are talking about quantization (quantumize) and became the birth of quantum mechanics)
But this chapter will not discuss thermal radiation from the perspective of late 19C scientists,
but rather view thermal radiation from the perspective of 20C quantum mechanics.
So, originally, viewing thermal radiation from classical mechanics is also correct in the order of history,
but rather than viewing it that ‘wrongly’, we’ll just flat-out view it ‘correctly’.
The reason??? Because it’s easy lol lol lol lol
If you view it from classical mechanics, it might trigger a hair-loss emergency
So, we view it from the modern physics perspective. Then let’s goooooooooo
First, in this posting
I’ll be organizing the Stefan-Boltzmann law.
At least this one can be derived from classical thermodynamics. It’s easy
How was this law obtained? —
you paint the inside of a box completely black, drill a tiny hole, and put light in.
And then you measure the temperature generated on the surface of the box…. something like that,
it’s just research on Black-body radiation….
intensity : I
(intensity, strength, energy emitted per unit surface & per unit time)
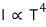
We found out that it’s proportional to the 4th power of the temperature like this
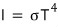
and we made an equation like this~~~~
Since we’ll derive everything including the constant in front here, don’t worry too much
So first, let’s derive that the intensity (intensity, strength, energy emitted per unit surface & per unit time) is proportional to the 4th power of temperature.
This can be simply derived from the 1st law of thermodynamics.

The meaning of the left side is “when the volume changes by this~much, how much does the energy change??”
That is, it refers to the internal energy density.
You can also call it energy density u
Gooood
then
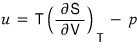
And the entropy differentiated by V in the middle, that one,
I’ll replace it with another form using Maxwell’s relations.
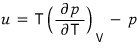
Oho it’s all ~ changed to p
Now I’ll express p as an equation in terms of u, and there’s a way.
Where did we do this before? — I organized it here http://gdpresent.blog.me/220584695823
Thermal & Statistical Mechanics As I Studied It #11. Ideal gas equation of state, pressure
Now we’re going to enter ‘Chapter 6. Pressure’. Rather than saying we’re learning about pressure, it’s just PV=nRT ideal gas equation of state…
blog.naver.com
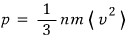
I organized it like this, but what we’re dealing with now
is filled with photon gases, and the speed of a photon is unfailingly always c.
Therefore
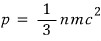
and yeah
mc squared, you see energy, right?!?!?!
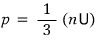
And when I bundle it like this, it’s the energy density~~~
(since it’s energy per unit times number density, you can call it energy density.)
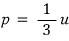
Ahh so for photons, energy density had this kind of relationship with pressure!!!
(For other gases whose speed isn’t c, it was 2/3….. just so you know)
Anyway the above equation is
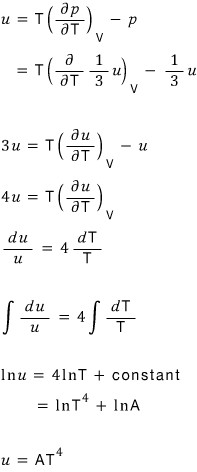
Right now we’re
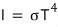
heading toward?!?!?! But
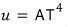
got derived.
It’s not energy per unit volume that we need, but ‘per unit area, per unit time’ that we need.
Alright then, Operation Convert u to I begins.

Let’s suppose photons are filled inside some cavity.
I’ll think about the flux of the “photon gas”. In the ideal gas case, at temperature T, the flux Φ was
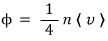
derived like this
Should I link the one I derived too… http://gdpresent.blog.me/220584775364 I derived it here.
But now since it’s filled with photon gas, the flux will be like this.
Whether the temperature is T or T’, because the photon’s speed is without exception c
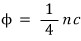
The energy of the above single flux, since the photon energy is
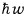
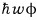
that’s what it is. (By Einstein’s photoelectric effect experiment, it was revealed that the photon energy has that kind of relationship with Planck’s constant)
Then that much would be the Intensity right
That is,

Aha then
Into the above intensity equation
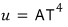
let’s plug this in.
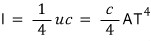
Alright alright alright so the Stefan-Boltzmann constant σ was cA over four!!!!!
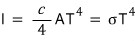
Like this we obtained the Stefan-Boltzmann law!!!!!
But then the Stefan-Boltzmann constant
that that sigma, how on earth are we supposed to obtain it!?!??!?!!!!!!!!!!
I’ll pick that up right in the next posting.lol lol lol lol
Originally written in Korean on my Naver blog (2016-06). Translated to English for gdpark.blog.