Maxwell's Construction
A quick look at Maxwell's construction — using ∫Vdp = 0 to pin down exactly where phase coexistence happens on the van der Waals curve.
Ah, this would be cleaner if I continued from the previous post, but I worry it’d get needlessly long,
so in this post I’ll briefly cover only Maxwell’s construction and move on.
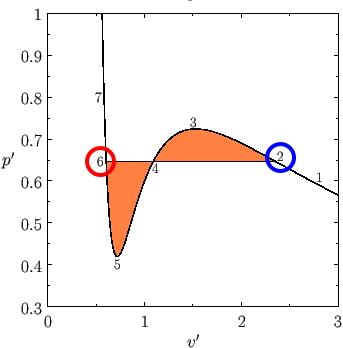
I’ll explain with this figure again.
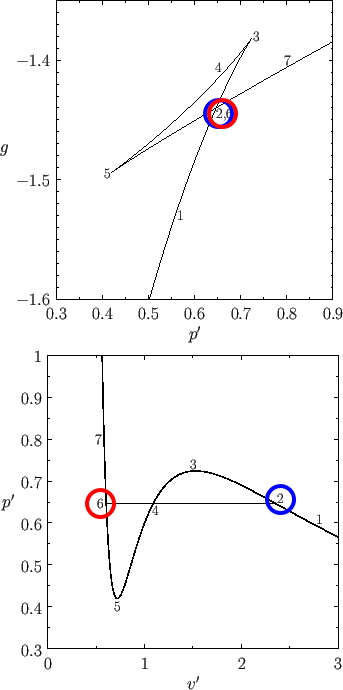
(source: https://farside.ph.utexas.edu/teaching/sm1/Thermalhtml/node123.html)
The fact that V/V_c differs at points 2 and 6 was interpreted as ’the phases are different'.
But, the Gibbs’ free energy at those two points is the same!!!!
We’ll use the above.
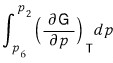
This integral, can I
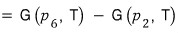
write it like this??? No problem at all, right
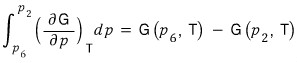
The left-hand side of this equation
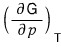
this, by the definition of the Gibbs function, is the volume V.
And if you look at the G-function part of the graph above,
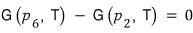
you can see this!!! Why??? Because the function values are the same!!! kk kk kk
agreed agreed byebyebye
That is,
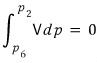
This means
the p and T that make the integral zero
are where “phase transition or phase coexistence” occurs!!!!!!!!!!!!!!!!!!!!
If you get the ordered pairs of p and T that make such phase coexistence happen and plot them, the graph apparently comes out looking like this.
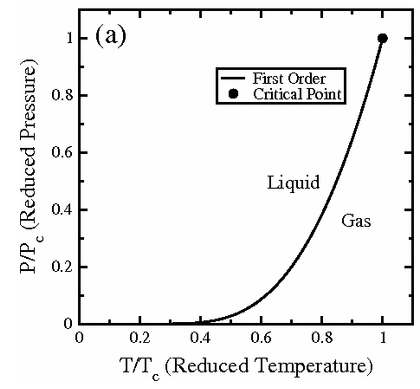
For now I’ll say just this much.
And a more detailed discussion will come in maybe the next-next post??, where I’ll cover just “phase transitions” in full detail.
Well then, zài jiàn 再见
Originally written in Korean on my Naver blog (2016-07). Translated to English for gdpark.blog.