Joule Coefficient and Joule Expansion of an Ideal Gas
Starting from the age-old puzzle of how to make things cold, we dig into Joule expansion to see why ideal gases don't cool down — but real gases do!
Suppose you’ve been given an assignment to heat something up.
You need to make some object hot. Then how would you make it hot????
You’d head straight for a lighter or a gas stove without hesitation, right. And if you don’t even have those, like the natives, you’d rub wood against wood and start a fire with friction heat.
Back in the day it was the same, and now it’s the same too — (unless it’s ultra-high temperature) you’d use fire to heat up and make hot whatever it is you want to heat.
Now then, back in modern times, what if you wanted to make some object “cold”???????????
How are you going to do it??????Are you going to leave it outside??????? Well, that means you’re trying to bring it into thermal equilibrium at room temperature 25°C,
then what are you going to do when you want to cool it below 25 degrees????
You’ll use a refrigerator?!?!???
Hmm… the refrigerator is probably the product of the question “how do we make things cold?”
That is, to the question we’re now asking, “how are we going to make things cold,” an appropriate answer would be “I’ll build this thing called a refrigerator in such-and-such a way!“
Is it starting to click now???Since olden times, making an object hot (unless it’s ultra-high temperature) was not a difficult task.
But making something cold was not easy in the past and isn’t easy now.
So the topic of this post will be “cooling.”
Alright, first we’ll look at the Joule expansion of a gas.
Not the Joule expansion of an ideal gas, but the Joule expansion of a real gas.
Because unlike the ideal-gas case, the Joule expansion of a real gas is related to ‘cooling.‘
First then, let’s look at Joule expansion.
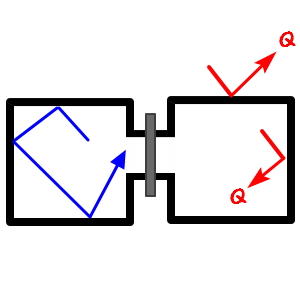
First, a synonym for Joule expansion was ‘free expansion.’
And the fact that there is no exchange of any energy in or out of the system is already a built-in assumption.
And letting it undergo (‘irreversible’) free expansion into the vacuum-connected space on the other side is what we call Joule expansion —
we’ve covered it before.
Last time we discussed entropy, and today we’re discussing temperature
Here we go.
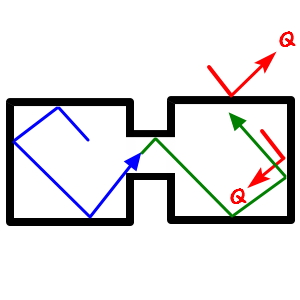
In the figure above, some particle marked with a blue arrow — lolololol — was moving with velocity v,
even if the pin-stopper dividing the two spaces opens and it crosses over to the other side
is there any room for the velocity to suddenly change from v to v’???!?!?!?!
There’s no such room anywhere,
that is,
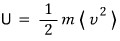
U is invariant.
But the internal energy U of an ideal gas is
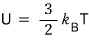
so it depends only on the temperature T
That is!!!! when an ideal gas undergoes Joule expansion, no change in temperature is induced
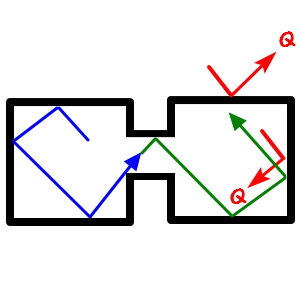
But real gases are different
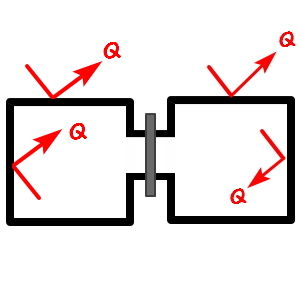
in a closed space
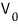
, an object moving with v, once the pin opens
and crosses over to the other side, when it sails around in there, there IS room for the velocity to change from v to v’!!!!!
Why?!?!?! Because real gases take into account the Potential that was not considered in the ideal-gas case.
Alrightalrightalrightalrightalrightalright
Since we’re thinking inside a container like this which is thermally insulated, the internal energy U is still invariant.
(You can also think of it as ‘Joule expansion is changing V while keeping U fixed.’)
But
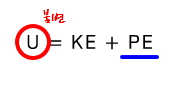
from
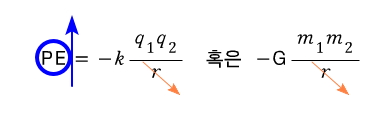
since
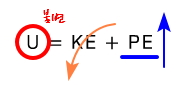
we get.
What KE ↓ means is that the change in temperature is ↓,
and we find that a real gas, not an ideal gas, undergoes Joule expansion and its temperature goes down!!!!!!!
So the Joule expansion of a real gas was related to “cooling” after all.
Now let’s organize this in a more physics-like way.
Joule Coefficient: we’re going to define something called μ_J.
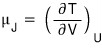
It’s defined like this, and you can take the meaning as follows.

Now, how are we going to analyze this — we’re going to use the ‘math’ we’ve used before.
Do you remember the “reciprocity theorem”?
http://gdpresent.blog.me/220587333984 The justification for using it is right here.

The Thermal·Statistical Mechanics I’ve Studied #23. Reciprocity — the reciprocal theorem, the reciprocity theorem (recip…
As I mentioned in the previous posting, I’ll prove the reciprocity theorem! Of course this will be a link-only posting hehehe. Something called x…
blog.naver.com
Then let’s get into the analysis.

> For those who forgot why the blue part is C_v (click)
└ collapse
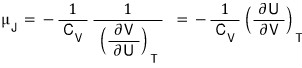
It’s organized this far.
Now I’d like that one remaining derivative to be expressed in terms of state functions we use often.

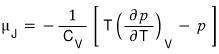
Let’s see what value μ_J takes for an ideal gas.
For simplicity, let’s suppose there’s 1 mol of an ideal gas.
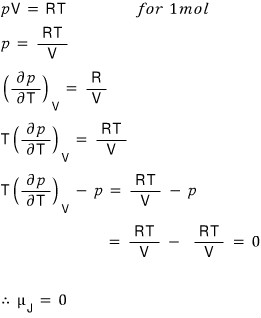
A very obvious conclusion has come out.
The Joule coefficient being 0 is
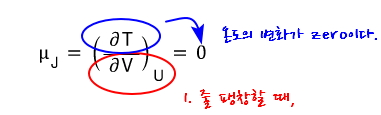
It matches the conclusion we intuitively expected way~~~ up at the beginning, so that’s nice.
Then now, let’s set aside the ideal gas and
look at what value the Joule coefficient takes for a van der Waals gas, which is a bit closer to a real gas.
For 1 mol of a van der Waals gas
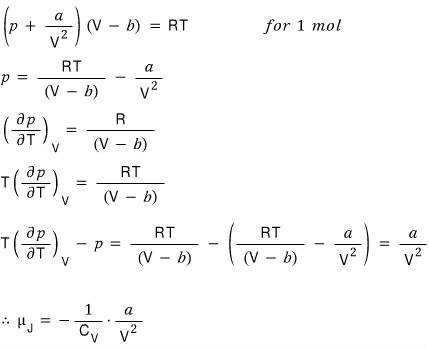
I’ll show this in an exercise later, but
the heat capacity of a van der Waals gas is

so the Joule coefficient is
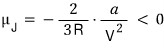
The Joule coefficient is negative!!!!?!?!?!?
So it really does cool down@@@@@@@@@@@@@@@@@@@
Then by how much does it cool?!?!?!?!

If we write it like this, I think we can state the temperature change due to Joule expansion!!!

Since V_2 > V_1, that’s why I wrote it that way.
The quantitative amount of temperature change depends on ‘a’
which is to say, it depends on the nature of the particles.
Meaning it differs by species — H, He, Na, N_2 ······.
<It depends on the attraction between gases!!!! - for Van der Waals gas>lolololol
Originally written in Korean on my Naver blog (2016-07). Translated to English for gdpark.blog.