Superheating and Supercooling
We dig into what actually happens below T_c — using Gibbs free energy to figure out which state a system really lands in when the P-V curve gets all weird and dented.
We’ve been looking at where the drastic changes of some system occur…
So we found the ‘critical temperature’ — the temperature at which it behaves like an ideal gas and then crosses over to the Van der Waals gas.
(Please refer to the previous posting.)
We were able to guess that at temperatures lower than that temperature (T_c), some strange phenomenon occurs that isn’t seen when thinking in terms of the ideal gas.
So now let’s think about it this way
Alright, fine. At temperatures below T_c, we get something we’ve never seen before
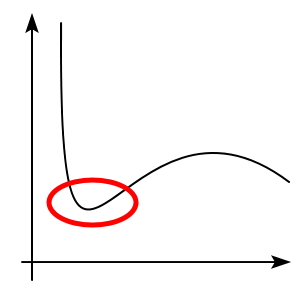
this sort of dented region appears,….
but then what the heck??????????
this is wild
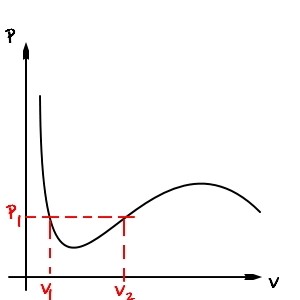
At ‘some given pressure’, should it be at V1? Or at V2?!?!?!?!?
It’s not like this is the mixed state from quantum mechanics…..
At pressure p_1, whether V1 or V2, it has to be in one of the two!?!?!
Which state on earth is it supposed to be in!!!!!!!
Here’s how we think about it!
The discussion of “where the state of the system heads toward” is usually discussed in terms of free energy
and that free energy heads toward its minimum
isn’t that how we’ve been discussing it????
So I’m going to look at the Gibbs free energy.
First, what we have is the Van der Waals equation of state
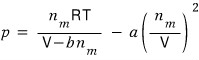
that’s what it is.
From this equation I’ll derive the Helmholtz function, and then draw out the Gibbs function.
(Wait, why look at Gibbs after deriving Helmholtz??? The reason is that the Gibbs function is a function of p and V.)
How do we derive the Helmholtz function?
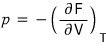
Using this relation, we’ll derive F by integrating both sides.
And we’ll derive G with G = F + pV
Start
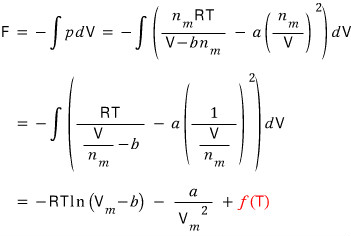
The red f(T) — since we integrated a partial derivative, there’d be a term in T, so I just slapped it in there!!
And G is
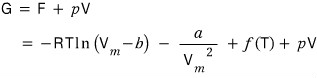
Now I’ve changed this equation into an equation meaning ‘with respect to 1 mol~’.
And I’ll let T be a temperature lower than T_c, T = αT_c (0 < α < 1).
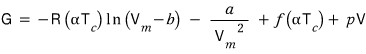
(The red function isn’t important, so it gets ignored, apparently)
If we express this function as a function of p,
it’s drawn like this, apparently.
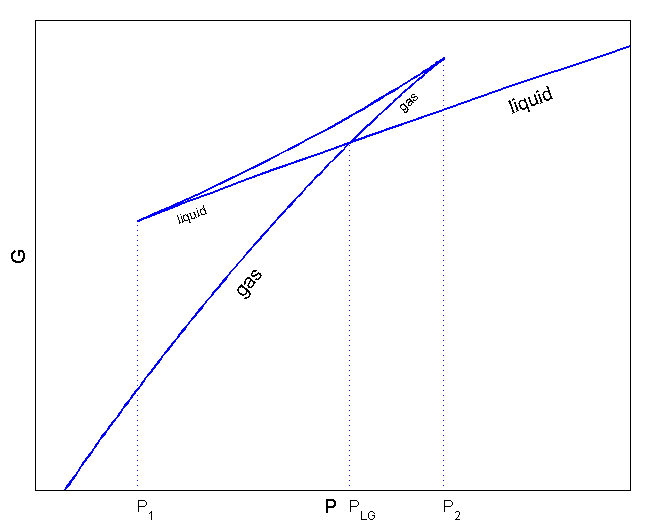
(Source: https://commons.wikimedia.org/wiki/File:Van_der_Waals_GibbsP.png)
Ahh……now we have to interpret this graph,….um…. just simply hehehehehehe
We have to take this and the graph we drew above and place them side by side and talk while comparing them.
But, I managed to find some good material.
For the case α=0.9, I found G(p) drawn with reduced variables, and a p-V graph!!haha
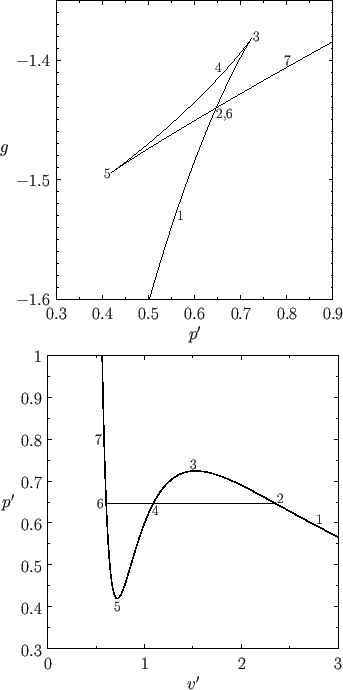
(Source: https://farside.ph.utexas.edu/teaching/sm1/Thermalhtml/node123.html)
Here’s how you plug in reduced variables.

The Van der Waals equation of state becomes this.

It transforms like this. hehehe
In the equation, a and b both disappear. Instead, the ranges of the variables change slightly.
In other words, I think you can consider it a kind of scaling.
Alright anyway so
Time for the explanation. This is going to be fun
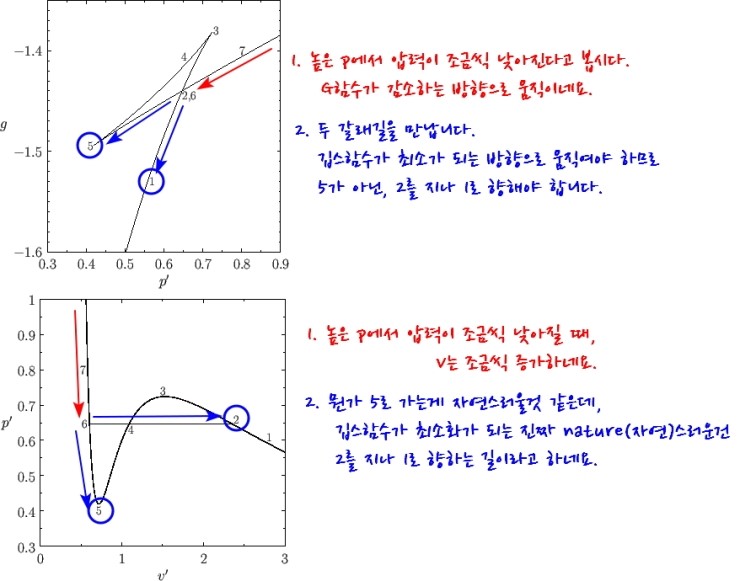
So, the “truly nature-like” path 2 — the volume suddenly diverges enormously,
what on earth does this mean?!?!?!!????!!?!
It’s exactly “phase transition”, right?!??!?!
Yup, that’s rite. Apply enormous pressure, and it becomes a liquid; ease the pressure gently, and at some moment the liquid becomes a gas….
The ‘phase transition’ that the ideal gas can’t explain — the Van der Waals model makes it possible!!!!!!!!!!!!!!!!!!!
Nownownownownownow then, does that mean the path heading to 5 is completely impossible?!?!?!
No. It’s not impossible.
Well then, let’s think about it,
Look carefully — we’ve learned that when the slope of G changes at the fork, a phase transition occurs
But if at the fork it goes straight~~~ on toward 5, that means a phase transition did not occur.
In other words, the liquid’s pressure keeps~ dropping and the gas should have gone ppyung!!!, but it’s still a liquid
We call this kind of liquid superheated.
Conversely, let’s go up. Up
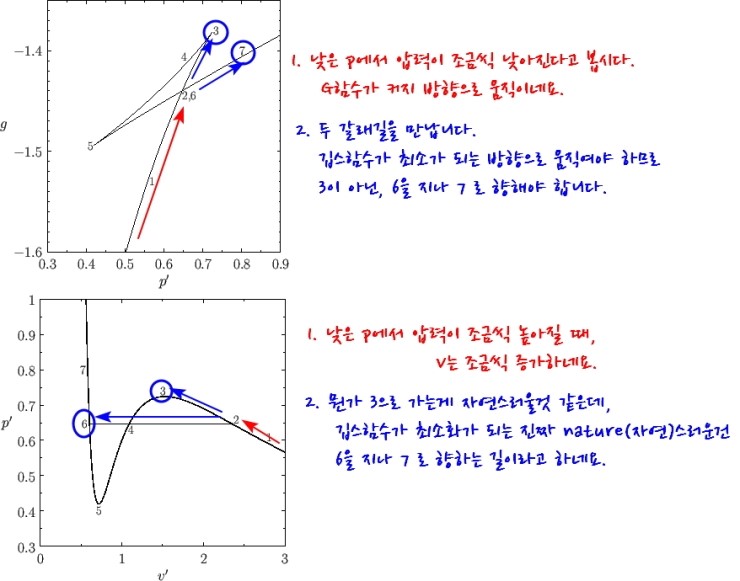
And then the gas that heads toward 3 here!!!!
As you apply more and more pressure, at some point it should go ppyung!!!!!! and if it’s a gas become a liquid, but it hasn’t yet.
This kind of state is called supercooled.
Want to see supercooled water with your own eyes?!?!?!??!?
Supercooled wadur is water that, at a temperature where it should be in the ice state, is still in the liquid state.
When it’s in this supercooled state, if you leave it alone~~~~ quietly, apparently it stays as water for a long time.
(Since it’s not in the state where the Gibbs function is minimized, it definitely won’t be a stable state.)
But@@@@@@@@@@@@@@@
if you apply a small shock to this water, that’s when the phase transition occurs in bursts!!!!!!!!!!
Whoa!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!
See it with your own eyes!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!
This kind of shock causing it to turn to ice — we can see it above our heads too.
When an airplane passes over your head, a white line appears, right?????????
That’s not the airplane spraying something as it passes,
the airplane is passing through supercooled water vapor.
Supercooled vapor is water vapor that is at a temperature where it should be water, but is still in the water vapor state.
So, due to the shock (pressure) received when the airplane passes, apparently water molecules form in bursts!!!!@@@@@@@@@@@@@@@@@
So cool!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!


Here just refer to this for fun
※Caution: fun※
Now let’s look at the case when no shock is applied.
There’s water here, and if we keep lowering the temperature of this water, it should become ice, right????
But, when we take the water and lower the temperature
the first water molecules to undergo the phase transition are, apparently, the “water molecules around impurities”.
In other words, super super super super pure water apparently doesn’t undergo phase transition easily!!!!!!
So!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!
when it’s not raining, airplanes go up into the sky,
or hot air balloons are launched
or missiles are shot up
and up into the sky, they spray dry ice
or silver iodide
— they spray these, apparently.
The reason is, by spraying a kind of impurity, to make them act as condensation nuclei hehehehe
Well then, even for super super super super super pure water with absolutely no impurities —
really nothing-but-H2O water — if the temperature drops way too much, phase transition must occur, right?
But when there are no impurities around, just what on earth is the first water molecule to undergo the phase transition…..
Nownownownownownow
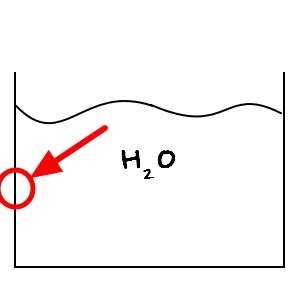
Let me zoom in and draw this part of the beaker.
No matter how precisely a beaker is made, when you look at it under a microscope
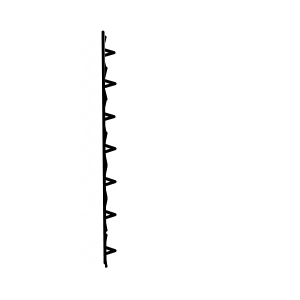
it will inevitably have a shape like this.
One of those bumps acts ‘as if it were an impurity’, apparently!!!!
The water molecules that were right next to it are the first to undergo phase transition, apparently!!!!
Originally written in Korean on my Naver blog (2016-07). Translated to English for gdpark.blog.