Ultrarelativistic Gas
A casual walkthrough of relativistic effects — γ, momentum, energy — and how they reshape the math when gas particles scream close to the speed of light.
A title that suggests we’re now going to take relativistic effects into account when looking at gases.
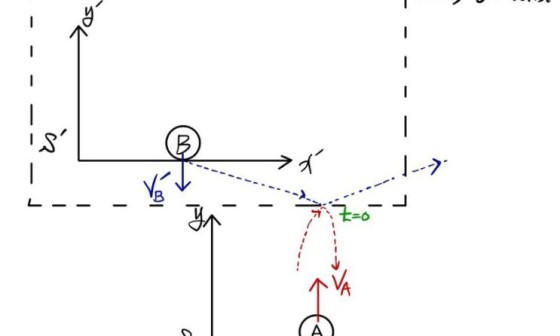
A relativistic “effect” means that as velocity v gets closer and closer to the natural-world limit speed c,
something looks different…. or feels different, that kind of effect,
and by “looks different” I mean things like momentum, mass, energy, etc.
p → γp
m → γm
E → γE
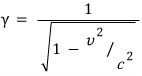
— these look different, that’s what I mean.
For details,
http://gdpresent.blog.me/220457496946
Relativistic mass, relativistic momentum, relativistic energy [ Modern physics I studied #5 ]
To spoil just a tiny bit what we’re going to do here, we’re going to look at “the relative change of mass” whoa!!!!! Mass is…
gdpresent.blog.me
it’s here. (It’s special relativity, based on the Lorentz transformation.)
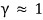
because of this, we were able to say “there’s no relativistic effect,” whereas when velocity v is extremely fast (close to the speed of light)
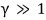
because of this,
considering relativistic effects is the way to avoid errors.
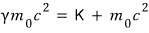
The left-hand side is the “total energy (total).”
So
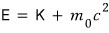
I’ll write it like this.
Then this equation gets derived.
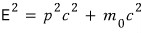
This is also allll in the link I attached above.
Alright, here we go.
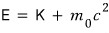
in this,
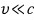
when this is the case,
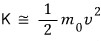
the fact that this gets approximated is also in the link up there.
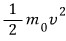
we express
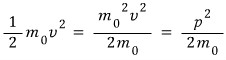
like this and plug it into the general equation above.
Expressing K in that form means we’ve totally ignored relativistic effects, right?!?!!
So
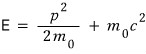
this equation represents the non-relativistic limit!!!
That is,
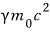
this total energy, as v gets smaller and smaller, converges to
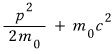
it converges to this!!!
You can derive it with high-school math too~
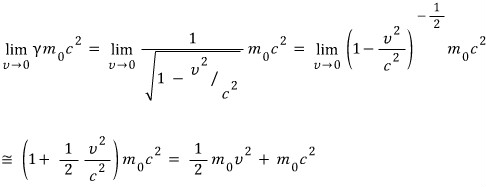
Ah, there’s a Taylor approximation in there so I guess I shouldn’t have said high-school math.. heh.hehe
Alright alright alright, and when the velocity gets super super fast and approaches the ultimate speed, the speed of light,
what we learned about where the energy converges is
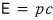
we learned that it converges to this.
That is, just as we called the convergence of E with respect to p the non-relativistic limit,
E=pc, which represents the convergence of E when v is large, is called the ultrarelativistic limit.
<Note: the relationship between p and E is called the dispersion relation, right~~~? hehehehehe>
If we represent these limits on one coordinate axis, it looks like this
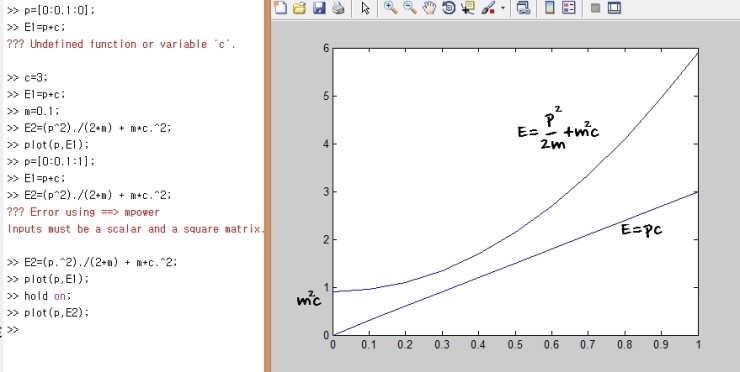
Since I wanted to stop taking photos of textbooks and uploading them, I roughly plugged some numbers into MATLAB and drew it myself,
and each of those limits is there.
So a pair (p,E) outside those lines cannot exist.
Ah, and the actual (p,E) is somewhere around the inside there,
and if p«mc, it’s close to the non-relativistic limit; if p»mc, it’s close to the relativistic limit.
So if we overlay and draw the dispersion relation of a particle with mass m,
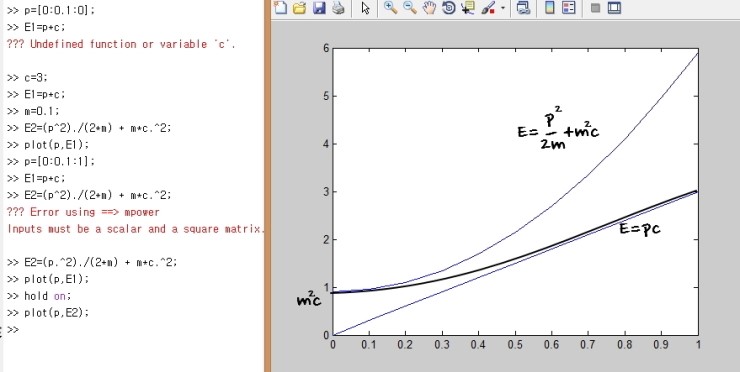
well, that’s what I’m saying.
Actually, up until now I’d been casually taking the non-relativistic limit
and deriving the partition function of the gas, internal energy, and various state functions,
but in this posting, instead of the non-relativistic limit value, I’m going to derive the various state functions using the relativistic limit value.
However, apparently at the undergraduate level the relativistic limit isn’t used much.
But,,, I’m a wimp…..
Still, later on if something happens
you never know how life goes lolololololol so I want to at least organize it before moving on hehehehehe
In case one day I suddenly need to use it????
Oho, so then.
Now we look at the gas.
Clearly the gas here is an ideal gas, so it’s not Fermi-Dirac, not Bose-Einstein — it follows the Boltzmann distribution!!!
Now, since we’re trying to look at the ultrarelativistic limit of the gas,
we latch onto this curiosity:
I think you can understand it as finding the answer to the question “The motion of gas is of course not the speed of light, but if v→c?? Where would it converge??“
So we’re going to use the dispersion relation E=pc,
and we’re going to express this as an equivalent wave using the de Broglie matter-wave formula.
That’s okay, right????
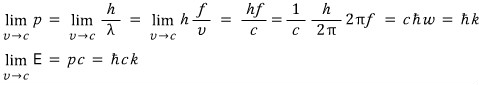
This is the momentum of a photon and the energy of a photon,
but since we’re going to use the Boltzmann distribution below, I didn’t want to use the word “photon.”
So I just derived it like that.
Now I’m going to derive the partition function.
Here g(k) is the density of states, and this was derived from the standing-wave condition,
and it is
this.
http://gdpresent.blog.me/220691412313 (The detailed derivation is here.)
Thermal & statistical mechanics I studied #36. Density of states, statistical mechanics of ideal gas
Let’s take a rough~ look at what we’ve done so far: find Z, compute U and F, take the derivative with respect to T…
blog.naver.com
Ah, and as previewed, I used the Boltzmann distribution.
Now I’ll do the partition-function integral.
Into this,

if we just plug this in well,
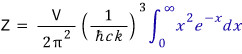
the blue gamma function shows up!!!
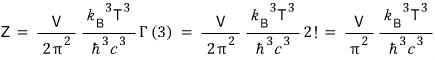
I’m going to write this like this~~~~
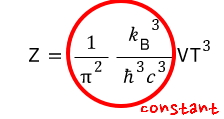
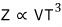
huh?!?!?!!! When relativistic effects weren’t considered,
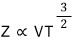
do you remember that it was this?!?
Submitting evidence. http://gdpresent.blog.me/220691598659
Thermal & statistical mechanics I studied #38. Quantum concentration, thermal wavel…
This time, past density of states, I’m going to move on with the curriculum. The concept we’ll learn this time is simple, but its meaning is…
blog.naver.com
Anyway, unlike the non-relativistic case, in the ultrarelativistic case
the partition function of the ideal gas is proportional to VT³ with T squared on top.
Why is that??? I’ll reflect on it later!!!!
Okay so now more to do
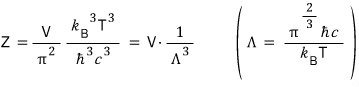
Apparently, you substitute the non-V portion of the partition function with this capital lambda and write it like this.
(I think it’ll be good to cross-reference this with the contents of the linked article attached above. For details please refer to the attached article.)
Having defined the partition function of the ultrarelativistic gas like this,
the ultrarelativistic gas —
not a gas whose speed is close to c, but a gas whose speed is just c — I’ll derive its U, C, F, p, H, S, G, and so on.
First, I’ll assume there are N indistinguishable ultrarelativistic gas particles.
And another necessary assumption is that the number of thermally accessible energy levels is much greater than the number of gas molecules.
Because then

yes~~~~ So we’ve organized the partition function for N gas particles, and now we’ll organize the actual state functions.
I’ll derive U first.
Since we’re going to derive U, I’ll use the 3rd equation among those above, not the 2nd!!!!

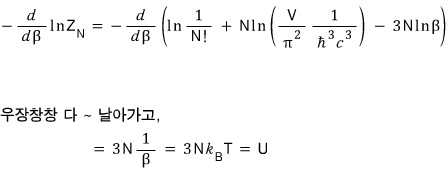
For reference, the internal energy U of a non-relativistic gas was
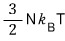
and the internal energy of the ultrarelativistic gas is 2× different from that, as you can see.
Then, now, heat capacity!!!!
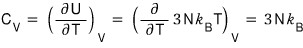
Likewise, in the non-relativistic case the constant-volume heat capacity was (3/2)kT,
a 2× difference from the above.
Then now I’ll derive the Helmholtz function F.
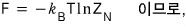
This time I want to express it simply using the 2nd equation from the top, not the 3rd.
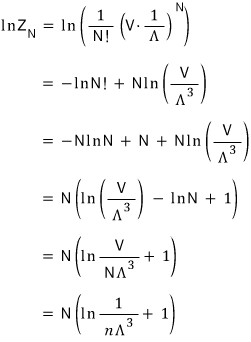
Therefore the Helmholtz function F is
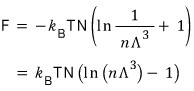
By the Maxwell relations, if you partially differentiate the Helmholtz function F with respect to V and slap on a (-), you get pressure.
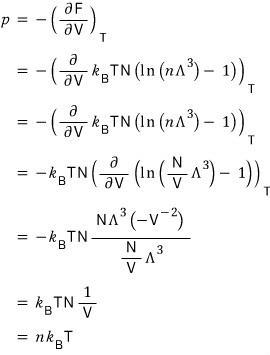
In the non-relativistic model the pressure p was also p=nkT, right?!!??!! Weird, huh?
Even if the gas inside a container of volume V and temperature T is all moving at speed c,
the pressure is p=nkT, the same as when the speed is v rather than the speed of light.
Amaaazing.
Weeeird and wiiild.
From this we can see that pressure does not depend on the variable of speed
and depends only on temperature and number density n.
Now, if we mash up the energy U = 3NkT with the pressure p = nkT,
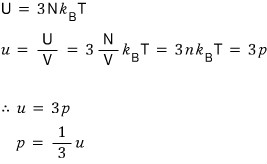
Yet again, in the non-relativistic case p was (3/2)u!!!!~~~
(When the temperature is the same, p is the same but there’s a difference in the energy??? You could say it like that hehe)
And the reason the energy density differs by 2× between the non-relativistic and ultrarelativistic cases —
mc² vs. one-half-mv²
you can think of it as that kind of connection!!!!!
Quick quick, let’s also find the entropy S!!!!
As we’ve used a lot before,
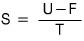
I’ll use this.
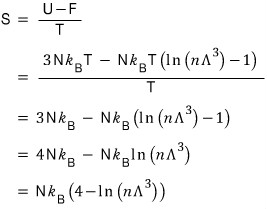
I’ll also derive the enthalpy H!
I’ll derive H as U + pV~
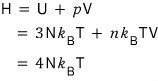
The Gibbs function G is what’s left.
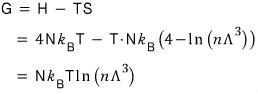
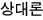
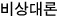
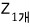
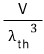
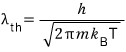
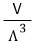
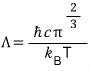
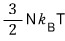
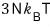
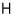
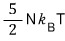
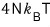
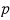
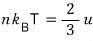
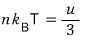
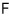
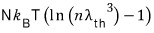
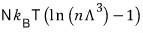
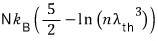
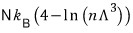
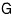
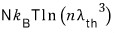
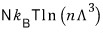



Now I’m going to talk about something a little more fun.
What I’m trying to explain with this is —
I want to think for a moment about right after the birth of our universe — Big to the Bang!!!
First, the universe is expanding.
“Redshift” is evidence of that, so the fact that the universe is expanding is acceptable enough.
Also, although the universe is expanding, surely the universe must be adiabatic!
It probably doesn’t exchange energy with the outside of the universe. (Let’s call it the law of conservation of energy.)
That is, the universe is expanding adiabatically, and “adiabatic” means
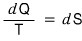
since this is so,
it means “isentropic expansion.”
But for a non-relativistic gas (something with mass, like matter),
For a non-relativistic gas, “isentropic” means "
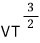
constant”.
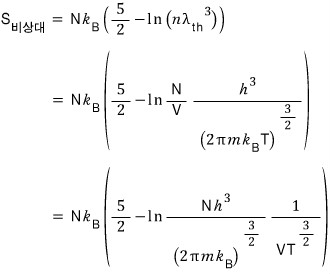
On the other hand, the entropy for an ultrarelativistic gas is
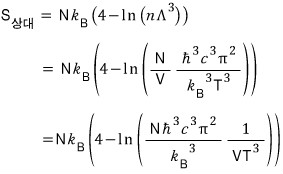
So for an ultrarelativistic gas, isentropic is represented by "
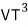
constant”.
Alright alright, as the universe expands in its early stages the gases cool down,
and this tells us that the cooling rate of a non-relativistic gas (particles) and an ultrarelativistic gas (photons) differ.
It tells us that the cooling rate of a particle-like non-relativistic gas was faster than that of a photon-like ultrarelativistic gas by the square.
That is, in the early state of the universe, matter cooled faster and its motion decreased,
and so in the universe the density of non-relativistic gas grew higher and higher compared to ultrarelativistic gas until it became the current state — that’s what we can roughly guess….
(I really hate the universe.. lo.lololol I’m totally uninterested in the universe but this was a bit intriguing so I wrote it up lololol)
Originally written in Korean on my Naver blog (2016-06). Translated to English for gdpark.blog.