Van der Waals Gas Equation of State
We crack open two ideal gas assumptions — molecular size and intermolecular forces — to build the Van der Waals equation of state from intuition up.
Now we’re going to break out of the ideal gas.
There’s a gas model that breaks two of the assumptions of the ideal gas,
and this model appears in the high school Chemistry II textbook, gets taught in freshman general chemistry in college,
and is a gas model also taught and used in many other disciplines — the “Van der Waals gas.”
So, which 2 assumptions out of the 5 ideal gas assumptions does it smash,
the first is the size of the gas. The second is the interaction force between gases.
<As something I should mention before going in for reference, here we are NOT saying we’ll deal with real gases.
We’re saying we’ll deal with the ‘Van der Waals gas,’ which is not a real gas but the ideal gas with two of its assumptions opened up.
But there’s no need to be discouraged. Even just opening those two assumptions allows us to explain very important phenomena that actually occur but couldn’t be explained with the ideal gas^^*>
So, the original ideal gas equation of state
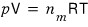
we’re going to think about how this gets transformed.
First, let’s go with intuition.
First,
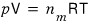
was an equation for n(mol) of ideal gas contained when the container’s volume is V, the temperature is T, and the pressure is p.
But,
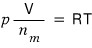
writing it like this,
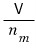
let’s think about this first.
What does it mean to divide the total volume V by n(mol)?
It will mean the volume occupied by 1 mol of ideal gas out of the total volume V.
We call this ‘molar volume’ and
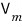
I’ll write it like this.
Now, the volume that 1 mol of ideal gas occupies
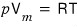
we can say is determined by this,
but the Van der Waals gas will consider the size that the ideal gas didn’t,
so
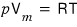
the
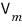
in this should be interpreted as somewhat larger.
(Please think of it as considering the size and at the same time the repulsive force resulting from it)
Because the bigger the gas size, the bigger — even slightly — the volume 1 mol occupies will be, y’know.
I thought briefly with intuition about how the ideal gas equation of state should change.
Now let’s lightly think about the interaction force with intuition. Of attractive and repulsive forces, let’s think about attraction first.
Of attractive and repulsive forces, the attractive one dominates in actual interactions between particles.
Generally, repulsion depends on
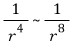
so attraction, which is proportional to the inverse square of distance, is more dominant.
So, since attraction is dominant,
in the ideal gas equation of state
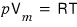
p should be interpreted as slightly underestimated!!!!
Right??? Since we’re suddenly considering an attraction we’d thought didn’t exist, the kids will rampage a bit less.
Okay so this kind of intuition is done,
let’s think a teeeeny~~~ bit more.
If the volume occupied by 1 mol is large, will the gases receive more mutual attraction?!?!?!?!
Since we’re thinking about size, it should be fine to think of our ‘molar volume’ as practically size, and let’s think like this.
Do bigger-sized things receive more mutual attraction, or do smaller-sized things receive more mutual attraction?
I think drawing two pictures will let us know simply.
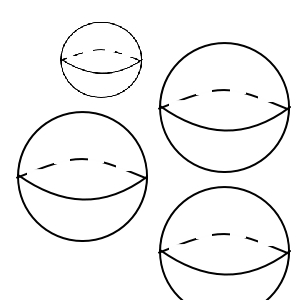

Naturally the smaller ones will receive more mutual attraction.
That is,

we’ve confirmed this.
Also, doesn’t mutual attraction also have an effect from the ’number of moles’??
The bigger the crowd, the more mutual attraction will be received.
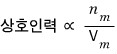
we’ve confirmed up to here.
Also also also also also, the smaller the total volume, the more mutual attraction will be received!!!
(It’s the form of breaking down what’s called density into two stages.)
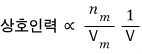
we’ve thought up to here.
Then let’s organize.
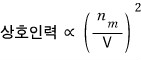
Now let’s call the coefficient that completes that proportionality into an equation a.
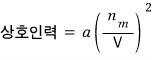
In the original ideal gas equation of state
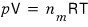
the pressure p is
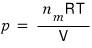
and let’s underestimate the pressure by subtracting the mutual attraction.
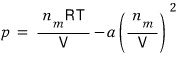
We’ve corrected by underestimating the pressure like this.
Let’s also correct the volume due to repulsion.
According to what we’ve thought about so far, the volume is
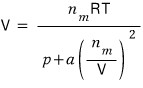
and what we’re going to do here is, we’ll add the effect that V becomes slightly larger due to repulsion.
As mentioned before, since repulsion only arises when the distance between molecules, between particles, is ridiculously close,
we’ll say that repulsion depends only on the number of moles.
And let’s call the coefficient b
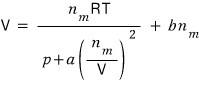
we correct just like this.
※Caution※
We are not discussing real gas right now, we’re discussing the Van der Waals gas.
Therefore the Van der Waals gas equation of state is
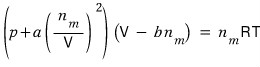
or
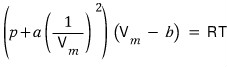
can be expressed equivalently!!! hehe
Since this is statistical mechanics time right now, let’s also derive the Van der Waals gas equation of state statistical-mechanically before moving on.
The partition function for ideal gas N is
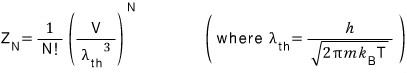
But here, like we corrected above, let’s replace V with V-bn.
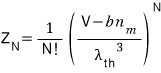
But this partition function only represents ’translational motion’!!!
Van der Waals gas, unlike the ideal gas, doesn’t only have kinetic, it has its own intrinsic force.
(That force definitely means there’s energy, and that energy will be captured by the Hamiltonian!)
That force is the mutual attraction!!!
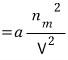
Yup, we did consider this~
Huh but?!?! It’s still ‘force,’ so it’s not energy. Let’s integrate over volume.
Since pressure × volume → energy, the volume integral of mutual ‘force’ can be called energy.
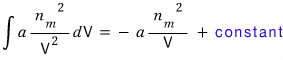
Argh, the constant term I’ve colored in blue will be the energy when the volume is 0.
Let’s say that energy is zero and not consider it… (for the protection of mental health)
It carries an energy of
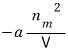
and by the definition of partition function, the partition function for this energy is
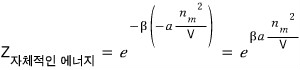
That is!!! The total partition function is the product of the partition functions in each state!!!
You remember, right?!?!

Now let’s play with that partition function a bit and derive up to the Van der Waals gas equation of state!
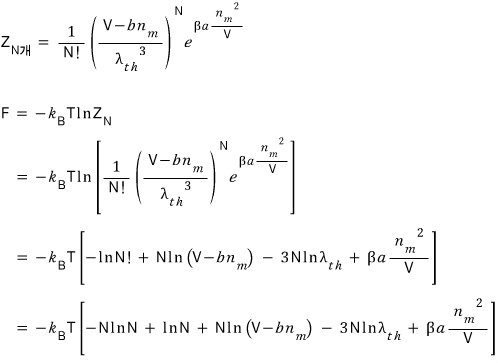
In Maxwell’s relations there was
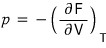
there was~, remember?
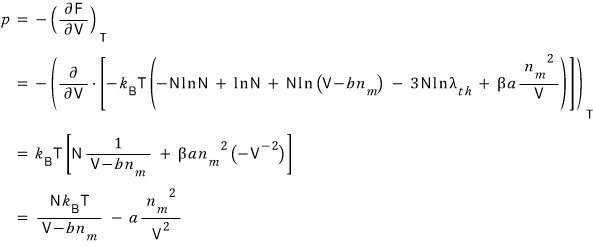
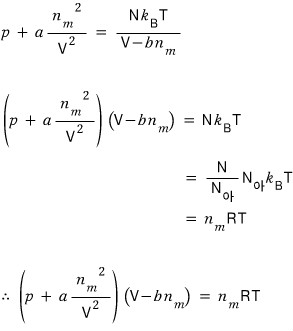
Yolololo~~~~~
Originally written in Korean on my Naver blog (2016-06). Translated to English for gdpark.blog.